Advanced Synthesis of Cyano-Modified Biphenyl Dianhydride Intermediates for Next-Gen Polyimide Manufacturing
Advanced Synthesis of Cyano-Modified Biphenyl Dianhydride Intermediates for Next-Gen Polyimide Manufacturing
The rapid evolution of the electronics and aerospace sectors demands polymer materials that can withstand extreme thermal environments while maintaining processability for complex fabrication methods. Patent CN102329290A introduces a groundbreaking class of modified biphenyl dianhydride intermediates containing cyano side chains, addressing the critical trade-off between thermal stability and solubility in aromatic polyimides. Traditional biphenyl-based polyimides often suffer from poor solubility due to rigid chain packing, necessitating high-temperature processing that limits their application scope. This proprietary technology leverages strategic functionalization to disrupt intermolecular forces, enabling the production of high-performance polymers that are easier to process without sacrificing mechanical integrity. As a leading entity in fine chemical synthesis, understanding the nuances of this patent provides a significant competitive edge in sourcing reliable polyimide intermediate suppliers capable of delivering next-generation materials.
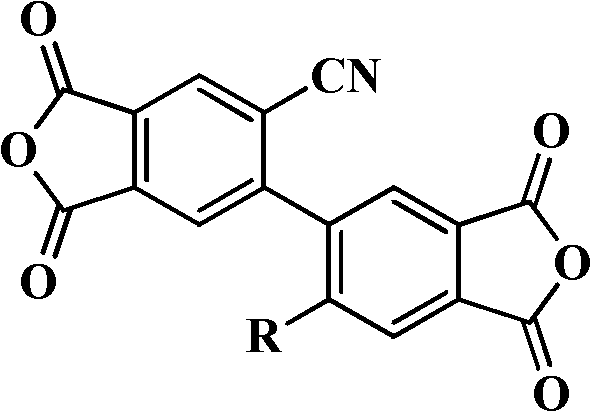
The core innovation lies in the versatile structural framework where the substituent R can be tailored as -OR1, -SR1, or -CN, allowing for precise tuning of dielectric and mechanical properties. This flexibility is paramount for manufacturers seeking cost reduction in electronic chemical manufacturing, as it permits the customization of material properties for specific end-use applications such as flexible printed circuits or high-temperature insulation layers. The synthesis pathway described in the patent is robust and scalable, utilizing standard industrial reagents and conditions that facilitate seamless technology transfer from laboratory to commercial production lines. By mastering this chemistry, supply chain leaders can secure a steady stream of high-purity biphenyl dianhydride derivatives that meet stringent quality specifications required by top-tier polymer producers globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aromatic polyimides relied heavily on unmodified monomers like 3,3',4,4'-biphenyltetracarboxylic dianhydride (BPDA), which impart exceptional thermal resistance but result in polymers with notoriously poor solubility. This insolubility creates significant bottlenecks in the commercial scale-up of complex polymer additives, as it often requires the use of aggressive, high-boiling solvents or processing in the precursor polyamic acid form followed by difficult thermal imidization. Furthermore, the high melting points associated with these rigid structures complicate melt-processing techniques, restricting the manufacturing of intricate components. Conventional modification strategies often involved the incorporation of bulky aliphatic groups which, while improving solubility, frequently degraded the thermal stability and mechanical strength of the final material, rendering them unsuitable for high-performance applications. These limitations have long plagued R&D teams striving to balance processability with performance in advanced material formulations.
The Novel Approach
The methodology outlined in CN102329290A offers a sophisticated solution by introducing polar cyano (-CN) or heteroatom-containing side chains directly onto the biphenyl backbone. This approach effectively disrupts the symmetry and packing efficiency of the polymer chains, thereby enhancing solubility in common organic solvents like NMP or DMAc without introducing thermally weak aliphatic linkages. The preservation of the aromatic core ensures that the intrinsic thermal stability and mechanical modulus remain intact, solving the classic dichotomy of polyimide design. Moreover, the electron-withdrawing nature of the cyano group can positively influence the dielectric properties of the resulting polymer, a critical factor for high-frequency electronic applications. This novel route not only expands the library of available monomers but also provides a streamlined pathway for creating specialized materials that meet the rigorous demands of modern optoelectronics and aerospace engineering.
Mechanistic Insights into CuCN-Mediated Cyanation and Cyclization
The synthetic route begins with the esterification of biphenyl tetraacid dianhydride in a methanol-sulfuric acid composite solvent at 80-95°C, forming the tetramethyl ester which serves as a soluble handle for subsequent functionalization. The critical step involves the electrophilic bromination of this ester using potassium bromate in concentrated sulfuric acid at controlled low temperatures (-10 to 10°C), ensuring selective substitution at the 2,2' positions to prevent over-bromination which could lead to impurities. Following isolation, the dibromo-intermediate undergoes a Rosenmund-von Braun reaction with cuprous cyanide in polar aprotic solvents such as DMF or DMAc at 150-160°C. This nucleophilic substitution is kinetically demanding and requires rigorous nitrogen protection to prevent oxidation of the copper catalyst, ultimately yielding the dicyano-tetraester with high fidelity. The precision of this step is vital for maintaining the purity profile required for electronic grade materials.
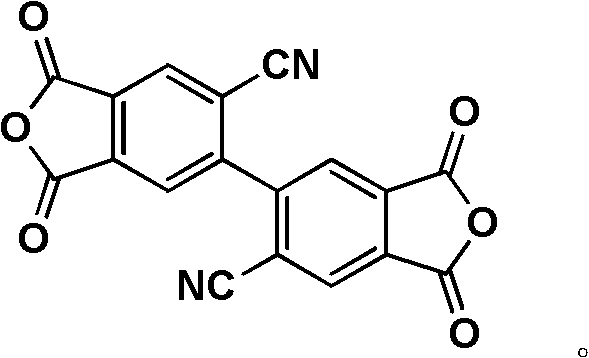
Subsequent hydrolysis of the nitrile-containing tetraester is achieved using alkali hydroxides in an aqueous acetonitrile system at mild temperatures (10-20°C), regenerating the tetracarboxylic acid functionality essential for polymerization. The final cyclodehydration to form the dianhydride ring can be accomplished via two distinct methods: thermal sublimation under reduced pressure at 200-280°C or chemical dehydration using acetic anhydride at 130-150°C. The choice between these methods depends on the thermal stability of the specific side chain; for instance, thermally sensitive ether linkages may prefer the chemical route to avoid degradation. This mechanistic versatility allows manufacturers to optimize the process for yield and purity, ensuring that the final high-purity biphenyl dianhydride intermediate meets the exacting standards necessary for downstream polymerization into high-performance polyimides.
How to Synthesize 2,2'-Dicyano-Biphenyl Dianhydride Efficiently
The synthesis of this specialized intermediate requires careful control of reaction parameters to maximize yield and minimize impurity formation, particularly during the bromination and cyanation stages. The patent details a five-step sequence that transforms readily available starting materials into a highly functionalized monomer suitable for advanced polymer synthesis. Operators must adhere strictly to temperature profiles and stoichiometric ratios, such as maintaining the methanol to sulfuric acid volume ratio between 20:1 and 20:3 during esterification, to ensure complete conversion. Detailed standardized synthesis steps see the guide below for specific operational parameters and safety protocols.
- Esterify biphenyl tetraacid dianhydride with methanol and sulfuric acid at 80-95°C to form the tetramethyl ester.
- Perform controlled bromination using potassium bromate in sulfuric acid at low temperatures (-10 to 10°C) to introduce bromine substituents.
- Execute Rosenmund-von Braun reaction with cuprous cyanide at 150-160°C to substitute bromine with cyano groups.
- Hydrolyze the ester groups using alkali hydroxide in aqueous acetonitrile to regenerate the tetracarboxylic acid.
- Cyclize the acid via dehydration with acetic anhydride or vacuum sublimation at 200-280°C to yield the final dianhydride.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this synthesis route offers substantial strategic benefits for procurement and supply chain management, primarily driven by the use of commodity chemicals and scalable unit operations. The reliance on standard reagents like methanol, sulfuric acid, and cuprous cyanide eliminates the need for exotic or supply-constrained catalysts, thereby enhancing supply chain reliability and reducing the risk of production stoppages due to raw material shortages. Furthermore, the ability to utilize either thermal sublimation or chemical dehydration for the final step provides operational flexibility, allowing manufacturers to select the most energy-efficient or equipment-compatible method for their specific facility. This adaptability translates directly into cost reduction in electronic chemical manufacturing by optimizing utility consumption and minimizing capital expenditure on specialized high-temperature sublimation apparatus if chemical dehydration is chosen.
- Cost Reduction in Manufacturing: The process avoids the use of expensive transition metal catalysts often required in cross-coupling reactions, relying instead on cost-effective cuprous cyanide which can be managed efficiently with proper waste treatment protocols. By eliminating the need for complex purification steps associated with metal removal, the overall production cost is significantly lowered while maintaining high product purity. Additionally, the high yields reported in the patent examples suggest a material-efficient process that minimizes waste generation, further contributing to economic viability and environmental compliance in large-scale operations.
- Enhanced Supply Chain Reliability: Since the starting material, biphenyl tetraacid dianhydride, is a well-established industrial chemical, the upstream supply chain is robust and less susceptible to volatility compared to routes requiring bespoke precursors. The modular nature of the synthesis, where different side chains (R groups) can be introduced using similar reaction conditions, allows for agile manufacturing responses to changing market demands for specific polyimide properties. This flexibility ensures that suppliers can maintain continuous production schedules even when switching between different variants of the intermediate, securing a stable supply for long-term contracts.
- Scalability and Environmental Compliance: The reaction conditions, predominantly involving reflux and standard filtration, are easily scalable from kilogram to multi-ton batches using conventional stainless steel reactors. The use of aqueous workups and standard organic solvents facilitates established waste treatment procedures, ensuring that the process aligns with increasingly stringent environmental regulations. The potential to recover and recycle solvents like acetonitrile and toluene further enhances the sustainability profile of the manufacturing process, making it an attractive option for companies committed to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the application and handling of these modified dianhydride intermediates, derived directly from the patent's experimental data and beneficial effects. Understanding these aspects is crucial for R&D teams evaluating the feasibility of incorporating these monomers into their existing polymer formulations. The answers provided reflect the specific advantages observed in the patent examples, such as improved solubility and thermal performance.
Q: How does the cyano side chain improve polyimide processability?
A: The introduction of polar cyano groups disrupts the regular packing of polymer chains, significantly improving solubility in common organic solvents without compromising the inherent thermal stability of the aromatic backbone.
Q: What are the key reaction conditions for the cyanation step?
A: The cyanation utilizes cuprous cyanide in polar aprotic solvents like DMF or DMAc at elevated temperatures (150-160°C) under nitrogen protection to ensure complete substitution of the bromo-intermediate.
Q: Can this intermediate be used for flexible display applications?
A: Yes, the resulting polyimides exhibit excellent thermal stability (high Tg) and improved solubility, making them ideal candidates for colorless PI films and flexible substrate materials in optoelectronics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2'-Dicyano-Biphenyl Dianhydride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance monomers play in the development of next-generation polyimides for electronics and aerospace applications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 2,2'-dicyano-biphenyl dianhydride meets the exacting standards required for electronic grade materials, minimizing defects in your final polymer products.
We invite you to collaborate with our technical procurement team to explore how this innovative intermediate can enhance your product portfolio. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are ready to provide specific COA data and route feasibility assessments to support your R&D initiatives and help you secure a competitive advantage in the global market for advanced polymer materials.
