Advanced Febuxostat Manufacturing: Safer Routes and Commercial Scalability
Advanced Febuxostat Manufacturing: Safer Routes and Commercial Scalability
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical active pharmaceutical ingredients (APIs) that balance efficiency with stringent safety standards. Patent CN102002016A introduces a significant improvement in the synthesis of Febuxostat, a potent xanthine oxidase inhibitor used for treating chronic hyperuricemia and gout. This technical disclosure outlines a novel seven-step synthetic route that strategically bypasses the use of highly toxic cyanating reagents, such as potassium cyanide, which are prevalent in conventional methodologies. By shifting the synthetic strategy to utilize 4-hydroxybenzonitrile as a starting material and employing a controlled HCl/DMF system for thioamide formation, the process achieves a remarkable enhancement in operational safety and environmental compliance. For R&D directors and procurement specialists, this represents a pivotal shift towards greener chemistry without compromising the structural integrity or yield of the final API intermediate.
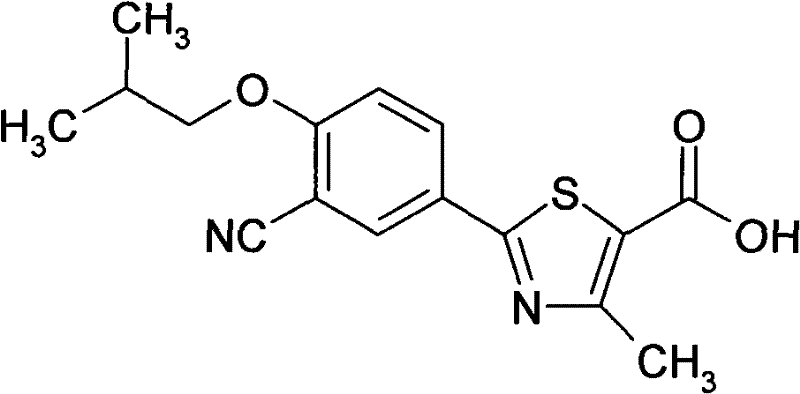
Furthermore, the elimination of high-pressure catalytic hydrogenation steps, often required in alternative routes, simplifies the equipment requirements and reduces the capital expenditure associated with specialized high-pressure reactors. The patent emphasizes that every step in this improved sequence yields a solid product, a critical feature for supply chain stability as solids are inherently easier to transport, store, and handle compared to unstable oils or liquids. This characteristic not only streamlines logistics but also ensures consistent quality control across different production batches. As a reliable pharmaceutical intermediates supplier, understanding these nuanced process improvements is essential for evaluating the long-term viability and cost-effectiveness of sourcing strategies for gout therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Febuxostat and its key intermediates has relied heavily on hazardous chemical transformations that pose significant risks to both personnel and the environment. Traditional routes, as referenced in prior art such as JP6345724 and EP0513379, frequently employ potassium cyanide for the introduction of the cyano group, a reagent classified under state key control due to its extreme toxicity. The handling of such substances necessitates rigorous operator protection protocols and complex three-waste treatment systems, driving up operational costs and regulatory burdens. Additionally, certain legacy pathways involve high-pressure catalytic hydrogenation, which imposes strict requirements on reactor design and safety monitoring, thereby limiting the flexibility of manufacturing facilities. These conventional methods often result in lower overall yields and generate substantial hazardous waste, creating a bottleneck for sustainable commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the improved method detailed in CN102002016A offers a streamlined and safer alternative that fundamentally redesigns the synthetic logic. By initiating the synthesis with 4-hydroxybenzonitrile, the process ingeniously incorporates the nitrile functionality from the outset, completely obviating the need for external cyanating agents in later stages. The core innovation lies in the formation of the thiazole ring through a condensation reaction that avoids severe toxicity, followed by a Duff reaction to introduce the necessary aldehyde group ortho to the phenolic hydroxyl. This approach not only mitigates the risks associated with toxic reagents but also utilizes cheap, common reagents that are easily sourced from global markets. The resulting pathway is characterized by mild reaction conditions, avoiding high temperatures and pressures, which translates directly into reduced energy consumption and enhanced process safety for industrial applications.
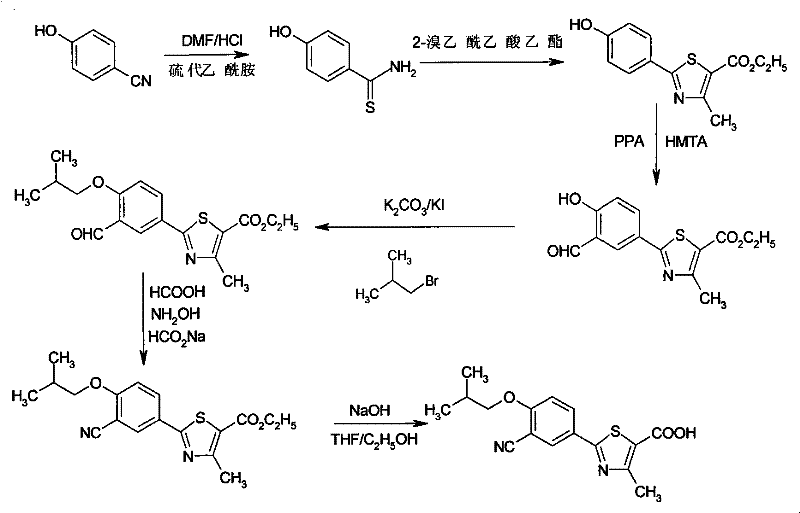
Mechanistic Insights into Thiazole Ring Closure and Duff Formylation
The success of this synthetic route hinges on the precise control of reaction conditions during the thiazole ring closure and subsequent formylation steps. The formation of p-hydroxythiobenzamide from 4-hydroxybenzonitrile and thioacetamide is catalyzed by hydrogen chloride in DMF, where the concentration of HCl and reaction temperature are critical parameters. Experimental data indicates that maintaining the HCl mass fraction between 15-20% and the temperature around 80-85°C optimizes the yield to over 70%, preventing the formation of dark oily by-products that occur at higher concentrations or temperatures. This specific solvent system facilitates the nucleophilic attack and cyclization required to build the heterocyclic core, ensuring high purity of the intermediate before it proceeds to the next stage. Such mechanistic control is vital for R&D teams aiming to replicate the process with minimal impurity profiles.
Following the ring closure, the introduction of the aldehyde group via the Duff reaction using polyphosphoric acid (PPA) and hexamethylenetetramine is another key mechanistic feature. This electrophilic aromatic substitution occurs selectively at the ortho position relative to the hydroxyl group, driven by the activating nature of the phenol and the specific conditions provided by the PPA medium. The subsequent alkylation of the phenolic hydroxyl with isobutyl bromide and the conversion of the aldehyde to a nitrile group using hydroxylamine and formic acid complete the construction of the carbon skeleton. Each transformation is designed to proceed with high selectivity, minimizing the generation of regioisomers or side products that would complicate downstream purification. This level of chemical precision ensures that the final hydrolysis step yields Febuxostat with a chromatographic purity exceeding 99.5%, meeting the stringent specifications required for pharmaceutical-grade materials.
How to Synthesize Febuxostat Efficiently
The synthesis of Febuxostat via this improved route involves a logical sequence of seven distinct chemical transformations, each optimized for yield and ease of isolation. The process begins with the formation of the thioamide precursor, followed by cyclization to the thiazole ester, formylation, alkylation, nitrile formation, and finally ester hydrolysis. Detailed operational parameters, such as the specific ratio of solvents and the precise temperature ramps for each step, are critical for achieving the reported high yields. For technical teams looking to implement this methodology, adherence to the specified workup procedures, such as pouring reaction mixtures into ice water for crystallization, is essential for maximizing recovery. The comprehensive guide below outlines the standardized steps derived from the patent embodiments to ensure reproducible results.
- React 4-hydroxybenzonitrile with thioacetamide in HCl/DMF solution to form p-hydroxythiobenzamide.
- Cyclize with ethyl 2-chloroacetoacetate in ethanol to form the thiazole ring intermediate.
- Perform Duff reaction using polyphosphoric acid and hexamethylenetetramine to introduce the aldehyde group.
- Alkylate the phenolic hydroxyl with isobutyl bromide, then convert the aldehyde to a nitrile group.
- Hydrolyze the ethyl ester under alkaline conditions to yield the final Febuxostat acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this improved synthesis route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain risk profile by removing controlled, high-toxicity raw materials from the bill of materials. Sourcing potassium cyanide often involves complex permitting, specialized logistics, and heightened security measures, all of which add friction and cost to the procurement process. By eliminating this dependency, manufacturers can secure a more resilient supply of raw materials, reducing the likelihood of production stoppages due to regulatory delays or supplier shortages. Furthermore, the use of common, low-cost reagents like thioacetamide and isobutyl bromide ensures stable pricing and broad availability, shielding the project from volatile market fluctuations associated with specialty chemicals.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of expensive safety infrastructure and waste treatment protocols. Without the need for high-pressure hydrogenation equipment or extensive cyanide detoxification systems, the capital expenditure for setting up production lines is significantly lowered. Additionally, the ability to isolate solid intermediates at every step reduces solvent consumption and energy costs associated with distilling large volumes of liquid products. The overall yield improvement, culminating in a total recovery rate that is competitive with industry standards, further drives down the cost per kilogram of the final API. These factors combine to create a highly cost-efficient manufacturing model that enhances profit margins for downstream drug producers.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route directly translates to improved supply continuity. Since the process avoids extreme operating conditions like high temperature and high pressure, the risk of equipment failure or unplanned maintenance downtime is minimized. The stability of the solid intermediates allows for flexible production scheduling, where batches can be paused or stored without degradation, providing greater agility in responding to market demand. This reliability is crucial for maintaining consistent inventory levels and meeting the just-in-time delivery expectations of major pharmaceutical clients. Consequently, partners adopting this technology can offer more dependable lead times and stronger service level agreements.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with modern green chemistry principles. The absence of severe toxicity and the generation of manageable waste streams simplify the regulatory approval process for new manufacturing sites. Scaling up from pilot plant to commercial production is facilitated by the straightforward nature of the unit operations, such as filtration and crystallization, which are easily adaptable to larger reactor volumes. This scalability ensures that the supply can grow in tandem with the commercial success of the final drug product, supporting long-term business growth without encountering technical bottlenecks related to process complexity or environmental restrictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Febuxostat synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on purity, safety, and operational feasibility. Understanding these details is essential for stakeholders evaluating the potential integration of this route into their existing manufacturing portfolios.
Q: How does this synthesis route improve safety compared to conventional methods?
A: This method eliminates the use of highly toxic potassium cyanide (KCN) and avoids high-pressure catalytic hydrogenation, significantly reducing operator risk and waste treatment complexity.
Q: What is the expected purity of the final Febuxostat product?
A: The patent specifies that through recrystallization steps involving ethanol and acetone, the chromatographic purity can reach higher than 99.5% with single impurities below 0.1%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process avoids extreme conditions like high temperature and pressure. All intermediates are solid products, which facilitates easy transportation, storage, and scalable manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Febuxostat Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like CN102002016A to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this safer, greener chemistry are fully realized in practice. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of Febuxostat intermediate meets the highest quality standards, with impurity profiles tightly controlled to support successful regulatory filings. Our commitment to technical excellence allows us to navigate the complexities of thiazole chemistry and formylation reactions with precision, delivering products that consistently exceed client expectations.
We invite you to collaborate with us to leverage these technological advantages for your supply chain. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your transition to this improved manufacturing process is seamless and economically sound. Let us be your trusted partner in delivering high-quality, cost-effective solutions for the next generation of gout therapeutics.
