Breakthrough in Biphenyl Dianhydride Isomer Production: A Cost-Effective Nickel Catalytic Route
The global demand for high-performance polyimide materials, particularly for flexible printed circuits and advanced aerospace composites, has placed immense pressure on the supply chain of critical monomers like biphenyl tetracarboxylic dianhydride (BPDA). Among the various isomers, the asymmetric 3,4'-BPDA is highly valued for its ability to lower the viscosity of polyimide precursors, yet it remains notoriously difficult and expensive to procure in high purity. A recent technological breakthrough documented in patent CN113461646A introduces a novel preparation and separation method that fundamentally alters the economic landscape of producing these isomers. By shifting from traditional noble metal catalysis to a robust nickel-based system and employing a ingenious solubility-driven separation strategy, this innovation addresses the long-standing bottleneck of isomer purification. For industry stakeholders, this represents a pivotal shift towards more sustainable and cost-efficient manufacturing of electronic grade chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of biphenyl dianhydrides has relied heavily on palladium-catalyzed coupling reactions, which incur substantial raw material costs due to the volatility and scarcity of noble metals. Beyond the catalyst expense, the conventional synthetic routes typically generate a complex mixture of 3,3'-, 3,4'-, and 4,4'-isomers that possess nearly identical physical properties. The boiling points and solubility coefficients of these isomers are so similar that standard purification techniques like fractional distillation or simple recrystallization often fail to achieve the necessary separation efficiency. Consequently, manufacturers are forced to utilize labor-intensive chromatographic methods or accept lower purity grades, which severely limits the application of asymmetric BPDA in high-end electronics. This inability to efficiently isolate the desired 3,4'-isomer has kept market prices artificially high, often exceeding ten times the cost of the symmetric 4,4'-congener.
The Novel Approach
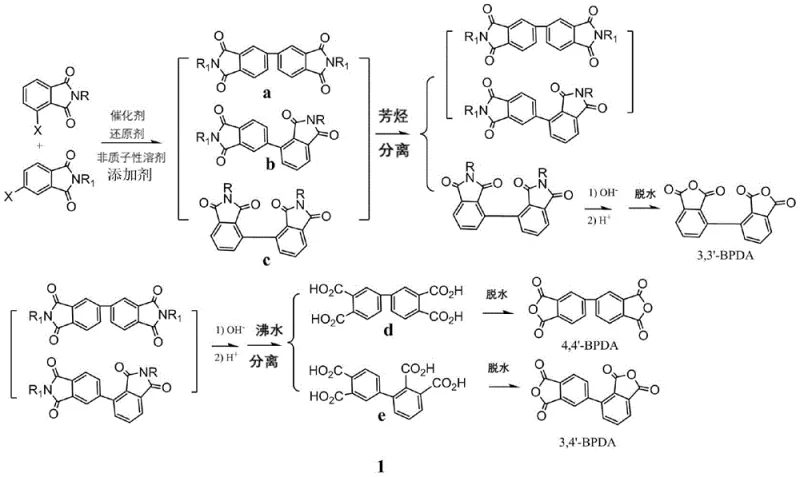
The methodology outlined in the patent data circumvents these thermodynamic limitations through a clever molecular design strategy that modifies the solubility of intermediates prior to the final dehydration step. Instead of attempting to separate the final dianhydrides directly, the process utilizes a mixture of aliphatic 3-halogenated phthalimide and aryl 4-halogenated phthalimide as starting materials. This specific combination ensures that the resulting coupling products have distinct solubility profiles in aromatic hydrocarbons. The bis-aliphatic amine-terminated intermediate (precursor to 3,3'-BPDA) becomes highly soluble, while the mixed and bis-aryl terminated intermediates remain insoluble, allowing for a clean physical separation via hot filtration. This strategic divergence in physical properties transforms an intractable chemical separation problem into a manageable unit operation.
Mechanistic Insights into Nickel-Catalyzed Reductive Coupling
At the heart of this synthesis lies a nickel-catalyzed reductive coupling mechanism that replaces the expensive palladium systems traditionally used for biaryl bond formation. The reaction employs a nickel salt, such as nickel chloride or nickel acetate, in conjunction with metallic zinc as a stoichiometric reductant within an anhydrous aprotic solvent environment like DMAc or NMP. The catalytic cycle likely involves the in situ generation of low-valent nickel species which facilitate the oxidative addition into the carbon-halogen bonds of the phthalimide substrates. Subsequent reductive elimination forms the critical carbon-carbon bond between the phthalimide rings. The presence of specific ligand additives, ranging from phosphines to nitrogen-containing heterocycles as depicted in the patent formulas, stabilizes the active nickel species and enhances the turnover frequency, ensuring high conversion rates even at moderate temperatures between 50°C and 100°C.
Crucially, the mechanism extends beyond bond formation to include a sophisticated impurity control strategy driven by the N-substituents on the phthalimide ring. By differentiating the nitrogen substituents—using an aliphatic group for the 3-position and an aryl group for the 4-position—the process engineers a disparity in intermolecular forces among the product isomers. The 3,3'-coupled product, bearing two aliphatic groups, exhibits significantly higher solubility in non-polar aromatic solvents compared to the rigid, planar 4,4'-isomer or the asymmetric 3,4'-isomer which retain aryl groups. This difference allows the 3,3'-intermediate to remain in the filtrate while the others precipitate, effectively removing one-third of the isomeric mixture before hydrolysis. The remaining mixture is then subjected to alkaline hydrolysis, where the resulting tetracarboxylic acids are separated based on their differential solubility in boiling water, ensuring that the final dehydration yields discrete, high-purity dianhydride isomers.
How to Synthesize Biphenyl Dianhydride Efficiently
The synthesis protocol described in the patent offers a streamlined pathway for producing high-purity BPDA isomers suitable for commercial polyimide manufacturing. The process begins with the precise mixing of nickel catalyst, ligand additives, and the specific halogenated phthalimide monomers under an inert atmosphere to prevent catalyst deactivation. Following the coupling reaction, the workup procedure leverages the engineered solubility differences to isolate the intermediates through sequential filtration and crystallization steps rather than complex chromatography. For a detailed breakdown of the specific molar ratios, temperature profiles, and solvent recovery steps required to replicate this high-efficiency route, please refer to the standardized synthesis guide below.
- Perform nickel-catalyzed coupling of mixed halogenated phthalimides using zinc as a reductant in an aprotic solvent.
- Separate the 3,3'-bis-aliphatic diimide intermediate from the mixture using aromatic hydrocarbon solubility differences.
- Hydrolyze the remaining intermediates and separate 4,4'- and 3,4'-tetracarboxylic acids based on hot water solubility before dehydration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nickel-catalyzed technology presents a compelling value proposition centered on cost stability and supply security. The transition from palladium to nickel catalysts immediately removes exposure to the volatile pricing of precious metals, which are subject to geopolitical supply shocks and mining constraints. Furthermore, the simplification of the purification train—from complex chromatography to straightforward filtration and crystallization—drastically reduces the processing time and energy consumption per kilogram of product. This operational efficiency translates directly into a more competitive cost structure, with the patent disclosure indicating that the preparation cost of asymmetric biphenyl dianhydride is reduced by more than 50% compared to previous methods. Such a reduction provides significant margin flexibility for downstream polymer manufacturers.
- Cost Reduction in Manufacturing: The elimination of noble metal catalysts and the replacement of energy-intensive separation techniques with simple solvent-based fractionation leads to substantial operational savings. By avoiding the need for expensive metal scavengers and complex purification columns, the overall cost of goods sold (COGS) is significantly lowered. This economic advantage allows suppliers to offer high-purity 3,4'-BPDA at price points that were previously unattainable, making high-performance polyimides more accessible for broader industrial applications.
- Enhanced Supply Chain Reliability: The robustness of the nickel-zinc catalytic system ensures consistent batch-to-batch reproducibility, which is critical for maintaining long-term supply contracts with electronics manufacturers. The reliance on commodity chemicals like zinc and nickel salts, rather than specialized organometallic complexes, mitigates the risk of raw material shortages. Additionally, the high selectivity of the separation process minimizes the generation of off-spec waste, ensuring that a larger proportion of the reactor output meets the stringent quality specifications required for electronic grade materials.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations such as reflux, filtration, and drying that are easily implemented in large-scale multipurpose reactors. The use of recoverable solvents like DMAc and xylene aligns with modern green chemistry principles by facilitating solvent recycling loops. Moreover, the avoidance of heavy metal contaminants in the final product simplifies wastewater treatment protocols and ensures compliance with increasingly strict environmental regulations regarding heavy metal discharge in chemical manufacturing zones.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel BPDA synthesis route. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the technology's viability. Understanding these nuances is essential for R&D teams considering the integration of this monomer into new polyimide formulations or for procurement specialists assessing supplier capabilities.
Q: Why is separating BPDA isomers traditionally difficult?
A: Traditional methods produce mixtures of 3,3'-, 3,4'-, and 4,4'-isomers which have extremely similar boiling points and solubility profiles, making purification via standard crystallization or distillation nearly impossible without significant yield loss.
Q: How does the nickel-catalyzed method reduce costs compared to palladium?
A: Nickel salts are significantly less expensive than noble metal palladium catalysts. Furthermore, the process eliminates the need for complex chromatographic separation steps, drastically lowering operational expenditures.
Q: What is the purity level achievable with this new separation technique?
A: The patent documentation indicates that this method allows for the isolation of biphenyl dianhydride isomers with a purity exceeding 98%, meeting the stringent requirements for high-performance polyimide applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biphenyl Dianhydride Supplier
As the demand for advanced electronic materials continues to surge, securing a stable source of high-purity monomers like 3,4'-BPDA is paramount for maintaining production continuity. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in catalytic coupling and crystallization technologies to deliver superior chemical intermediates. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet both pilot-scale development needs and full-volume manufacturing demands. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of biphenyl dianhydride meets the exacting standards required for next-generation polyimide films.
We invite potential partners to engage with our technical team to explore how this cost-effective synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to our nickel-catalyzed BPDA variants. We encourage you to contact our technical procurement team today to obtain specific COA data and route feasibility assessments tailored to your specific application requirements, ensuring a seamless transition to more efficient and sustainable material sourcing.
