Revolutionizing Polyimide Precursors: Efficient Synthesis and Separation of Biphenyl Dianhydride Isomers
The landscape of high-performance polyimide precursors is undergoing a significant transformation driven by the innovations disclosed in patent CN113603663B. This pivotal intellectual property introduces a groundbreaking method for preparing biphenyl dianhydride (BPDA) isomers through ultrasonic-assisted catalytic coupling, addressing one of the most persistent challenges in electronic material synthesis: the efficient separation of structural isomers. Traditionally, the production of asymmetric isomers like 3,4'-BPDA has been hindered by the相似 properties of reaction intermediates, leading to exorbitant costs and limited availability for advanced aerospace and semiconductor applications. By leveraging a sophisticated molecular design strategy combined with green catalytic principles, this technology enables the distinct isolation of 3,3'-, 3,4'-, and 4,4'-BPDA with exceptional purity levels exceeding 98 percent. For industry leaders seeking a reliable electronic chemical supplier, understanding this technological leap is crucial for securing supply chains for next-generation heat-resistant polymers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of biphenyl dianhydride isomers relied heavily on palladium-catalyzed coupling or non-selective nickel processes using mixed chlorophthalic acid derivatives. These conventional routes suffered from a critical flaw: the resulting intermediate mixtures possessed nearly identical physical and chemical properties, making chromatographic or crystallization-based separation economically unviable on an industrial scale. Specifically, previous attempts often yielded inseparable mixtures of biphenyl tetracarboxylic acids, forcing manufacturers to accept low-purity products or incur massive costs to isolate the valuable asymmetric 3,4'-isomer. Furthermore, the reliance on precious metal catalysts like palladium introduced significant cost volatility and supply chain risks, while traditional mechanical stirring often failed to maintain the activity of zinc reducing agents due to surface passivation by inorganic byproducts. These inefficiencies created a bottleneck where the price of specialized isomers could reach ten times that of standard 4,4'-BPDA, limiting their adoption in cost-sensitive high-volume applications.
The Novel Approach
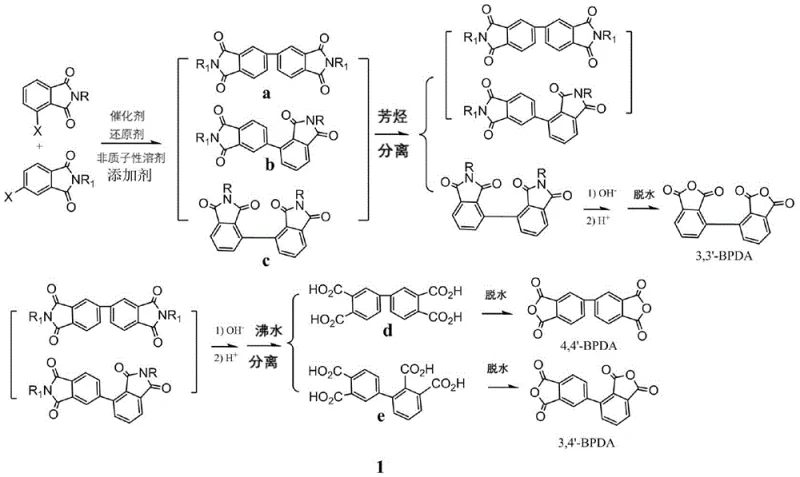
The methodology outlined in the patent circumvents these historical barriers through a clever manipulation of solubility characteristics at the intermediate stage. By reacting specific combinations of N-aryl (or methyl)-4-chlorophthalimide and N-propyl (or isopropyl)-3-chlorophthalimide, the process generates bis-imine intermediates with deliberately engineered solubility differences in 2-THF solvent. As illustrated in the reaction scheme below, the 3,3'-bis-imine intermediate exhibits high solubility, allowing it to be easily separated from the insoluble 3,4'- and 4,4'-isomers via simple hot filtration. This initial physical separation is followed by a hydrolysis step where the resulting tetra-acids display divergent solubility in hot water, enabling the final isolation of pure isomers without complex chromatography. This strategic approach not only simplifies the purification workflow but also drastically reduces the consumption of organic solvents and energy, representing a paradigm shift in cost reduction in polyimide precursor manufacturing.
Mechanistic Insights into Ultrasonic-Assisted Nickel Catalysis
At the heart of this synthesis lies a robust nickel-catalyzed reductive coupling mechanism that is significantly enhanced by ultrasonic irradiation. The reaction employs inexpensive nickel salts, such as nickel chloride or bromide, in conjunction with metallic zinc as the stoichiometric reducing agent. Under standard mechanical stirring, the efficiency of zinc powder often diminishes rapidly as the surface becomes coated with insoluble zinc chloride (ZnCl2) byproduct, which blocks active sites and halts the electron transfer necessary for the coupling reaction. However, the application of ultrasonic waves at frequencies and power levels between 40 to 500 watts creates intense cavitation effects within the 2-THF solvent. These microscopic implosions effectively scour the surface of the zinc particles, continuously removing the passivating ZnCl2 layer and exposing fresh, reactive metal surfaces to the catalytic cycle. This dynamic maintenance of catalyst activity ensures that the reductive coupling proceeds to completion within a short timeframe of 0.5 to 3 hours at moderate temperatures ranging from 60 to 100 degrees Celsius.
Beyond the activation of the reducing agent, the ultrasonic field plays a vital role in controlling the impurity profile of the final product. The enhanced mass transfer facilitated by acoustic streaming ensures a homogeneous distribution of reactants and catalyst species, minimizing the formation of higher-order oligomers or homocoupling side products that often plague batch reactions. The specific choice of ligands, such as triphenylphosphine or 2,2'-bipyridine derivatives, further stabilizes the low-valent nickel species, promoting the selective cross-coupling of the distinct chlorophthalimide substrates. This precise control over the reaction environment is what allows for the high total yields of 87 to 96 percent reported in the patent data. For R&D directors focused on purity and杂质谱 (impurity profiles), this mechanism offers a reproducible pathway to high-purity biphenyl dianhydride isomers that meets the stringent specifications required for electronic grade materials without the need for extensive downstream polishing.
How to Synthesize Biphenyl Dianhydride Isomers Efficiently
The practical implementation of this technology involves a streamlined sequence of coupling, separation, hydrolysis, and dehydration steps that are amenable to large-scale production. The process begins with the preparation of the reaction mixture under an inert nitrogen atmosphere, combining the specifically designed N-substituted chlorophthalimide monomers with the nickel catalyst system and zinc powder in anhydrous 2-THF. Following the ultrasonic-assisted coupling phase, the reaction mixture is subjected to hot filtration to isolate the soluble 3,3'-isomer fraction from the insoluble precipitate containing the 3,4'- and 4,4'-isomers. Each fraction is then independently hydrolyzed using aqueous sodium hydroxide, followed by acidification to precipitate the corresponding biphenyltetracarboxylic acids. The final dehydration step, typically conducted at 200 degrees Celsius or via azeotropic distillation with trimethylbenzene, converts these acids into the target dianhydrides. For a detailed breakdown of the specific molar ratios, temperature profiles, and workup procedures, please refer to the standardized synthesis guide below.
- Perform ultrasonic-assisted reductive coupling of N-substituted chlorophthalimides using Ni catalyst and Zinc in 2-THF at 60-100°C.
- Separate the soluble 3,3'-bis-imine intermediate from the insoluble 3,4'- and 4,4'-bis-imine mixture via hot filtration in 2-THF.
- Hydrolyze the separated intermediates and utilize differential solubility in hot water to isolate pure 3,3',4,4'- and 2,3',3,4'-biphenyltetracarboxylic acids before dehydration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ultrasonic-assisted nickel catalysis route offers compelling economic and operational benefits that extend far beyond simple yield improvements. The substitution of expensive palladium catalysts with abundant nickel salts immediately lowers the raw material cost base, while the elimination of complex chromatographic purification steps reduces the consumption of high-grade silica and organic eluents. The ability to separate isomers based on intrinsic solubility differences rather than energy-intensive distillation or recrystallization cycles translates into substantial cost savings in utility consumption and waste treatment. Furthermore, the high recovery rate of the 2-THF solvent, reported to exceed 90 percent in the patent examples, creates a closed-loop system that minimizes environmental impact and reduces the frequency of solvent replenishment orders. These factors collectively contribute to a more resilient and cost-efficient supply chain for high-purity biphenyl dianhydride isomers.
- Cost Reduction in Manufacturing: The transition from precious metal catalysis to a nickel-zinc system fundamentally alters the cost structure of BPDA production. By eliminating the need for costly palladium ligands and reducing the catalyst loading to mere mole percentages, the direct material costs are significantly optimized. Additionally, the simplified separation protocol removes the need for multiple recrystallization steps or preparative HPLC, which are traditionally the most expensive unit operations in fine chemical manufacturing. The overall process efficiency means that the same production capacity can yield a higher volume of saleable product, effectively lowering the cost per kilogram and making the previously premium-priced 3,4'-BPDA isomer more accessible for broader industrial applications.
- Enhanced Supply Chain Reliability: Dependence on single-source suppliers for specialized catalysts or difficult-to-separate intermediates poses a significant risk to production continuity. This new method utilizes commodity chemicals such as zinc powder, nickel chloride, and common phosphine ligands, which are readily available from multiple global vendors, thereby diversifying the supply base and mitigating shortage risks. The robustness of the ultrasonic coupling reaction also ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by off-spec material that requires reprocessing. For supply chain planners, this reliability translates into more predictable lead times for high-purity biphenyl dianhydride isomers and the ability to maintain lower safety stock levels without compromising production schedules.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges related to heat transfer and mixing efficiency, but the use of ultrasonic assistance addresses these issues at the molecular level. The technology facilitates rapid reaction kinetics even in larger vessels, ensuring that the benefits observed in the laboratory can be translated to commercial scale-up of complex electronic chemicals. Moreover, the process aligns with increasingly stringent environmental regulations by minimizing the generation of heavy metal waste associated with palladium residues and reducing the overall volume of organic solvent waste. The high atom economy and solvent recovery rates support sustainability goals, making this manufacturing route attractive for companies aiming to reduce their carbon footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this technology into existing production lines or for procurement specialists negotiating supply agreements for advanced polyimide precursors.
Q: How does this patent solve the separation issue of 3,4'-BPDA?
A: The patent utilizes a unique molecular design where different N-substituents (e.g., propyl vs. phenyl/methyl) create distinct solubility profiles for the bis-imine and tetra-acid intermediates, allowing physical separation that was previously impossible with mixed chlorides.
Q: What are the advantages of using Nickel over Palladium in this synthesis?
A: Nickel catalysts are significantly more cost-effective than Palladium systems. Furthermore, the ultrasonic assistance enhances the activity of the Zinc reducing agent, preventing surface passivation by ZnCl2 and ensuring high conversion rates without requiring excessive catalyst loading.
Q: Is this process scalable for industrial production?
A: Yes, the process operates at moderate temperatures (60-100°C) and uses recoverable solvents like 2-THF with recovery rates exceeding 90%. The simplified separation steps reduce waste generation, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biphenyl Dianhydride Isomers Supplier
As the demand for high-performance polyimides continues to surge in the aerospace, flexible electronics, and 5G communication sectors, securing a stable source of high-quality dianhydride monomers is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced catalytic technologies like the one described in CN113603663B to deliver superior chemical solutions. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet both pilot-scale research needs and full-scale manufacturing demands with equal proficiency. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify the isomeric composition and impurity profiles of every batch, guaranteeing that our clients receive materials that perform consistently in their final polymer applications.
We invite global partners to collaborate with us to optimize their material sourcing strategies and achieve significant competitive advantages. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team today to request specific COA data for our BPDA isomer portfolio and to discuss route feasibility assessments for your next-generation polyimide projects. Together, we can drive innovation and efficiency in the electronic materials supply chain.
