Advanced Cyclization Technology for Citalopram: Enhancing Purity and Commercial Scalability
Advanced Cyclization Technology for Citalopram: Enhancing Purity and Commercial Scalability
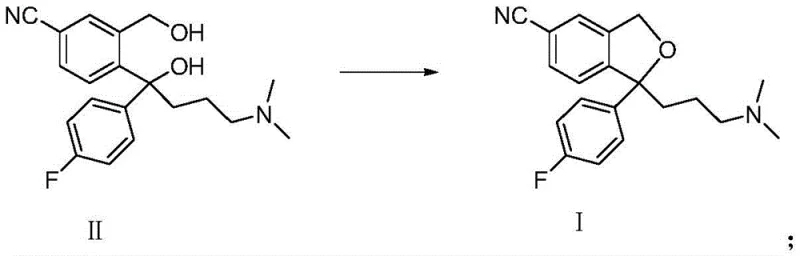
The pharmaceutical industry constantly seeks robust manufacturing routes for critical antidepressants like Citalopram and its active enantiomer, S-Citalopram. A pivotal advancement in this domain is detailed in patent CN104262306B, which discloses a highly efficient industrial process for the cyclization of diol precursors. This technology addresses long-standing challenges in solvent selection and workup efficiency, offering a pathway to significantly higher yields and purity profiles. By shifting away from traditional solvents like toluene or tetrahydrofuran, this method utilizes specific C4-C7 ketone solvents to create a homogeneous reaction environment that facilitates superior mass transfer. For R&D directors and procurement specialists, understanding this shift is crucial, as it represents a tangible opportunity to optimize the supply chain for this high-volume pharmaceutical intermediate. The patent outlines a method that not only improves reaction kinetics but also drastically simplifies the downstream processing, thereby reducing the overall environmental footprint and operational costs associated with large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Citalopram via cyclization of diol compounds has been plagued by significant inefficiencies related to solvent choice. Traditional methods often rely on solvents such as toluene, which exhibit poor solubility for the polar diol starting materials and the target product. This low solubility necessitates the use of excessive solvent volumes to maintain a viable reaction mixture, leading to bloated reactor occupancy times and increased waste generation. Furthermore, the poor solubility complicates the extraction process, often requiring repeated acid and alkali washing steps that degrade overall yield. Alternatively, some processes utilize highly water-miscible solvents like tetrahydrofuran (THF) or acetonitrile. While these solvents dissolve the reactants well, they create a nightmare for post-reaction workup; because they mix freely with water, the reaction mixture must be concentrated to dryness before a new extraction solvent can be introduced. This additional concentration step is energy-intensive, time-consuming, and introduces opportunities for product degradation, ultimately capping the achievable purity and yield at suboptimal levels.
The Novel Approach
The innovative process described in the patent overcomes these hurdles by employing a mixed solvent system comprising water and a C4-C7 ketone, such as methyl isobutyl ketone (MIBK) or methyl ethyl ketone. This specific class of solvents strikes a perfect balance: they possess high solubility for the organic diol reactants and the resulting Citalopram product, yet they remain immiscible enough with water to allow for direct phase separation. This unique property eliminates the need for energy-intensive concentration steps prior to extraction. As demonstrated in the patent examples, switching to this ketone-based system elevates the yield from roughly 70-75% in conventional methods to an impressive 83-85%. The reaction proceeds smoothly under alkaline conditions with aryl or alkyl sulfonyl chlorides, and the product can be isolated simply by separating the organic layer, washing it with water, and concentrating. This streamlined workflow not only boosts productivity but also ensures a cleaner impurity profile, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Ketone-Mediated Cyclization
The core of this technological breakthrough lies in the physicochemical properties of the C4-C7 ketone solvent system during the nucleophilic substitution and subsequent cyclization. In this mechanism, the diol precursor reacts with a sulfonyl chloride (such as p-toluenesulfonyl chloride) in the presence of a base like sodium hydroxide. The base deprotonates one of the hydroxyl groups, generating an alkoxide intermediate that attacks the sulfur atom of the sulfonyl chloride, forming a sulfonate ester in situ. This sulfonate group serves as an excellent leaving group, facilitating an intramolecular nucleophilic attack by the second hydroxyl group (or its alkoxide form) onto the benzylic carbon. This intramolecular displacement closes the isobenzofuran ring, releasing the sulfonate anion and yielding the final Citalopram structure. The choice of ketone solvent is critical here; it stabilizes the transition states through dipole interactions while maintaining the reactants in a single organic phase, thereby maximizing the collision frequency between the reactive species and driving the equilibrium towards the cyclic product.
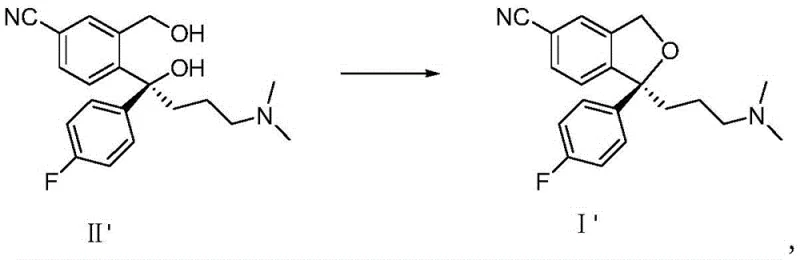
Furthermore, the solvent system plays a pivotal role in impurity control and stereochemical integrity, particularly for the synthesis of S-Citalopram. The high solubility of the product in the ketone phase prevents premature precipitation, which can often trap impurities or lead to occlusion of unreacted starting materials. By maintaining a homogeneous solution throughout the reaction temperature range of -5°C to 40°C, the process ensures that side reactions, such as elimination or over-sulfonylation, are minimized. The ease of phase separation post-reaction allows for the efficient removal of inorganic salts and water-soluble byproducts simply by washing with water. This "clean" workup is essential for achieving the high purity levels (>99.5%) reported in the patent data. For the chiral variant, the mild conditions preserve the stereocenter, ensuring that the S-enantiomer is produced with high optical purity, which is a non-negotiable requirement for the efficacy of the final antidepressant medication.
How to Synthesize Citalopram Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize the benefits of the ketone solvent system. The process begins by dissolving the diol compound in the selected ketone solvent, followed by the addition of an aqueous base. Temperature control is vital; the reaction is typically initiated at low temperatures (around 3°C) to manage the exotherm upon addition of the sulfonyl chloride, then allowed to warm to room temperature to ensure completion. The beauty of this method lies in its operational simplicity: once the reaction is complete, the mixture naturally separates into two distinct layers. The organic layer, containing the dissolved product, is easily isolated, washed to remove residual base and salts, and concentrated. This straightforward protocol minimizes operator error and equipment complexity, making it highly suitable for GMP manufacturing environments where reproducibility is paramount.
- Dissolve the diol compound (II) or S-diol compound (II') in a C4-C7 ketone solvent such as methyl isobutyl ketone at room temperature.
- Add an aqueous alkaline solution (e.g., NaOH) and cool the mixture to between -5°C and 40°C before adding the sulfonyl chloride reagent.
- Upon reaction completion, separate the layers directly, wash the organic phase with water, and concentrate to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ketone-based cyclization process offers compelling economic and logistical advantages. The primary driver for cost reduction is the drastic simplification of the downstream processing. By eliminating the need for solvent swapping or drying-to-dryness steps required by THF-based methods, the facility saves significant energy costs and reduces batch cycle times. Moreover, the high recovery rate of C4-C7 ketones due to their moderate boiling points means that solvent consumption per kilogram of product is significantly lowered. This efficiency translates directly into a lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for high-purity pharmaceutical intermediates. Additionally, the use of less toxic solvents compared to methylene chloride reduces the regulatory burden and costs associated with hazardous waste disposal and worker safety monitoring.
- Cost Reduction in Manufacturing: The elimination of complex concentration and re-dissolution steps inherently lowers utility consumption and labor hours per batch. Since the ketone solvent effectively extracts the product directly from the reaction mixture, there is no need for multiple solvent exchanges, which are notoriously expensive in terms of both material loss and energy usage. The high yield (up to 85%) further amplifies these savings by maximizing the output from every kilogram of expensive chiral or achiral starting material purchased. This efficiency creates a robust margin buffer that can absorb fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The solvents utilized in this process, such as methyl isobutyl ketone and methyl ethyl ketone, are commodity chemicals produced on a massive global scale. Unlike specialized or restricted solvents, their supply is stable and resilient to geopolitical disruptions. This availability ensures that production schedules for Citalopram intermediates can be maintained without interruption. Furthermore, the simplified process flow reduces the risk of batch failures due to operational complexity, guaranteeing a consistent and reliable flow of material to downstream formulation partners.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial tonnage is straightforward because the phase separation behavior remains consistent regardless of scale. The reduced toxicity profile of the solvent system aligns perfectly with modern green chemistry initiatives and strict environmental regulations. By minimizing the generation of hazardous waste and lowering the emission of volatile organic compounds (VOCs), manufacturers can operate with fewer environmental permits and lower compliance costs. This sustainability angle is increasingly becoming a key differentiator when qualifying suppliers for major pharmaceutical contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cyclization technology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a transparent view of the process capabilities. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios.
Q: Why are C4-C7 ketone solvents preferred over toluene for Citalopram synthesis?
A: C4-C7 ketones offer significantly higher solubility for both the diol reactants and the final Citalopram product compared to toluene. This eliminates the need for excessive solvent volumes and simplifies the post-reaction extraction process, leading to higher yields (up to 85% vs 75%).
Q: How does this process improve environmental compliance?
A: By replacing highly toxic solvents like methylene chloride and reducing the total volume of organic solvents required, the process minimizes hazardous waste generation. Additionally, the moderate boiling point of ketones allows for efficient solvent recovery and recycling.
Q: What is the typical purity achievable with this cyclization method?
A: The patented method consistently achieves HPLC purities exceeding 99.5%, with specific examples demonstrating up to 99.8% purity for S-Citalopram, meeting stringent pharmaceutical standards without complex purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Citalopram Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, high-yield synthetic routes in the competitive landscape of antidepressant manufacturing. Our technical team has extensively analyzed the advancements presented in patent CN104262306B and possesses the expertise to implement this ketone-mediated cyclization at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this process are fully realized in practice. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying the stringent purity specifications required for API intermediates, guaranteeing that every batch meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this optimized route can reduce your overall procurement costs. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you secure a sustainable, cost-effective, and high-quality supply of Citalopram intermediates for your global operations.
