Advanced Continuous Flow Synthesis of Nirmatrelvir Pyrrolidone Intermediates for Commercial Scale-up
Advanced Continuous Flow Synthesis of Nirmatrelvir Pyrrolidone Intermediates for Commercial Scale-up
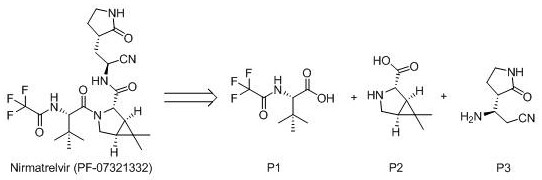
The global pharmaceutical landscape has been profoundly reshaped by the urgent demand for effective antiviral therapies, particularly following the emergence of SARS-CoV-2. At the forefront of this medical response is Nirmatrelvir (PF-07321332), the active protease inhibitor component of Paxlovid, which has demonstrated exceptional efficacy in reducing hospitalization risks. The synthesis of this complex molecule relies heavily on the availability of high-quality chiral intermediates, specifically the pyrrolidone fragment known as (2S,3S)-2-amino-3-[(2-pyrrolidinonyl)]-propionitrile. A groundbreaking patent, CN114230504B, filed in June 2022, introduces a transformative manufacturing methodology that shifts the production paradigm from traditional batch processing to a sophisticated continuous flow regime. This technological leap addresses critical bottlenecks in safety, efficiency, and scalability that have long plagued the synthesis of such sensitive pharmaceutical building blocks.
This patent details a robust four-step continuous sequence that integrates lithium-hydrogen exchange, heterogeneous hydrogenation, high-pressure amination, and dehydration into a unified production line. For R&D directors and process chemists, the significance lies in the meticulous optimization of reaction parameters that allow for the stable handling of hazardous reagents like organolithiums and high-pressure hydrogen gas. By transitioning these unit operations into a continuous environment, the process not only mitigates the inherent risks associated with exothermic reactions and pressurized gases but also establishes a foundation for reproducible, high-purity manufacturing. The strategic implementation of fixed-bed reactors and micro-reactor technology ensures that the thermal and mass transfer limitations of conventional kettle reactors are effectively overcome, paving the way for a more resilient supply chain.
The transition from legacy batch methodologies to this novel continuous flow approach represents a fundamental upgrade in process chemistry engineering. Conventional batch synthesis of pyrrolidone intermediates typically involves discrete, isolated steps that require significant manual intervention, extensive solvent handling, and large equipment footprints. In a traditional setting, the initial lithiation step often necessitates massive ultra-low temperature reactors to manage the exotherm and prevent decomposition, creating substantial energy burdens and safety hazards. Furthermore, the subsequent hydrogenation step usually requires high-pressure autoclaves, which pose significant operational risks and are difficult to scale linearly due to heat transfer limitations. These batch processes are also prone to the "amplification effect," where reaction outcomes change unpredictably as the vessel size increases, leading to inconsistent yields and impurity profiles that complicate regulatory approval.
In stark contrast, the novel approach outlined in patent CN114230504B leverages the intrinsic advantages of continuous flow chemistry to dismantle these barriers. By utilizing a series of connected reactor modules, the process achieves precise control over residence time and temperature, allowing the lithium-hydrogen exchange to proceed safely at relatively higher temperatures than would be feasible in a batch tank. The integration of a fixed-bed reactor for the hydrogenation step eliminates the need for filtering pyrophoric catalysts and enables continuous operation under pressure with enhanced safety margins. Moreover, the continuous amination step utilizes the system's ability to maintain high pressure to increase ammonia solubility, drastically accelerating reaction rates that are typically sluggish in batch systems. This holistic integration results in a streamlined workflow that minimizes intermediate isolation, reduces solvent consumption, and delivers a product with superior consistency.
Mechanistic Insights into Continuous Flow Cascade Synthesis
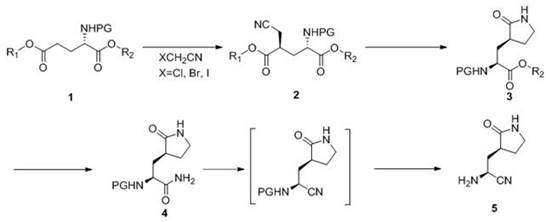
The core of this innovative synthesis lies in the precise orchestration of four distinct chemical transformations within a continuous flow environment. The first stage involves the generation of a lithium enolate from a protected amino ester precursor using a strong base such as LiHMDS, followed immediately by nucleophilic substitution with a halogenated acetonitrile. In a flow reactor, the rapid mixing capabilities ensure that the highly reactive lithiated intermediate encounters the electrophile almost instantaneously, minimizing side reactions like self-condensation or elimination. The system is designed to operate at cryogenic temperatures ranging from -20°C to -78°C, but the efficient heat exchange surface area of the flow reactor allows for much tighter temperature control than a jacketed batch vessel, ensuring the stereochemical integrity of the chiral center is preserved throughout the cyanomethylation process.
Following the formation of the nitrile intermediate, the process moves to a heterogeneous catalytic hydrogenation step performed in a fixed-bed reactor packed with catalysts such as Raney Cobalt or Ruthenium on Carbon. This step is critical as it simultaneously reduces the nitrile group and facilitates an intramolecular cyclization to form the pyrrolidone ring. The continuous flow setup allows for the precise regulation of hydrogen pressure and liquid flow rates, ensuring complete conversion while preventing over-reduction or catalyst deactivation. Subsequent steps involve a high-temperature, high-pressure amination reaction where the ester moiety is converted to a primary amide using aqueous ammonia. The ability of the flow system to withstand pressures up to 30 bar allows for the use of ammonia concentrations that would be gaseous and unmanageable in open batch systems, driving the equilibrium towards the desired amide product. Finally, a dehydration step using agents like cyanuric chloride converts the amide to the final nitrile functionality, completing the synthesis of the key pharmacophore with high efficiency.
How to Synthesize (2S,3S)-2-amino-3-[(2-pyrrolidinonyl)]-propionitrile Efficiently
The synthesis of this critical Nirmatrelvir intermediate requires a sophisticated understanding of flow chemistry parameters to maximize yield and safety. The patented process outlines a clear pathway starting from readily available protected amino esters, transforming them through a sequence of lithiation, cyclization, amination, and dehydration. Each step is optimized for continuous operation, utilizing specific molar ratios, temperature zones, and residence times to ensure robust performance. For process engineers looking to implement this technology, the key lies in the modular design of the reactor system, which allows for independent control of each reaction stage. The detailed standardized synthesis steps for implementing this continuous flow protocol are provided in the guide below.
- Perform continuous lithium-hydrogen exchange and nucleophilic cyanomethylation using LiHMDS and halogenated acetonitrile in a flow reactor at -20 to -78°C.
- Execute hydrogenation and cyclization of the intermediate in a fixed-bed reactor using catalysts like Raney Cobalt or Ru/C at 50-150°C.
- Conduct continuous amination by reacting the cyclic intermediate with aqueous ammonia solution at elevated temperatures (80-180°C) and pressures (5-30 bar).
- Complete the synthesis via continuous dehydration using agents like cyanuric chloride to generate the final nitrile product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this continuous flow technology offers profound strategic benefits that extend far beyond simple chemical yield improvements. The shift away from batch processing fundamentally alters the cost structure and risk profile of manufacturing high-value pharmaceutical intermediates. By eliminating the need for specialized, capital-intensive equipment such as large-scale ultra-low temperature freezers and high-pressure batch hydrogenation autoclaves, the process significantly reduces capital expenditure requirements. Furthermore, the continuous nature of the reaction minimizes the inventory of hazardous intermediates held on-site at any given time, lowering insurance costs and regulatory compliance burdens associated with storing dangerous chemicals. This lean manufacturing approach translates directly into a more agile and cost-effective supply chain capable of responding rapidly to market fluctuations.
- Cost Reduction in Manufacturing: The continuous flow process drives down manufacturing costs through multiple mechanisms, primarily by enhancing resource efficiency and reducing waste generation. The precise control over reaction stoichiometry and residence time minimizes the formation of by-products, which in turn reduces the load on downstream purification units and lowers solvent consumption. Additionally, the ability to run the hydrogenation step in a fixed-bed mode eliminates the costly and time-consuming filtration and disposal of spent catalyst slurries typical of batch processes. The energy efficiency is also markedly improved, as the high surface-to-volume ratio of flow reactors allows for rapid heating and cooling, avoiding the prolonged energy draw of maintaining large reaction masses at extreme temperatures for extended periods.
- Enhanced Supply Chain Reliability: Supply continuity is a critical concern for global pharmaceutical companies, and this technology offers a robust solution to production bottlenecks. Unlike batch processes that are susceptible to variability between batches and scale-up failures, continuous flow systems provide a steady state of production with consistent quality output. The modular nature of the equipment means that capacity can be increased simply by running the system for longer durations or by numbering up reactor modules, without the need for constructing new large-scale facilities. This flexibility ensures that suppliers can meet surging demand for antiviral intermediates without compromising on delivery timelines or product specifications, thereby securing the supply chain against disruptions.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, the continuous process aligns perfectly with modern green chemistry principles. The reduction in solvent usage and the minimization of waste streams significantly lower the environmental footprint of the manufacturing process. The closed nature of the flow system prevents the release of volatile organic compounds and toxic gases, such as ammonia, into the workplace environment, ensuring higher safety standards for operators. Furthermore, the absence of scale-up effects means that the process developed at the laboratory scale can be transferred to commercial production with minimal re-validation, accelerating the time-to-market for new drug formulations and ensuring compliance with stringent regulatory purity standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous flow synthesis of pyrrolidone intermediates. These insights are derived directly from the technical specifications and beneficial effects described in the patent literature, providing clarity on how this advanced manufacturing technique resolves traditional production challenges. Understanding these nuances is essential for stakeholders evaluating the feasibility and advantages of adopting this technology for their own supply chains.
Q: How does continuous flow technology improve safety in pyrrolidone intermediate synthesis?
A: The continuous flow process eliminates the need for large-scale ultra-low temperature reactors and high-pressure batch hydrogenation vessels. By managing dangerous reagents like organolithiums and high-pressure hydrogen in small, controlled volumes within tubular reactors, the thermal mass is minimized, significantly reducing the risk of runaway reactions and improving overall operational safety.
Q: What are the purity advantages of this continuous manufacturing route?
A: The plug flow characteristics of the continuous reactor system greatly reduce back-mixing, which is a common source of side reactions in batch processes. This precise control over residence time and mixing ensures a narrower residence time distribution, leading to fewer impurities, simplified downstream purification, and consistently higher product purity suitable for pharmaceutical applications.
Q: Is this process scalable for industrial production of Nirmatrelvir intermediates?
A: Yes, the process is designed for seamless scale-up. Unlike batch reactions which suffer from amplification effects (changes in heat and mass transfer as volume increases), continuous flow reactors maintain consistent reaction parameters regardless of scale. Numbering up reactor modules allows for linear capacity expansion without re-optimizing reaction conditions, ensuring reliable supply continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nirmatrelvir Intermediate Supplier
As the pharmaceutical industry continues to evolve towards more efficient and sustainable manufacturing practices, the expertise to translate complex patent technologies into commercial reality becomes invaluable. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, possessing the technical acumen and infrastructure to master the continuous flow synthesis of critical antiviral intermediates. Our team of expert process chemists and engineers has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial application is seamless. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite global partners to collaborate with us to leverage these advanced manufacturing capabilities for your supply chain needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our continuous flow expertise can drive value and reliability in your Nirmatrelvir intermediate supply chain.
