Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-One Pharmaceutical Intermediates
Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-One Pharmaceutical Intermediates
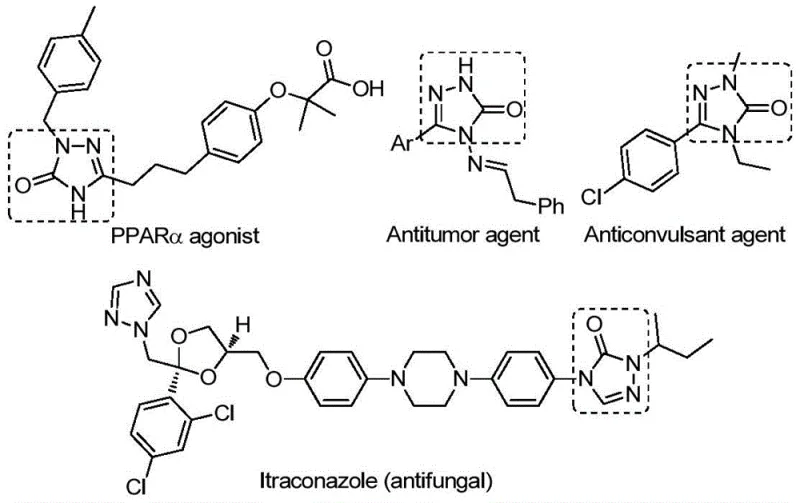
The pharmaceutical industry continuously seeks robust synthetic methodologies to access complex heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. A significant breakthrough in this domain is documented in Chinese Patent CN112538054B, which discloses a highly efficient preparation method for 1,2,4-triazole-3-one compounds. This specific class of five-membered nitrogen-containing heterocycles is ubiquitous in medicinal chemistry, known for exhibiting a diverse array of biological activities including antifungal, anti-inflammatory, antitumor, antiviral, and anticonvulsant properties. As illustrated in the structural diversity of bioactive molecules below, these cores are critical for developing PPARα agonists, antitumor agents, and antifungal drugs like Itraconazole.
The innovation presented in this patent addresses long-standing challenges in the synthesis of these valuable intermediates. By leveraging a transition metal palladium-catalyzed carbonylation tandem cyclization reaction, the method transforms inexpensive and readily available starting materials—specifically chlorinated hydrazones and sodium azide—into high-value products. For procurement managers and R&D directors alike, this represents a pivotal shift towards more cost-effective and reliable pharmaceutical intermediate supplier strategies, enabling the streamlined production of complex heterocycles essential for modern drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthetic landscape for 1,2,4-triazole-3-one compounds has been fraught with inefficiencies that hinder large-scale manufacturing. Traditional literature reports describe several common pathways, such as the cyclization of benzoyl hydrazide with urea under potassium hydroxide action, or the tandem cyclization of hydrazides with isocyanates. Other methods involve the condensation of thioamides with hydrazines at elevated temperatures or the reaction of acyl isocyanates with monosubstituted hydrazines. These legacy processes suffer from severe drawbacks, including harsh reaction conditions that demand specialized equipment,冗杂 (complicated) multi-step sequences that reduce overall throughput, and the necessity for pre-activation of reaction substrates which increases raw material costs. Furthermore, these conventional routes often exhibit low yields and narrow substrate scopes, limiting their utility for generating diverse libraries of analogs required for structure-activity relationship (SAR) studies in early-stage drug development.
The Novel Approach
In stark contrast, the novel methodology outlined in the patent introduces a streamlined, one-pot catalytic system that circumvents the pitfalls of prior art. The core of this innovation lies in the use of chlorohydrazones, which are easily synthesized from corresponding acid chlorides, hydrazines, triphenylphosphine, and carbon tetrachloride, serving as stable and accessible precursors. The reaction employs a sophisticated palladium catalytic system comprising Pd2(dba)3 and the bidentate ligand Xantphos, alongside TFBen (phenyl formate derivative) acting as a safe carbon monoxide substitute. This setup allows for a smooth carbonylation tandem cyclization at moderate temperatures of 100°C to 120°C. The result is a process with exceptional substrate compatibility, capable of tolerating various functional groups such as alkyl, alkoxy, halogen, and trifluoromethyl substituents on both aryl and alkyl chains. This versatility ensures that a wide range of 1,2,4-triazole-3-one derivatives can be accessed efficiently, supporting the rapid iteration needed in pharmaceutical research.
Mechanistic Insights into Pd-Catalyzed Carbonylation Tandem Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific targets. The reaction is believed to initiate with the oxidative addition of the palladium(0) catalyst into the carbon-chlorine bond of the chlorohydrazone substrate, generating a reactive divalent palladium intermediate. Subsequently, the TFBen additive undergoes thermal decomposition to release carbon monoxide in situ, which then inserts into the carbon-palladium bond to form an acyl-palladium species. This key intermediate reacts with sodium azide to generate an acyl azide compound. Following this, a Curtius rearrangement occurs, converting the acyl azide into a highly reactive isocyanate intermediate. The final step involves an intramolecular nucleophilic addition where the hydrazine nitrogen attacks the isocyanate carbon, closing the ring to yield the final 1,2,4-triazole-3-one scaffold. This elegant cascade minimizes the formation of side products and maximizes atom economy.

Impurity control is another critical aspect managed by this catalytic system. The choice of ligand, specifically Xantphos, plays a vital role in stabilizing the palladium center and preventing the formation of palladium black or other inactive species that could lead to incomplete reactions or difficult-to-remove metal residues. Additionally, the use of aprotic solvents like 1,4-dioxane is paramount; polar aprotic solvents such as dimethyl sulfoxide (DMSO) or dimethylformamide (DMF) were found to be detrimental to the reaction efficiency. By strictly adhering to the optimized solvent and ligand system, manufacturers can achieve high purity profiles, reducing the burden on downstream purification steps and ensuring the final product meets stringent quality specifications required for active pharmaceutical ingredients (APIs).
How to Synthesize 1,2,4-Triazole-3-One Efficiently
To implement this synthesis effectively, precise adherence to the reaction parameters is essential. The process begins with the careful weighing of the palladium catalyst and ligand to ensure the correct molar ratio, typically 1:2 for Pd2(dba)3 to Xantphos. The reaction mixture, containing the chlorohydrazone, sodium azide, and the CO source in 1,4-dioxane, must be heated uniformly to maintain the kinetic energy required for the tandem cyclization. While the patent demonstrates success on a millimole scale, the simplicity of the operation suggests straightforward scalability. The detailed standardized synthesis steps, including specific workup procedures like filtration and silica gel mixing followed by column chromatography, are outlined in the guide below to ensure reproducibility and high yield.
- Combine Pd2(dba)3 catalyst, Xantphos ligand, TFBen carbon monoxide substitute, chlorohydrazone substrate, and sodium azide in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to a temperature between 100°C and 120°C and maintain stirring for a duration of 16 to 30 hours to ensure complete conversion.
- Upon completion, perform filtration and silica gel mixing followed by column chromatography purification to isolate the high-purity 1,2,4-triazole-3-one product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary advantage lies in the significant simplification of the supply chain for raw materials. Chlorohydrazones and sodium azide are commodity chemicals that are widely available from multiple global suppliers, reducing the risk of single-source dependency. Furthermore, the elimination of hazardous gaseous carbon monoxide cylinders in favor of the solid TFBen substitute enhances workplace safety and reduces regulatory compliance costs associated with handling toxic gases. This shift not only streamlines logistics but also lowers the barrier for entry for facilities that may lack specialized gas handling infrastructure, thereby expanding the pool of potential contract manufacturing organizations (CMOs) capable of executing this chemistry.
- Cost Reduction in Manufacturing: The economic implications of this method are profound. By utilizing cheap and easy-to-obtain starting materials, the direct material cost is drastically reduced compared to routes requiring pre-activated or exotic reagents. Moreover, the high reaction efficiency and yields, which can reach up to 96% for certain substrates as demonstrated in the patent data, mean less waste and higher throughput per batch. The operational simplicity also translates to lower labor costs and reduced energy consumption, as the reaction proceeds effectively at standard heating temperatures without the need for cryogenic conditions or extreme pressures. These factors collectively contribute to a substantially lower cost of goods sold (COGS) for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: In an era where supply chain resilience is paramount, this method offers superior reliability. The robustness of the catalytic system against various functional groups means that supply disruptions for specific niche precursors are less likely to halt production, as the chemistry is adaptable to a wide range of substituted chlorohydrazones. The use of common solvents like 1,4-dioxane further ensures that solvent supply remains stable. Additionally, the shortened reaction timeline compared to multi-step traditional methods accelerates the production cycle, allowing for faster turnaround times and more responsive inventory management to meet fluctuating market demands for API intermediates.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this process is highly favorable. The tandem nature of the reaction reduces the number of isolation and purification steps, thereby minimizing solvent usage and waste generation. The post-treatment process involving simple filtration and chromatography is well-established and easily scalable from gram to kilogram scales. The avoidance of heavy metal contaminants through effective ligand stabilization simplifies the purification process, ensuring that the final product meets rigorous heavy metal limits without expensive scavenging treatments. This alignment with green chemistry principles not only reduces environmental impact but also facilitates smoother regulatory approvals and audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on reaction optimization and practical application. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing workflows.
Q: What represents the primary advantage of this palladium-catalyzed method over traditional synthesis routes?
A: Unlike traditional methods requiring harsh conditions or pre-activated substrates, this novel approach utilizes cheap chlorohydrazones and operates under milder conditions with significantly higher reaction efficiency and broader substrate compatibility.
Q: Which solvent system provides the optimal conversion rates for this carbonylation reaction?
A: While various organic solvents can dissolve the raw materials, aprotic solvents are preferred. Specifically, 1,4-dioxane has been identified as the optimal solvent, facilitating higher conversion rates compared to polar aprotic solvents like DMF or DMSO.
Q: Is this synthetic route scalable for industrial production of API intermediates?
A: Yes, the patent explicitly states that the method is operationally simple and has been successfully scaled to the 1 mmol level and beyond, demonstrating strong potential for industrial application and medium-scale production in drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole-3-One Supplier
As the global demand for complex heterocyclic intermediates continues to rise, partnering with an experienced CDMO is critical for success. NINGBO INNO PHARMCHEM stands at the forefront of this sector, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing palladium-catalyzed reactions to ensure maximum yield and purity, leveraging state-of-the-art rigorous QC labs to verify that every batch meets stringent purity specifications. We understand the critical nature of API intermediates in the drug development timeline and are committed to delivering consistent quality that supports your clinical and commercial goals.
We invite you to collaborate with us to leverage this advanced synthetic technology for your projects. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target molecules. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary candidates. Let us help you accelerate your path to market with reliable, high-quality chemical solutions.
