Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-one Pharmaceutical Intermediates
Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-one Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic methodologies for constructing nitrogen-containing heterocycles, particularly the 1,2,4-triazole-3-one scaffold, which serves as a critical pharmacophore in numerous bioactive molecules. As detailed in patent CN112538054B, a groundbreaking preparation method has been developed that leverages transition metal palladium catalysis to achieve efficient carbonylation tandem cyclization. This technology represents a significant leap forward for manufacturers acting as a reliable pharmaceutical intermediate supplier, addressing long-standing challenges in yield and operational safety. The disclosed method utilizes readily available chlorohydrazones and sodium azide as starting materials, reacting them in the presence of a palladium catalyst and a carbon monoxide substitute within an organic solvent at elevated temperatures. By shifting away from traditional, hazardous gas-phase carbonylation, this process offers a streamlined pathway to high-purity heterocyclic compounds essential for antifungal, antitumor, and anticonvulsant drug development.
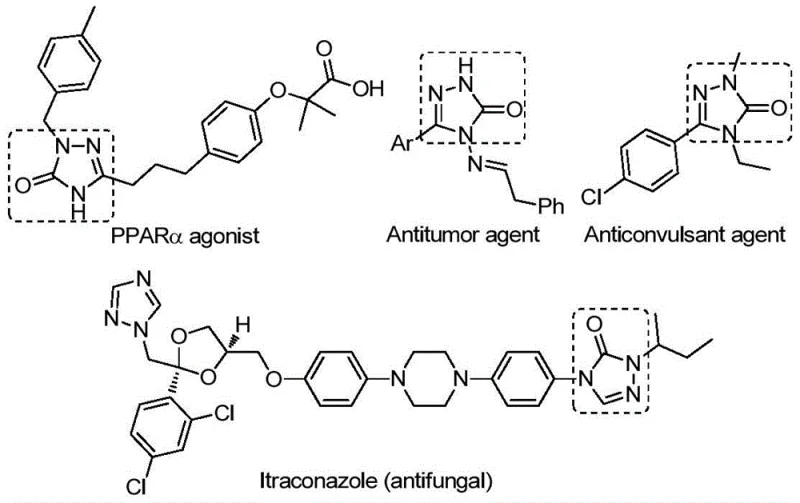
The strategic importance of the 1,2,4-triazole-3-one core cannot be overstated, as it appears in a diverse array of therapeutic agents ranging from PPARα agonists to potent antifungal drugs like Itraconazole. The ability to synthesize these structures with high fidelity and minimal impurities is paramount for R&D directors focused on accelerating drug discovery timelines. The patent highlights that this novel approach not only simplifies the operational workflow but also expands the chemical space accessible to medicinal chemists by accommodating a wide variety of substituents on the hydrazone backbone. This versatility ensures that supply chain heads can rely on a single, adaptable platform for producing multiple analogues, thereby reducing the complexity of managing diverse synthetic routes for different pipeline candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazole-3-one ring system has been plagued by significant synthetic inefficiencies that hinder cost-effective manufacturing. Traditional protocols often rely on the cyclization of benzoyl hydrazides with urea under strongly basic conditions, or the tandem reaction of hydrazides with isocyanates, which frequently necessitates the use of toxic and volatile reagents. Another common route involves the condensation of thioamides with hydrazines at excessively high temperatures, leading to thermal degradation of sensitive functional groups and the formation of complex impurity profiles that are difficult to purge. Furthermore, methods utilizing acyl isocyanates often require pre-activation steps that add unnecessary unit operations, increasing both the time and capital expenditure required for production. These legacy processes typically suffer from narrow substrate scope, meaning that slight modifications to the molecular structure can cause the reaction to fail completely, forcing process chemists to restart method development from scratch for every new analogue.
The Novel Approach
In stark contrast, the methodology described in CN112538054B introduces a sophisticated palladium-catalyzed carbonylation strategy that elegantly bypasses these historical bottlenecks. By employing chlorohydrazones as the electrophilic partner and sodium azide as the nitrogen source, the reaction proceeds through a well-defined catalytic cycle that constructs the triazole ring and installs the ketone functionality in a single pot. The use of TFBen (1,3,5-tricarboxylic acid phenol ester) as a solid carbon monoxide surrogate is a masterstroke of process safety, eliminating the need for high-pressure CO gas infrastructure while ensuring a steady, controlled release of the carbonyl source. This innovation allows the reaction to proceed smoothly at 100°C in common aprotic solvents like 1,4-dioxane, achieving high conversion rates without the extreme conditions that degrade product quality. The result is a versatile platform capable of synthesizing derivatives substituted with alkyl, aryl, and heteroaryl groups, providing a robust solution for cost reduction in pharmaceutical intermediate manufacturing.

Mechanistic Insights into Pd-Catalyzed Carbonylation Tandem Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The reaction initiates with the oxidative addition of the low-valent palladium catalyst, specifically Pd2(dba)3 coordinated with the bidentate ligand Xantphos, into the carbon-chlorine bond of the chlorohydrazone substrate. This step generates a reactive organopalladium(II) intermediate, which is stabilized by the wide bite angle of the Xantphos ligand, preventing premature catalyst deactivation. Subsequently, carbon monoxide, released in situ from the thermal decomposition of TFBen, inserts into the carbon-palladium bond to form an acyl-palladium species. This acyl intermediate then undergoes nucleophilic attack by the azide ion, leading to the formation of an acyl azide compound. The thermal energy of the reaction drives a Curtius rearrangement, converting the acyl azide into a highly reactive isocyanate intermediate. Finally, an intramolecular nucleophilic addition occurs where the pendant hydrazine nitrogen attacks the isocyanate carbon, closing the five-membered ring and yielding the final 1,2,4-triazole-3-one product with the regeneration of the active palladium catalyst.
From an impurity control perspective, this mechanism offers distinct advantages over non-catalytic thermal cyclizations. The specificity of the palladium insertion ensures that side reactions such as homocoupling of the hydrazine or polymerization of the isocyanate are minimized. The use of a solid CO source prevents local concentrations of carbon monoxide from becoming too high, which could otherwise lead to over-carbonylation or the formation of urea byproducts. Moreover, the mild reaction temperature of 100°C is sufficient to drive the Curtius rearrangement without promoting the decomposition of thermally labile substituents like halogens or esters that might be present on the aromatic rings. This precise control over the reaction trajectory results in a cleaner crude profile, significantly reducing the burden on downstream purification units and ensuring that the final high-purity pharmaceutical intermediates meet stringent regulatory specifications for heavy metals and organic impurities.
How to Synthesize 1,2,4-Triazole-3-one Efficiently
The practical execution of this synthesis is designed for scalability and ease of operation, making it ideal for both laboratory optimization and pilot plant campaigns. The protocol involves charging a reaction vessel with the palladium catalyst system, the solid CO source, and the substrates in a suitable solvent, followed by heating to reflux. The simplicity of the reagent setup allows for rapid screening of different chlorohydrazone derivatives to build structure-activity relationship libraries. For detailed operational parameters regarding stoichiometry, specific temperature ramps, and workup procedures, please refer to the standardized guide below.
- Charge a reaction vessel with Pd2(dba)3 catalyst, Xantphos ligand, TFBen (CO source), chlorohydrazone substrate, and sodium azide in an aprotic solvent like 1,4-dioxane.
- Heat the reaction mixture to 100-120°C and maintain stirring for 16 to 30 hours to allow for oxidative addition, CO insertion, and cyclization.
- Upon completion, filter the mixture, concentrate, and purify the crude residue via silica gel column chromatography to isolate the target 1,2,4-triazole-3-one.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates into tangible strategic benefits that extend beyond mere chemical yield. The shift from hazardous gaseous reagents to stable solid surrogates fundamentally alters the risk profile of the manufacturing process, allowing for production in facilities that may not be equipped for high-pressure gas handling. This flexibility opens up a wider network of potential contract manufacturing organizations (CMOs), enhancing supply chain resilience and reducing dependency on specialized, high-cost vendors. Furthermore, the use of commodity chemicals like sodium azide and easily synthesized chlorohydrazones ensures that raw material costs remain low and predictable, shielding the project from volatile market fluctuations associated with exotic reagents.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide equipment drastically reduces capital expenditure (CAPEX) requirements for reactor setup and maintenance. By utilizing a solid CO surrogate, the process avoids the need for specialized gas delivery systems, leak detection infrastructure, and the associated safety audits, leading to substantial operational cost savings. Additionally, the high atom economy of the tandem cyclization minimizes waste generation, lowering the costs associated with solvent recovery and waste disposal. The robustness of the catalyst system also implies that lower catalyst loadings may be feasible upon further optimization, further driving down the cost of goods sold (COGS) for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including chlorohydrazones and sodium azide, are commercially available from multiple global suppliers, mitigating the risk of single-source bottlenecks. Unlike proprietary reagents that may have long lead times or limited availability, these commoditized inputs ensure a steady flow of materials necessary for continuous production. The stability of the reagents also simplifies logistics, as they do not require cryogenic shipping or special hazard classifications that delay customs clearance. This reliability is critical for maintaining just-in-time inventory levels and ensuring that clinical trial materials are delivered without interruption.
- Scalability and Environmental Compliance: The reaction conditions are inherently scalable, having been demonstrated to work efficiently from milligram to gram scales with consistent results. The use of 1,4-dioxane, a common industrial solvent, facilitates easy integration into existing solvent recovery loops, aligning with green chemistry principles by reducing the environmental footprint of the synthesis. The straightforward workup procedure, involving simple filtration and chromatography, avoids complex distillation steps that are energy-intensive and prone to thermal hazards. This ease of scale-up ensures that the transition from process development to commercial manufacturing is seamless, supporting the commercial scale-up of complex pharmaceutical intermediates with minimal technical risk.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for stakeholders evaluating this technology for their supply chains.
Q: What are the safety advantages of using TFBen over carbon monoxide gas?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) acts as a solid carbon monoxide surrogate, releasing CO in situ under heating. This eliminates the need for handling high-pressure CO gas cylinders, significantly reducing explosion risks and simplifying reactor requirements for industrial scale-up.
Q: How does this method improve substrate compatibility compared to traditional routes?
A: Traditional methods often require harsh basic conditions or high temperatures that degrade sensitive functional groups. This Pd-catalyzed protocol operates under milder thermal conditions (100°C) and tolerates diverse substituents including halogens, alkyl groups, and heterocycles like furan, enabling the synthesis of complex drug scaffolds.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes commercially available reagents like sodium azide and chlorohydrazones. The workup involves simple filtration and standard chromatography, avoiding complex distillation or cryogenic steps, which facilitates translation from gram-scale laboratory synthesis to multi-kilogram commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating the delivery of life-saving medicines to patients. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. We are committed to delivering stringent purity specifications through our rigorous QC labs, which are equipped with state-of-the-art analytical instrumentation to verify the identity and quality of every batch. By leveraging the efficiencies of the Pd-catalyzed carbonylation route, we can offer competitive pricing and reliable lead times for your 1,2,4-triazole-3-one requirements.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to evaluate the feasibility of new analogues, we are ready to provide specific COA data and route feasibility assessments. Partner with us to secure a sustainable and efficient supply of high-quality intermediates that empower your drug development pipeline.
