Advanced Synthesis of High-Resolution Photoresist Resin Monomers for Semiconductor Manufacturing
The semiconductor industry is constantly pushing the boundaries of miniaturization, driving an urgent demand for photoresist materials with superior resolution and etching resistance. Patent CN111100007A introduces a breakthrough class of photoresist resin monomers synthesized from carboxylic acid compounds, specifically designed to overcome the limitations of traditional linear polymers. Unlike conventional monomers that rely solely on deprotection of acid-sensitive groups, these novel structures incorporate an acetal linkage capable of cleaving the polymer backbone upon exposure. This fundamental shift in chemical architecture allows for the generation of small molecular fragments during development, drastically improving the contrast and edge roughness of the final lithographic pattern. The general structural formula, designated as Formula I, represents a versatile platform where R can be varied to tune solubility and reactivity profiles for specific lithography applications.
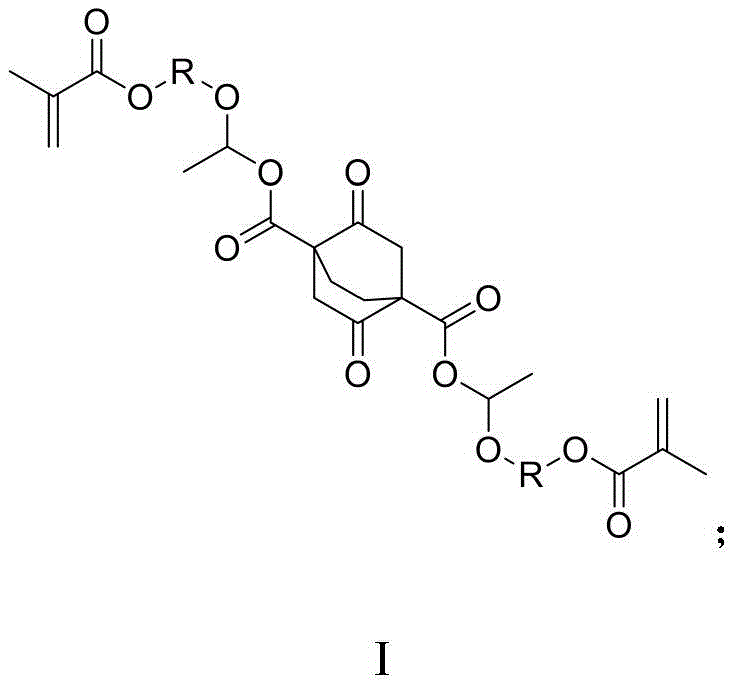
As a reliable electronic chemical supplier, understanding the nuances of this molecular design is critical for delivering high-purity OLED material and semiconductor precursors. The innovation lies not just in the final structure but in the robust synthetic pathway that enables its production. By utilizing readily available vinyl ether alcohols and methacryloyl chloride, the process ensures a stable supply chain while maintaining rigorous quality standards. This patent provides a comprehensive roadmap for manufacturing these advanced materials, addressing the critical need for cost reduction in electronic chemical manufacturing without compromising on the performance metrics required for next-generation chip fabrication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional photoresist resins are typically formed by copolymerizing monomers that possess a single acid-sensitive group, resulting in a linear polymer chain. While effective for larger feature sizes, these linear structures face significant challenges as feature sizes shrink to the nanometer scale. The primary limitation is that the solubility difference between exposed and unexposed areas relies entirely on the conversion of a protecting group, leaving the polymer backbone intact. This often leads to insufficient dissolution rates in the developer and poor contrast, manifesting as line edge roughness (LER) that can ruin circuit functionality. Furthermore, many conventional resins lack the necessary thermal stability and etching resistance required for aggressive plasma etching processes, limiting their utility in advanced node manufacturing. The inability of the main chain to break down into smaller, more soluble fragments restricts the resolution potential of the material.
The Novel Approach
The novel approach detailed in the patent fundamentally alters the dissolution mechanism by incorporating an acetal structure directly into the monomer design. Upon exposure to light and subsequent acid generation from the photoacid generator (PAG), this acetal linkage is cleaved, effectively cutting the polymer main chain into smaller segments. This depolymerization-like effect dramatically increases the solubility of the exposed regions in the alkaline developer, creating a sharp contrast with the unexposed areas. Additionally, the incorporation of a rigid 2,5-dioxobicyclo[2.2.2]octane core provides exceptional etching resistance, protecting the underlying substrate during pattern transfer. The synthetic route is elegantly simple, avoiding complex catalytic cycles in favor of straightforward esterification and addition reactions, which facilitates easier purification and higher overall yields.
Mechanistic Insights into Esterification and Thermal Addition
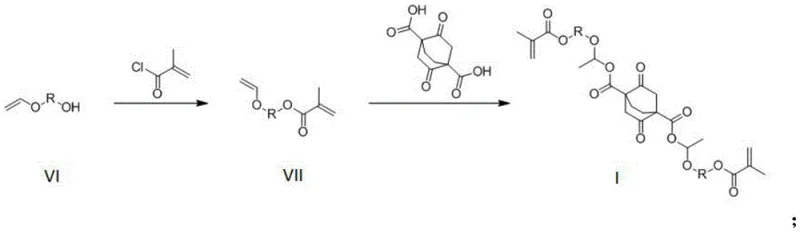
The synthesis mechanism is a two-stage process that prioritizes atom economy and operational simplicity. The first stage involves the esterification of a vinyl ether alcohol (Formula VI) with methacryloyl chloride. This reaction is typically conducted in a solvent like dichloromethane with triethylamine acting as an acid scavenger to neutralize the generated HCl. The nucleophilic attack of the hydroxyl group on the acid chloride carbonyl carbon forms the methacrylate ester intermediate (Formula VII). This step is crucial as it installs the polymerizable double bond and preserves the acid-labile vinyl ether moiety, which will later form the acetal linkage. Careful temperature control at 0°C initially prevents premature polymerization or side reactions, ensuring the integrity of the sensitive vinyl group.
The second stage is a thermal addition reaction where two equivalents of the intermediate react with one equivalent of 2,5-dioxobicyclo[2.2.2]octane-1,4-dicarboxylic acid. This step creates the central bicyclic core flanked by the acid-sensitive arms. The reaction proceeds at elevated temperatures, typically between 75°C and 85°C, facilitating the formation of the ester bonds between the carboxylic acid groups of the bicyclic core and the hydroxyl groups generated or present in the system, ultimately forming the acetal-bridged structure. The use of inhibitors like p-tert-butylcatechol is essential during this heating phase to prevent radical polymerization of the methacrylate groups. The result is a symmetrical monomer with high functionality, ready for copolymerization into high-performance photoresist resins.
How to Synthesize Photoresist Resin Monomer Efficiently
The practical execution of this synthesis requires precise stoichiometric control and adherence to specific thermal profiles to maximize yield and purity. The patent outlines a clear protocol starting from commercially available vinyl glycol ethers, making the barrier to entry for production relatively low. Operators must ensure anhydrous conditions during the esterification phase to prevent hydrolysis of the acid chloride. Following the formation of the intermediate, the subsequent coupling with the bicyclic dicarboxylic acid demands careful monitoring of temperature to drive the equilibrium towards the product while minimizing thermal degradation. For a detailed breakdown of the specific molar ratios, solvent volumes, and workup procedures, please refer to the standardized synthesis guide below.
- Perform esterification between a vinyl ether alcohol compound and methacryloyl chloride in the presence of triethylamine and dichloromethane at 0°C to room temperature to obtain the intermediate.
- Conduct an addition reaction by mixing the intermediate with 2,5-dioxobicyclo[2.2.2]octane-1,4-dicarboxylic acid and an inhibitor like p-tert-butylcatechol.
- Stir the mixture at 75-85°C for approximately 1 hour, followed by reduced pressure distillation to isolate the final high-purity photoresist resin monomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers substantial strategic benefits over more complex organometallic or high-pressure catalytic processes. The reliance on commodity chemicals such as methacryloyl chloride, triethylamine, and dichloromethane ensures that raw material sourcing is robust and less susceptible to geopolitical supply disruptions. The absence of expensive transition metal catalysts eliminates the need for costly metal scavenging steps and rigorous heavy metal testing, which are often bottlenecks in pharmaceutical and electronic grade chemical production. This simplification of the downstream processing directly translates to reduced operational expenditures and faster batch turnover times.
- Cost Reduction in Manufacturing: The process utilizes mild reaction conditions and avoids the use of precious metal catalysts, which significantly lowers the direct material costs associated with production. The high yields reported in the examples, exceeding 80% for the intermediate and over 82% for the final product, indicate an efficient conversion of raw materials into value-added products. Furthermore, the purification methods described, such as column chromatography and distillation, are standard unit operations that do not require specialized or custom-built equipment, thereby reducing capital expenditure requirements for new production lines.
- Enhanced Supply Chain Reliability: The starting materials, specifically the vinyl ether alcohols and the bicyclic dicarboxylic acid, are derived from established chemical supply chains with high availability. By avoiding exotic reagents that might have long lead times or single-source dependencies, manufacturers can maintain a consistent inventory level and reduce the risk of production stoppages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further stabilizing the supply output and ensuring reliable delivery schedules to downstream photoresist formulators.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-ton commercial production without fundamental changes to the chemistry. The use of common organic solvents allows for established recovery and recycling protocols, minimizing waste generation and aligning with strict environmental regulations. The elimination of heavy metals from the process stream simplifies wastewater treatment and reduces the environmental footprint of the manufacturing facility, making it a sustainable choice for long-term production of high-purity electronic chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photoresist monomer technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process engineers and procurement specialists. Understanding these details is essential for evaluating the feasibility of integrating this material into existing manufacturing workflows.
Q: How does the acetal structure in this monomer improve photoresist resolution?
A: The acetal structure undergoes acid-catalyzed cleavage upon exposure, disconnecting the polymer main chain into small fragments. This significantly increases solubility in the developer and reduces edge roughness, leading to superior pattern resolution compared to linear polymers.
Q: What are the key advantages of using 2,5-dioxobicyclo[2.2.2]octane derivatives?
A: The multi-ring bicyclic structure provides exceptional etching resistance, which is critical for deep sub-micron processing. Additionally, the polyester-based nature enhances solubility in fat-soluble solvents, ensuring uniform spin-coating performance.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process utilizes standard organic synthesis techniques such as esterification and thermal addition without requiring exotic catalysts or extreme pressures. The workup involves simple filtration and distillation, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Photoresist Resin Monomer Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance monomers play in the advancement of semiconductor technology. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to mass manufacturing is seamless. We are committed to delivering materials with stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our capability to handle complex organic syntheses, including the esterification and addition reactions described in CN111100007A, positions us as a strategic partner for your electronic material needs.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your next-generation photoresist development projects.
