Advanced Nickel-Catalyzed Synthesis of Biphenyl Dianhydride for Industrial Polyimide Applications
The global demand for high-performance polyimide materials continues to surge, driven by applications in flexible electronics and aerospace engineering, yet the supply chain for their critical monomer, Biphenyl Dianhydride (BPDA), has long been constrained by costly and hazardous synthesis routes. Patent CN113582954B introduces a transformative methodology that leverages metal coordination chemistry to overcome these historical bottlenecks, specifically utilizing aminohalogenated phthalimide monomers in a nickel-catalyzed coupling system. This innovation represents a paradigm shift from traditional palladium-dependent processes, offering a robust pathway for cost reduction in polyimide precursor manufacturing while simultaneously enhancing operational safety profiles. By ingeniously exploiting the coordination capability of the amino group within the substrate, the process effectively manages inorganic by-products that typically plague reductive coupling reactions. For R&D directors and procurement strategists alike, this technology signals a move towards more sustainable and economically viable production of high-purity electronic chemical intermediates, addressing both the financial and technical hurdles that have limited market expansion.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of BPDA has relied heavily on noble metal catalysts such as palladium, which impose exorbitant raw material costs and necessitate complex recovery systems to meet purity standards. Alternative routes involving Grignard reagents require the use of low-boiling, highly flammable solvents like tetrahydrofuran or diethyl ether, creating significant safety liabilities and explosion risks during large-scale operations. Furthermore, conventional nickel or palladium-catalyzed couplings using zinc powder often suffer from severe efficiency losses due to the adsorption of inorganic salt by-products, such as zinc halides, onto the surface of the active metal reducing agent. This surface passivation phenomenon drastically reduces the effective contact area between the catalyst and the reductant, forcing manufacturers to employ massive excesses of zinc powder—often exceeding 400% of stoichiometric requirements—to drive the reaction to completion. The resulting accumulation of solid waste not only complicates downstream processing but also poses fire hazards upon exposure to oxygen, rendering many traditional methods unsuitable for modern, environmentally conscious commercial scale-up of complex polymer additives.
The Novel Approach
In stark contrast, the methodology disclosed in CN113582954B utilizes a sophisticated design where the starting material itself acts as a ligand to modulate the reaction environment. By employing aminohalogenated phthalimides, the process ensures that the generated zinc halide by-products are sequestered into soluble organic-metal complexes rather than precipitating as passivating layers on the zinc surface. This fundamental change in reaction dynamics allows for the use of significantly lower catalyst loadings and near-quantitative consumption of the reducing agent, thereby streamlining the entire workflow. The reaction proceeds smoothly in high-boiling, non-protic solvents such as N,N-dimethylacetamide (DMAc) or N-methylpyrrolidone (NMP) at moderate temperatures ranging from 50°C to 100°C, eliminating the need for cryogenic conditions or hazardous ether solvents. As illustrated in the reaction scheme below, this approach facilitates a clean conversion to the biphenyl derivative, which is subsequently hydrolyzed and dehydrated to yield the target dianhydride with exceptional efficiency.
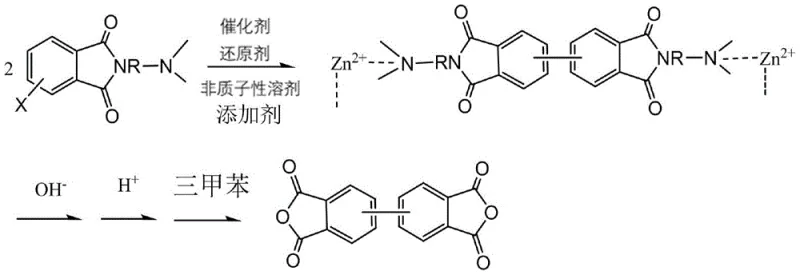
This streamlined pathway not only improves yield metrics, with reported isolation yields for the intermediate reaching 90-95%, but also drastically simplifies the workup procedure by avoiding the filtration of massive quantities of spent metal sludge. For supply chain managers, this translates to a more predictable and reliable production cycle with reduced downtime for reactor cleaning and waste disposal.
Mechanistic Insights into Nickel-Catalyzed Coupling with Metal Coordination
The core scientific breakthrough of this patent lies in the intricate interplay between the substrate's functional groups and the inorganic species generated during the reduction phase. In standard reductive coupling scenarios, the accumulation of zinc chloride (ZnCl2) creates a physical barrier on the zinc powder, effectively insulating the reducing agent from the catalytic cycle and stalling the reaction. However, the presence of the tertiary amino group on the phthalimide monomer introduces a powerful chelating effect that alters the solubility profile of these salts. The nitrogen lone pair coordinates with the zinc cation (Zn2+), forming a soluble organometallic complex that remains dissolved in the polar aprotic solvent matrix. This coordination prevents the nucleation and deposition of insoluble zinc halide crystals on the metal surface, thereby maintaining the high surface energy and reactivity of the zinc powder throughout the duration of the reaction.
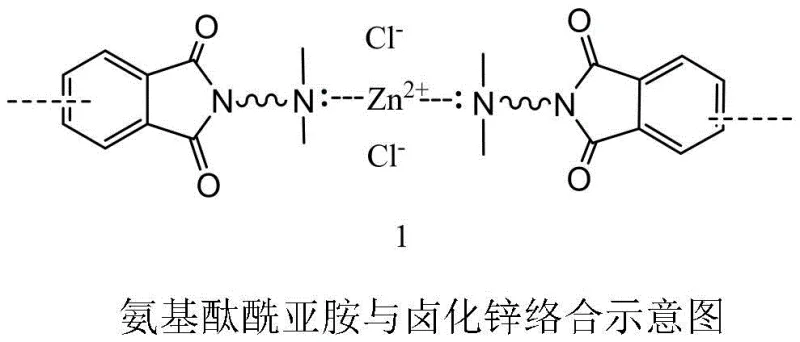
From a mechanistic standpoint, this solubilization effect ensures that the nickel catalyst maintains uninterrupted access to the active reducing sites, facilitating a continuous electron transfer process essential for the carbon-carbon bond formation. The stability of these intermediates also contributes to superior impurity control, as the homogeneous nature of the by-products prevents the formation of occluded impurities that are difficult to remove during crystallization. Consequently, the final product exhibits a cleaner impurity profile, which is critical for applications in semiconductor packaging where ionic contamination must be minimized. This level of mechanistic control allows for the precise tuning of reaction parameters, such as the molar ratio of additive to nickel salt (1:1 to 7:1), to optimize the kinetics without compromising the structural integrity of the sensitive phthalimide ring system.
How to Synthesize Biphenyl Dianhydride Efficiently
The synthesis protocol outlined in the patent provides a clear, three-stage roadmap for producing high-quality BPDA suitable for advanced material applications. The process begins with the catalytic coupling of the functionalized monomer, followed by a rigorous purification of the bis-imide intermediate, and concludes with hydrolysis and dehydration to form the anhydride. Detailed operational parameters, including specific solvent volumes and temperature ramps, are critical for maximizing the reported yields of 90% to 98% in the final steps. The following guide summarizes the standardized synthetic approach derived from the patent examples, serving as a foundational reference for process engineers looking to implement this technology.
- Perform the coupling reaction by mixing aminohalogenated phthalimide monomers with a nickel salt catalyst and zinc reducing agent in an aprotic solvent like DMAc at 50-100°C for 1-24 hours.
- Purify the resulting bisaminobiphenyl tetracarboximide intermediate through recrystallization using toluene or xylene to achieve high purity levels.
- Hydrolyze the purified imide using aqueous NaOH, acidify to precipitate the tetra-acid, and finally dehydrate using trimethylbenzene reflux to obtain the final dianhydride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement leaders evaluating the viability of this technology, the economic implications extend far beyond simple raw material substitution; they represent a structural optimization of the entire value chain. The elimination of expensive palladium catalysts and the drastic reduction in zinc waste generation directly correlate to a leaner cost structure, making the final product more competitive in price-sensitive markets. Furthermore, the shift away from volatile ether solvents to high-flash-point amide solvents significantly lowers insurance premiums and safety compliance costs associated with hazardous material handling. This process inherently supports a more resilient supply chain by reducing dependency on scarce noble metals and minimizing the logistical burden of hazardous waste transport and disposal.
- Cost Reduction in Manufacturing: The transition from palladium to nickel catalysis removes one of the most significant variable cost drivers in traditional BPDA synthesis, while the near-quantitative usage of the reducing agent eliminates the need for purchasing and disposing of excessive zinc powder. By avoiding the generation of large volumes of solid metal waste, facilities can reduce their expenditure on waste treatment services and extend the lifespan of their filtration equipment. Additionally, the ability to recover and recycle high-boiling solvents like DMAc further enhances the overall economic efficiency of the plant, ensuring that operating margins remain robust even during periods of raw material price volatility.
- Enhanced Supply Chain Reliability: Utilizing widely available base metals like nickel and zinc mitigates the geopolitical and supply risks often associated with precious metal sourcing, ensuring a more stable production schedule. The robustness of the reaction conditions, which tolerate moderate temperatures and do not require stringent moisture exclusion to the degree of Grignard chemistry, allows for more flexible manufacturing windows and reduced batch failure rates. This reliability is crucial for maintaining consistent delivery timelines to downstream polymer manufacturers who depend on a steady flow of monomer to keep their own production lines running without interruption.
- Scalability and Environmental Compliance: The absence of pyrophoric waste residues simplifies the scale-up process, as reactors do not require specialized inert atmosphere protocols for discharge, thereby accelerating batch turnover times. From an environmental perspective, the reduction in solid waste and the use of less volatile organic compounds align with increasingly stringent global regulations regarding industrial emissions and hazardous waste disposal. This compliance advantage future-proofs the manufacturing asset against tightening environmental legislation, securing long-term operational continuity without the need for costly retrofits or process modifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this nickel-catalyzed coordination method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and safety considerations. Understanding these nuances is essential for technical teams assessing the feasibility of integrating this route into existing production frameworks.
Q: How does the amino-coordination mechanism improve reaction efficiency?
A: The amino group on the monomer coordinates with by-product zinc halides, keeping them soluble in the reaction medium. This prevents the passivation of the zinc reducing agent surface, allowing for near-quantitative utilization of the reductant and significantly lowering catalyst requirements.
Q: What are the safety advantages over traditional Grignard-based methods?
A: This method utilizes high-boiling, non-flammable aprotic solvents like DMAc or NMP instead of highly volatile and explosive ethers or THF required for Grignard reagents. This drastically reduces fire hazards and simplifies solvent recovery processes.
Q: What purity levels can be achieved with this purification protocol?
A: Through optimized recrystallization steps using xylenes or toluene, the intermediate bis-imides can achieve purity levels between 95% and 99%, ensuring the final dianhydride meets stringent specifications for electronic grade polyimides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biphenyl Dianhydride Supplier
As the industry pivots towards more efficient and sustainable chemical manufacturing, NINGBO INNO PHARMCHEM stands at the forefront of translating these advanced patent technologies into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this nickel-catalyzed process are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped to analyze trace metal residuals and isomeric distributions, guaranteeing that every batch of BPDA meets the exacting standards required for high-performance polyimide films and electronic substrates.
We invite forward-thinking partners to collaborate with us on optimizing this synthesis route for their specific volume requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current supply chain constraints. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this innovative method can enhance your product competitiveness and operational stability.
