Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-One Pharmaceutical Intermediates
Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-One Pharmaceutical Intermediates
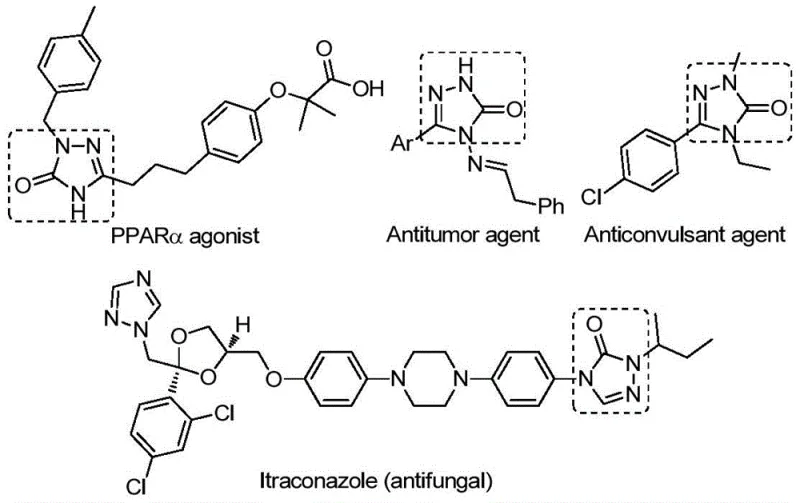
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for privileged scaffolds that exhibit diverse biological activities. Patent CN112538054B discloses a groundbreaking preparation method for 1,2,4-triazole-3-one compounds, a core structure prevalent in numerous bioactive molecules ranging from antifungal agents like Itraconazole to antitumor and anticonvulsant drugs. This technology represents a significant leap forward in heterocyclic chemistry, offering a streamlined, palladium-catalyzed carbonylation tandem cyclization strategy. By utilizing readily available chlorohydrazones and sodium azide as starting materials, this method circumvents the limitations of classical synthesis, providing a reliable pathway for generating high-purity pharmaceutical intermediates. For R&D directors and procurement specialists, understanding this innovation is crucial for optimizing supply chains and reducing the cost of goods sold (COGS) in complex API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazole-3-one ring has been fraught with synthetic challenges that hinder efficient scale-up. Traditional methodologies often rely on the cyclization of benzoyl hydrazides with urea under strong basic conditions, or the reaction of hydrazides with isocyanates. These conventional routes suffer from severe drawbacks, including the requirement for harsh reaction conditions, such as high temperatures and strong bases, which can degrade sensitive functional groups. Furthermore, many existing protocols involve multi-step sequences requiring the pre-activation of substrates, leading to lower overall yields and increased waste generation. The use of toxic isocyanates or thioamides also poses significant safety and environmental hazards, complicating regulatory compliance and increasing disposal costs. Consequently, these factors limit the substrate scope and make the commercial production of diverse derivatives economically unviable for many fine chemical manufacturers.
The Novel Approach
In stark contrast, the method described in patent CN112538054B introduces a highly efficient, one-pot transition metal-catalyzed protocol. This novel approach leverages a palladium-catalyzed carbonylation tandem cyclization reaction, utilizing chlorohydrazones and sodium azide as the primary building blocks. A key innovation is the use of TFBen (1,3,5-tricarboxylic acid phenol ester) as a safe and effective carbon monoxide surrogate, eliminating the need for handling hazardous high-pressure CO gas. The reaction proceeds smoothly in common organic solvents like 1,4-dioxane at moderate temperatures (100-120°C). This strategy not only simplifies the operational procedure but also dramatically expands the substrate compatibility, allowing for the introduction of various alkyl, aryl, and heteroaryl groups. As illustrated in the general reaction scheme below, this method transforms simple precursors into valuable heterocycles with high atom economy.

Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
For technical teams evaluating process feasibility, understanding the mechanistic underpinnings is vital for troubleshooting and optimization. The reaction likely initiates with the oxidative addition of the palladium(0) catalyst into the carbon-chlorine bond of the chlorohydrazone substrate, forming a reactive divalent palladium intermediate. Simultaneously, the CO surrogate, TFBen, decomposes under thermal conditions to release carbon monoxide in situ. This generated CO then inserts into the carbon-palladium bond, creating an acyl-palladium species. Subsequently, the interaction with sodium azide leads to the formation of an acyl azide intermediate, which undergoes a Curtius rearrangement to generate an isocyanate species. The final step involves an intramolecular nucleophilic attack by the hydrazone nitrogen onto the isocyanate carbon, closing the ring to form the stable 1,2,4-triazole-3-one core. This elegant cascade minimizes the accumulation of unstable intermediates, thereby enhancing the overall purity of the crude product.
From an impurity control perspective, this mechanism offers distinct advantages over stepwise syntheses. By conducting the carbonylation and cyclization in a single pot, the exposure of reactive intermediates to external quenchers or moisture is minimized. The use of specific ligands, such as Xantphos, stabilizes the palladium center and facilitates the reductive elimination step, ensuring high turnover numbers. Moreover, the mild reaction conditions prevent the decomposition of sensitive functional groups like halogens or alkoxy substituents, which are often prone to side reactions in harsher traditional methods. This results in a cleaner reaction profile, reducing the burden on downstream purification processes such as column chromatography or recrystallization, which is a critical factor for maintaining cost efficiency in large-scale production.
How to Synthesize 1,2,4-Triazole-3-One Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these valuable intermediates. The process begins by charging a reaction vessel with the palladium catalyst system, specifically tris(dibenzylideneacetone)dipalladium and the Xantphos ligand, along with the CO source TFBen. To this mixture, the chlorohydrazone substrate and sodium azide are added in an appropriate organic solvent. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are provided in the guide below to ensure reproducibility and safety during laboratory or pilot-scale operations.
- Combine palladium catalyst (Pd2(dba)3), ligand (Xantphos), carbon monoxide substitute (TFBen), chlorohydrazone, and sodium azide in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to 100-120°C and maintain stirring for 16 to 30 hours to allow the carbonylation and cyclization to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 1,2,4-triazole-3-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates into tangible strategic benefits. The shift from complex, multi-step traditional methods to this streamlined catalytic process addresses several critical pain points in the sourcing of pharmaceutical intermediates. By simplifying the synthesis, manufacturers can reduce lead times and mitigate the risks associated with supply chain disruptions caused by hard-to-source reagents. The reliance on commodity chemicals rather than specialized, custom-synthesized precursors enhances supply security and price stability.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the use of inexpensive and widely available starting materials. Chlorohydrazones and sodium azide are commodity chemicals with established global supply chains, unlike the specialized isocyanates or activated hydrazides required in older methods. Furthermore, the elimination of high-pressure carbon monoxide equipment reduces capital expenditure (CAPEX) and operational safety costs. The high reaction efficiency and yield mean less raw material is wasted, directly lowering the variable cost per kilogram of the final API intermediate. This cost structure allows for more competitive pricing in the global market without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. The process tolerates a wide range of functional groups, meaning that a single manufacturing line can be adapted to produce various derivatives simply by changing the starting chlorohydrazone. This flexibility reduces the need for dedicated production lines for each specific analog, optimizing asset utilization. Additionally, the avoidance of hazardous gases like CO simplifies logistics and storage requirements, removing a significant bottleneck often found in carbonylation chemistries. This ensures a more consistent and reliable flow of materials to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden inefficiencies, but this method is designed with scalability in mind. The use of standard solvents like 1,4-dioxane and moderate temperatures facilitates easy heat transfer and mixing in large reactors. From an environmental standpoint, the atom economy of the tandem reaction is superior to stepwise approaches, resulting in less chemical waste. The simplified workup procedure, involving filtration and standard purification, reduces the volume of solvent waste generated. This aligns with increasingly stringent environmental regulations and corporate sustainability goals, making it an attractive option for green chemistry initiatives within the pharmaceutical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for stakeholders evaluating this technology for potential integration into their manufacturing portfolios.
Q: What are the key advantages of this palladium-catalyzed method over traditional synthesis?
A: Unlike traditional methods that require harsh conditions, pre-activated substrates, or toxic isocyanates, this novel approach utilizes cheap chlorohydrazones and sodium azide under mild conditions with high atom economy and broad functional group tolerance.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses commercially available reagents, avoids high-pressure carbon monoxide gas by using TFBen as a safe CO surrogate, and operates in standard organic solvents, making it highly scalable for industrial applications.
Q: What types of substituents are compatible with this reaction?
A: The method demonstrates excellent substrate compatibility, tolerating various groups including alkyl (t-Bu, n-Bu), aryl (phenyl, naphthyl), and heteroaryl (furyl) groups, as well as halogen and alkoxy substituents on the aromatic rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole-3-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient intermediate synthesis plays in the broader drug development lifecycle. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are committed to delivering high-purity 1,2,4-triazole-3-one compounds that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our facility is equipped to handle the specific requirements of palladium-catalyzed reactions, including advanced containment systems and recovery protocols for precious metals.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Whether you require custom synthesis of novel analogs or large-scale supply of established intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how we can optimize your supply chain and accelerate your time to market.
