Advanced Phase Transfer Catalysis for Efficient Sulfonylurea Herbicide Intermediate Manufacturing
The global demand for high-efficiency, low-toxicity herbicides has driven intense innovation in the synthesis of sulfonylurea compounds, a cornerstone class of modern agrochemicals. Patent CN101659642B introduces a transformative methodology that leverages phase-transfer catalysis (PTC) to synthesize these critical intermediates with superior efficiency and safety profiles. This technical breakthrough addresses long-standing challenges in the industry by replacing hazardous phosgene-based routes with a streamlined, one-pot reaction system that operates under mild conditions. For R&D directors and process chemists, this patent represents a pivotal shift towards greener chemistry, offering a robust pathway to generate complex heterocyclic structures with enhanced selectivity. The integration of solid-liquid phase transfer catalysts fundamentally alters the reaction kinetics, ensuring that insoluble cyanate species can effectively interact with organic sulfonyl chlorides. This report analyzes the technical depth of this innovation, providing actionable insights for procurement and supply chain leaders seeking reliable agrochemical intermediate suppliers who prioritize both quality and operational safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of sulfonylurea herbicides relied heavily on the condensation of isocyanates with aminoheterocycles, a process fraught with significant safety and economic drawbacks. Early synthetic routes frequently utilized phosgene gas to generate the necessary isocyanate intermediates from sulfonamides, a practice that imposes severe regulatory burdens and requires specialized, corrosion-resistant equipment to handle the extreme toxicity of the reagent. Even when alternatives like diphosgene or triphosgene were employed to mitigate gas handling risks, the processes still necessitated expensive basic catalysts such as DBU, which could not be recovered post-reaction, thereby inflating raw material costs. Furthermore, alternative non-phosgene methods involving the reaction of sulfonyl chlorides with sodium cyanate in the presence of nucleophilic catalysts often suffered from heterogeneous reaction conditions. In these traditional solid-liquid systems, the poor interfacial contact between the insoluble cyanate salt and the organic sulfonyl species frequently resulted in sluggish reaction rates and suboptimal yields, often hovering around merely 66 percent, which is economically unviable for large-scale manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a sophisticated phase-transfer catalytic system to overcome the kinetic barriers of heterogeneous reactions. By introducing specific quaternary ammonium or phosphonium salts, the process effectively solubilizes the cyanate anions within the organic phase, dramatically increasing the frequency of productive collisions with the sulfonyl chloride electrophiles. This method allows for a convenient one-pot synthesis where the sulfonyl chloride, cyanate, and aminoheterocycle react sequentially or concurrently in solvents like acetonitrile or toluene without the need for isolating unstable isocyanate intermediates. The operational simplicity is further enhanced by the ability to run the reaction at moderate temperatures ranging from 40°C to 90°C, reducing energy consumption compared to high-temperature reflux protocols. This strategic shift not only eliminates the need for toxic phosgene but also simplifies the downstream processing, as the reaction mixture can often be worked up directly by solvent evaporation and filtration.
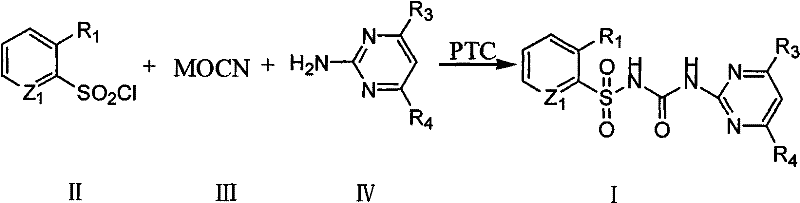
Mechanistic Insights into Phase-Transfer Catalyzed Sulfonylurea Formation
The core mechanistic advantage of this technology lies in the ability of the phase transfer catalyst (PTC) to act as a molecular shuttle across the phase boundary. In the absence of a PTC, the cyanate anion (OCN⁻) remains trapped in the solid lattice or aqueous interface, inaccessible to the lipophilic sulfonyl chloride dissolved in the organic solvent. The quaternary ammonium cation (Q⁺) of the catalyst exchanges with the metal cation of the cyanate salt to form a lipophilic ion pair [Q⁺][OCN⁻], which readily dissolves in the organic medium. Once in the organic phase, the naked cyanate anion exhibits heightened nucleophilicity, attacking the sulfur atom of the sulfonyl chloride to form a reactive sulfonyl isocyanate intermediate in situ. This intermediate then rapidly reacts with the added aminoheterocycle to form the final sulfonylurea linkage. This mechanism bypasses the diffusion limitations typical of solid-liquid reactions, ensuring that the concentration of the active nucleophile remains high throughout the reaction vessel, which is critical for driving the equilibrium towards product formation.
From an impurity control perspective, this mechanism offers distinct advantages over acid chloride or phosgene routes which are prone to generating chlorinated by-products and symmetric ureas. The mild conditions facilitated by the PTC minimize the thermal degradation of sensitive heterocyclic rings, such as the pyrimidine or pyridine moieties common in herbicide structures. Additionally, because the isocyanate is generated and consumed in situ without isolation, the risk of hydrolysis to the corresponding sulfonamide is significantly reduced, provided that moisture levels are controlled. The selectivity of the reaction is further tuned by the choice of solvent; polar aprotic solvents like acetonitrile stabilize the transition state without competing as nucleophiles, while the catalyst loading can be optimized between 1 to 5 mole percent to balance reaction rate against cost. This precise control over the reaction environment ensures a cleaner impurity profile, reducing the burden on purification units and increasing the overall mass balance of the process.
How to Synthesize Sulfonylurea Compounds Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing pilot and production facilities, requiring only standard glass-lined or stainless steel reactors. The process begins by charging the reactor with the sulfonyl chloride substrate and the chosen organic solvent, followed by the addition of the solid cyanate salt and the phase transfer catalyst. After an initial stirring period to allow for the formation of the reactive isocyanate species, the aminoheterocyclic coupling partner is introduced, and the mixture is heated to promote the condensation step. Detailed standardized operating procedures regarding specific stoichiometric ratios, agitation speeds, and crystallization parameters are essential for reproducibility.
- Dissolve pyridine or benzene sulfonyl chloride in a suitable organic solvent such as acetonitrile or toluene within a reactor.
- Add alkali metal cyanate (e.g., NaOCN) and a solid-liquid phase transfer catalyst (e.g., TBAB or TEBA) to the mixture and stir at ambient temperature.
- Introduce the aminoheterocyclic compound to the reaction system, heat to reflux (40-90°C), and maintain until completion before standard workup.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this phase-transfer catalytic route translates into tangible strategic benefits that extend beyond simple yield improvements. The elimination of phosgene and its solid surrogates removes a major bottleneck in raw material sourcing and storage, as these highly regulated toxins require specialized logistics and containment infrastructure that drive up overhead costs. By shifting to stable, commodity-grade reagents like sodium cyanate and common quaternary ammonium salts, manufacturers can secure a more resilient supply chain that is less susceptible to regulatory shutdowns or transportation restrictions. Furthermore, the one-pot nature of the reaction drastically reduces the number of unit operations required, minimizing solvent usage and waste generation, which aligns with increasingly stringent environmental compliance standards globally. This streamlined workflow allows for faster batch turnover times, enabling suppliers to respond more agilely to market fluctuations in herbicide demand.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the removal of expensive catalysts like DBU and the avoidance of complex isocyanate isolation steps. Traditional methods often incur high costs due to the need for stoichiometric amounts of activating agents and the subsequent disposal of hazardous waste streams associated with phosgene chemistry. In this novel process, the catalytic amount of PTC required is minimal, and the catalyst can potentially be recovered or recycled depending on the specific workup protocol, leading to substantial savings in auxiliary material costs. Additionally, the higher reaction yields mean that less raw material is wasted per kilogram of finished product, directly improving the cost of goods sold (COGS) and enhancing margin potential for high-volume agrochemical intermediates.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on hazardous reagents that face strict transportation quotas and seasonal production bans. By utilizing benign reagents such as alkali metal cyanates and widely available sulfonyl chlorides, this method diversifies the supplier base and reduces the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a broader range of temperatures and mixing efficiencies compared to sensitive phosgenation reactions, ensures consistent output even during periods of high production volume. This reliability is crucial for maintaining just-in-time inventory levels for downstream formulators who depend on steady flows of active ingredients for seasonal agricultural applications.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden inefficiencies, but the homogeneous nature of the PTC-mediated reaction facilitates smooth scale-up from laboratory to multi-ton production. The absence of gas evolution hazards simplifies reactor design and safety interlocks, allowing for larger batch sizes without proportional increases in capital expenditure. From an environmental standpoint, the reduction in hazardous waste and the use of recoverable solvents like toluene or acetonitrile support sustainability goals, making the manufacturing process more attractive to eco-conscious partners and regulators. This alignment with green chemistry principles future-proofs the supply chain against tightening environmental legislation, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is vital for stakeholders evaluating its potential for commercial adoption. The following questions address common concerns regarding safety, efficiency, and product quality, drawing directly from the experimental data and claims within the patent literature. These insights are intended to clarify how this technology differentiates itself from incumbent manufacturing practices and what it means for the quality of the final agrochemical product.
Q: How does phase transfer catalysis improve the yield of sulfonylurea synthesis compared to traditional methods?
A: Traditional methods often suffer from poor contact between solid cyanate salts and organic sulfonyl chlorides. Phase transfer catalysts facilitate the transport of cyanate anions into the organic phase, significantly increasing collision frequency and reaction yield without requiring toxic phosgene reagents.
Q: Is this synthesis method scalable for industrial production of agrochemical intermediates?
A: Yes, the process utilizes a one-pot protocol with common solvents like acetonitrile or toluene and avoids hazardous phosgene gas, making it highly suitable for safe commercial scale-up and continuous manufacturing environments.
Q: What are the primary purity advantages of this non-phosgene route?
A: By eliminating the use of phosgene and complex isocyanate preparation steps, the process reduces the formation of chlorinated by-products and urea impurities, resulting in cleaner crude products that require less intensive purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonylurea Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic methodologies requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of phase-transfer catalysis are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of sulfonylurea intermediate meets the exacting standards required for modern herbicide formulations, minimizing the risk of crop damage due to impurities. We are committed to leveraging innovations like CN101659642B to deliver superior value to our global clientele.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing footprint. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced PTC capabilities can enhance your product portfolio and drive sustainable growth in the agrochemical sector.
