Advanced Pyran-Based Photoresist Monomers: Technical Breakthroughs and Commercial Scalability for Semiconductor Manufacturing
Advanced Pyran-Based Photoresist Monomers: Technical Breakthroughs and Commercial Scalability for Semiconductor Manufacturing
The semiconductor industry is currently facing unprecedented challenges in pushing the boundaries of lithography resolution to meet the demands of next-generation integrated circuits. As feature sizes shrink, the performance of photoresist materials becomes the critical bottleneck determining yield and fidelity. Patent CN111777583A introduces a transformative approach to this problem by disclosing a novel degradable photoresist resin monomer synthesized from pyran-3,5-dione. This technology represents a significant leap forward in the design of acid-sensitive resin monomers, moving away from traditional linear architectures toward a more robust, cross-linkable framework. The core innovation lies in the unique molecular architecture which incorporates a central tetrahydropyran ring substituted with dual polymerizable groups. This structural motif not only enhances the mechanical and chemical stability of the resist film but also optimizes the differential solubility required for high-definition patterning. For R&D directors and procurement specialists in the electronic chemicals sector, understanding the nuances of this synthesis is vital for securing a reliable photoresist monomer supplier capable of delivering high-purity OLED material and semiconductor precursors.
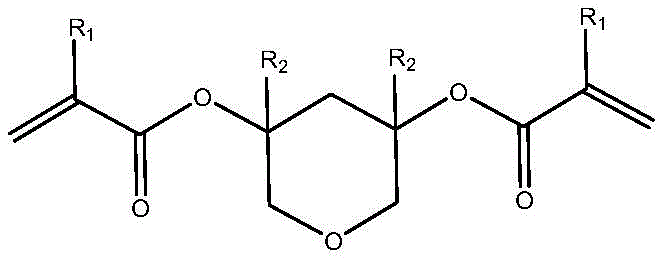
The development of advanced lithography materials requires a delicate balance between etching resistance and developability. Conventional photoresist resins often rely on linear polymers or monomers with single acid-sensitive groups, which inherently limits their performance envelope. These traditional materials frequently suffer from insufficient etching resistance, leading to pattern collapse or distortion during the aggressive plasma etching steps common in chip fabrication. Moreover, the dissolution contrast between exposed and unexposed regions in legacy systems is often marginal, resulting in line edge roughness that compromises device performance. The novel approach detailed in this patent overcomes these limitations by utilizing a bis-functional monomer that creates a three-dimensional network upon polymerization. This cross-linked structure provides superior mechanical integrity and etching resistance. Crucially, the design incorporates acid-labile linkages within the polymer backbone itself, ensuring that upon exposure and subsequent acid generation, the entire polymer chain fragments into low molecular weight species. This mechanism dramatically increases the dissolution rate in the developer, thereby sharpening the image and improving the overall resolution of the lithographic pattern.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of photoresist monomers has been dominated by methodologies that produce linear chains or lightly branched structures with limited functional density. These conventional methods often struggle to provide the necessary thermal and chemical stability required for modern dry etching processes. When a resist film lacks sufficient cross-linking density, it becomes susceptible to swelling or deformation when exposed to harsh etchants, leading to critical dimension variations. Furthermore, the reliance on single-point acid sensitivity means that the change in solubility upon exposure is gradual rather than abrupt. This lack of a sharp dissolution threshold manifests as poor edge acuity in the final developed pattern. From a manufacturing perspective, these deficiencies translate into lower yields and higher costs due to the need for thicker resist films or additional hardening steps, which complicates the process flow and increases the risk of defects in high-volume manufacturing environments.
The Novel Approach
In stark contrast, the methodology presented in patent CN111777583A leverages the unique reactivity of 2H-pyran-3,5(4H,6H)-dione to create a monomer with dual functionality. By introducing two polymerizable vinyl groups onto a rigid pyran scaffold, the resulting polymer forms a dense, three-dimensional network that is inherently more resistant to physical and chemical stress. The presence of the pyran ring also modulates the polarity of the molecule, enhancing its compatibility with standard casting solvents. This improved solubility profile ensures that the resist can be spun into uniform, defect-free films, which is a prerequisite for sub-micron patterning. Additionally, the strategic placement of the ester linkages adjacent to the quaternary carbons of the pyran ring creates a highly sensitive acid-cleavage point. This ensures that even a small amount of photo-generated acid can trigger a cascade of backbone scission events, leading to a massive increase in solubility. This binary switch-like behavior is essential for achieving the high contrast ratios needed for advanced node fabrication.
Mechanistic Insights into Grignard Addition and Esterification
The synthesis of this high-performance monomer proceeds through a concise two-step sequence that maximizes atom economy and minimizes waste. The first step involves a nucleophilic addition reaction where a Grignard reagent attacks the carbonyl groups of the starting pyran-dione. This transformation is highly sensitive to reaction conditions, requiring strict temperature control between 0°C and 30°C to prevent side reactions such as enolization or over-addition. The use of anhydrous tetrahydrofuran or diethyl ether as the solvent is critical to maintain the activity of the organomagnesium species. The resulting intermediate is a diol, where the original ketone functionalities have been converted into tertiary alcohols bearing the desired alkyl or cycloalkyl substituents. This step effectively sets the steric bulk and lipophilicity of the final monomer, parameters that are crucial for tuning the glass transition temperature and dissolution properties of the final resist polymer.
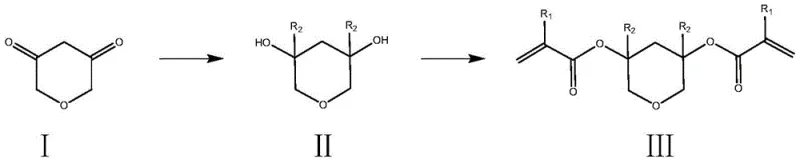
Following the formation of the diol intermediate, the second step is an esterification reaction that installs the polymerizable acrylate or methacrylate groups. This reaction is conducted under basic conditions, typically using triethylamine or pyridine as a proton scavenger to neutralize the hydrochloric acid byproduct generated from the acid chloride. The reaction temperature is carefully managed, ranging from 0°C to 70°C, to balance reaction kinetics with the stability of the acid-sensitive product. The choice of solvent in this step, such as THF, toluene, or chloroform, influences the solubility of the growing oligomers and prevents premature precipitation which could trap impurities. The mechanistic precision of this esterification ensures that both hydroxyl groups are converted efficiently, yielding a bis-functional monomer with high purity. Any residual mono-ester or unreacted diol would act as a chain terminator or defect site in the final polymer, so rigorous purification protocols, including aqueous workups and column chromatography, are employed to ensure the stringent purity specifications required for electronic grade materials.
How to Synthesize Degradable Photoresist Resin Monomer Efficiently
The practical implementation of this synthesis route offers a streamlined pathway for producing high-value electronic chemicals. The process begins with the preparation of the Grignard reagent, followed by its controlled addition to the cyclic diketone precursor. After quenching and isolation of the diol intermediate, the final esterification step converts the hydrophilic diol into the hydrophobic, polymerizable monomer. This sequence is robust and adaptable, allowing for the variation of R groups to fine-tune material properties for specific lithography applications. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and purification techniques, are outlined in the guide below to assist process engineers in replicating these results.
- Perform a Grignard reaction between 2H-pyran-3,5(4H,6H)-dione and an alkyl or cycloalkyl Grignard reagent in anhydrous THF or ether at 0-30°C to form the diol intermediate.
- Quench the reaction with water or dilute acid, separate the organic layer, and purify the intermediate diol via extraction and column chromatography.
- React the purified diol intermediate with acryloyl chloride or methacryloyl chloride in the presence of a base like triethylamine at 0-70°C to yield the final resin monomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits. The reliance on commodity chemicals such as pyran-dione, alkyl halides, and magnesium metal ensures a stable and diversified supply base, reducing the risk of raw material shortages. The two-step nature of the synthesis significantly reduces the number of unit operations compared to multi-step convergent syntheses often seen in complex fine chemical manufacturing. This simplification directly correlates to lower capital expenditure requirements for reactor trains and reduced labor costs for operation and monitoring. Furthermore, the mild reaction conditions eliminate the need for specialized high-pressure or cryogenic equipment, allowing for production in standard stainless steel reactors found in most fine chemical facilities. These factors combine to create a manufacturing process that is not only cost-effective but also resilient to market fluctuations.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts, which are often required for cross-coupling reactions in alternative monomer syntheses, results in substantial cost savings. By utilizing Grignard chemistry, the process avoids the need for costly palladium or nickel catalysts and the associated ligand systems. Additionally, the absence of heavy metals simplifies the downstream purification process, removing the need for expensive scavenging resins or complex filtration steps to meet trace metal specifications. The high atom efficiency of the addition reaction means that a larger proportion of the starting mass ends up in the final product, minimizing waste disposal costs. Qualitative analysis suggests that the simplified workflow and reduced reagent costs contribute to a significantly lower cost of goods sold, making this monomer a financially attractive option for high-volume photoresist production.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are widely available from multiple global suppliers, mitigating the risk of single-source dependency. 2H-pyran-3,5-dione and common alkyl halides are produced at scale for various industrial applications, ensuring consistent quality and availability. The robustness of the reaction conditions means that the process is less susceptible to minor variations in utility supply, such as cooling water temperature fluctuations, which can sometimes disrupt more sensitive catalytic processes. This operational stability translates into more predictable lead times and higher on-time delivery rates for customers. For supply chain planners, this reliability is crucial for maintaining continuous production lines in the fast-paced semiconductor industry where downtime is exceptionally costly.
- Scalability and Environmental Compliance: The synthesis is inherently scalable, having been demonstrated effectively from gram to kilogram scales in the patent examples without loss of efficiency. The use of common organic solvents like THF and ethyl acetate facilitates established solvent recovery and recycling protocols, aligning with modern green chemistry principles and environmental regulations. The aqueous workup steps generate waste streams that are relatively straightforward to treat compared to those containing heavy metals or persistent organic pollutants. This ease of waste management reduces the environmental footprint of the manufacturing process and lowers compliance costs. As regulatory pressures on chemical manufacturing intensify globally, having a process that is both scalable and environmentally benign provides a significant competitive advantage for long-term sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this novel photoresist monomer. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on performance metrics and process capabilities. Understanding these details is essential for stakeholders evaluating the integration of this material into existing fabrication workflows or supply chains.
Q: What are the primary advantages of this pyran-based monomer over traditional linear photoresist monomers?
A: This monomer features two polymerizable double bonds that enable the formation of a three-dimensional cross-linked network during polymerization. This structure significantly enhances etching resistance compared to linear polymers. Furthermore, the acid-labile nature of the backbone allows for precise degradation upon exposure, drastically improving the dissolution contrast between exposed and unexposed areas, which leads to superior pattern resolution and reduced edge roughness.
Q: Is the synthesis process scalable for industrial production of electronic chemicals?
A: Yes, the synthesis relies on robust and well-established chemical transformations: a Grignard addition followed by a standard esterification. The reaction conditions are mild, operating between 0°C and 70°C, which minimizes energy consumption and safety risks associated with high-temperature processes. The use of common solvents like THF and ether further facilitates solvent recovery and recycling, making the process highly amenable to large-scale commercial manufacturing.
Q: How does the pyran ring structure influence the solubility and processing of the final photoresist?
A: The inclusion of the oxygen-containing pyran ring imparts higher lipophilicity to the monomer compared to purely cycloalkane-based structures. This enhanced lipid solubility ensures that the resulting polymer resin exhibits excellent solubility in standard ester solvents used during the spin-coating process. Better solubility translates to more uniform film formation and fewer defects, which is critical for achieving high yields in semiconductor fabrication.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Photoresist Resin Monomer Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance monomers play in the advancement of semiconductor technology. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. We are committed to delivering materials that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our facility is equipped to handle the specific safety and handling requirements of reactive intermediates like Grignard reagents and acid chlorides, guaranteeing a safe and efficient production environment for your specialized electronic chemicals.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your project goals. Let us be your partner in driving innovation and efficiency in the electronic materials sector.
