Advanced Manufacturing of High-Purity Citalopram Intermediates via Solubility Differentiation
The pharmaceutical industry continuously seeks robust manufacturing pathways for high-volume antidepressants, and the synthesis of citalopram represents a critical area of process optimization. Patent CN1761658A introduces a transformative methodology for preparing 1-(4-fluorophenyl)-1,3-dihydro-5-isobenzofurancarbonitrile, a pivotal intermediate in the production of citalopram hydrobromide. This technical disclosure addresses a long-standing challenge in organic synthesis: the efficient separation of structurally similar by-products that traditionally compromise yield and purity. By leveraging specific solubility characteristics rather than relying solely on complex chromatographic techniques, this invention offers a scalable solution that aligns perfectly with the rigorous demands of modern good manufacturing practices (GMP). For R&D directors and procurement specialists, understanding this patented approach is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering consistent quality.
Furthermore, the strategic implementation of this process mitigates the risks associated with hazardous reagents often found in legacy synthetic routes. The transition from dangerous reducing agents to safer alternatives, coupled with a streamlined purification protocol, underscores a commitment to both operational safety and environmental compliance. As global demand for mental health medications continues to rise, the ability to produce key building blocks like 1-(4-fluorophenyl)-1,3-dihydro-5-isobenzofurancarbonitrile with high efficiency becomes a competitive advantage. This report analyzes the technical nuances of Patent CN1761658A to demonstrate how it facilitates cost reduction in pharmaceutical intermediates manufacturing while ensuring the highest standards of chemical integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
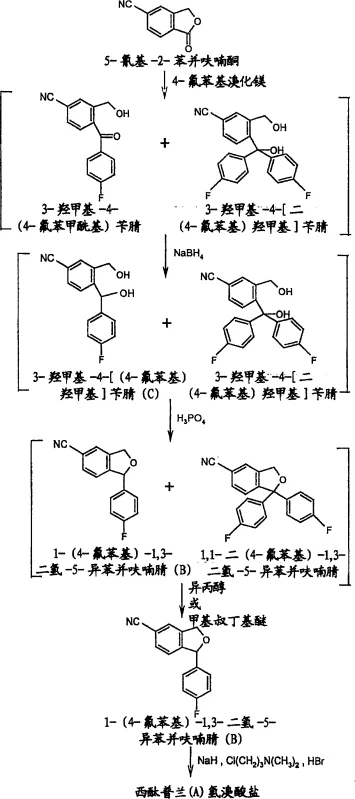
Historically, the synthesis of citalopram precursors has been plagued by the formation of persistent impurities that are chemically analogous to the desired product. Conventional methods, such as those described in earlier patents like US 4,136,193, typically involve the reaction of 5-cyano-2-benzofuranone with a Grignard reagent followed by reduction and cyclization. However, a significant drawback of these traditional routes is the unavoidable generation of 1,1-bis(4-fluorophenyl)-1,3-dihydro-5-isobenzofurancarbonitrile during the Grignard addition step. This bis-arylated by-product possesses physical and chemical properties that are remarkably similar to the target mono-arylated intermediate, making separation extremely difficult using standard crystallization or extraction techniques. Consequently, manufacturers often face substantial yield losses or are forced to employ expensive and time-consuming purification methods to meet regulatory purity specifications.
Moreover, the reliance on aggressive reducing agents like lithium aluminum hydride in older protocols introduces significant safety hazards and operational complexities. These reagents require strictly anhydrous conditions and specialized handling equipment, which increases the capital expenditure and operational risk for large-scale production facilities. The inability to effectively remove the bis-aryl impurity not only affects the final drug quality but also creates bottlenecks in the supply chain, as additional processing steps extend lead times and increase the overall cost of goods sold. For supply chain heads, these inefficiencies translate into vulnerability regarding supply continuity and price stability, highlighting the urgent need for a more refined synthetic strategy.
The Novel Approach
The methodology disclosed in Patent CN1761658A revolutionizes this landscape by introducing a targeted purification step based on differential solubility. Instead of attempting to prevent the formation of the bis-aryl impurity entirely, which is kinetically challenging, the process accepts its formation and subsequently removes it through a highly selective washing procedure. The core innovation lies in the discovery that the target intermediate, 1-(4-fluorophenyl)-1,3-dihydro-5-isobenzofurancarbonitrile, is practically insoluble in specific solvents such as isopropanol or methyl tert-butyl ether, whereas the troublesome 1,1-bis(4-fluorophenyl) impurity exhibits high solubility in these same media. This fundamental physicochemical difference allows for the isolation of the pure product through simple filtration, effectively bypassing the need for complex chromatographic separation.
In addition to the purification breakthrough, the process optimizes the reduction step by utilizing sodium borohydride in an aqueous alkaline medium. This modification replaces hazardous hydride reagents with a safer, more manageable alternative that can be operated at mild temperatures, specifically below 15°C. The combination of safer reaction conditions and a robust purification protocol results in a streamlined workflow that is inherently more suitable for commercial scale-up. By integrating these improvements, the novel approach ensures that the final intermediate achieves a purity level exceeding 98%, with the critical impurity content reduced to less than 0.5%, thereby setting a new benchmark for quality in the production of high-purity OLED material and pharmaceutical precursors.
Mechanistic Insights into Grignard Addition and Solubility-Based Purification
The chemical mechanism underpinning this process begins with the nucleophilic attack of 4-fluorophenylmagnesium bromide on the carbonyl carbon of 5-cyano-2-benzofuranone. While the primary pathway leads to the desired ketone intermediate, a competing reaction occurs where a second equivalent of the Grignard reagent attacks the newly formed ketone or reacts directly with the starting material to form a tertiary alcohol by-product. This side reaction generates 3-hydroxymethyl-4-[bis(4-fluorophenyl)hydroxymethyl]benzonitrile, which persists through the subsequent reduction step unchanged. Upon acid-catalyzed cyclization using 60% phosphoric acid, both the desired diol and the bis-aryl diol undergo dehydration and ring closure to form their respective isobenzofuran structures. The structural similarity between these two cyclic products is what traditionally makes them so difficult to separate.
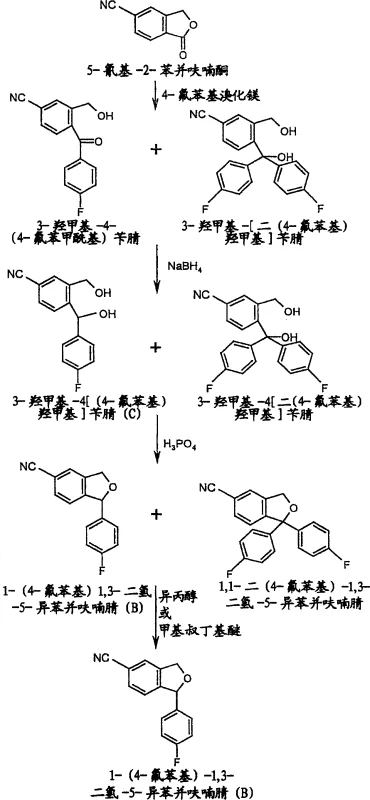
The breakthrough mechanism involves the exploitation of lattice energy differences between the mono-aryl and bis-aryl isobenzofurans. When the crude reaction mixture, containing both cyclic compounds, is treated with a solvent like isopropanol, the thermodynamic stability of the bis-aryl impurity in the solution phase is significantly higher than that of the target molecule. The target 1-(4-fluorophenyl)-1,3-dihydro-5-isobenzofurancarbonitrile precipitates out of the solution as a crystalline solid due to its low solubility, while the impurity remains dissolved in the mother liquor. This selective crystallization is driven by the specific intermolecular forces and steric hindrance presented by the extra phenyl ring in the impurity, which disrupts its ability to pack efficiently into a crystal lattice under these specific solvent conditions. This elegant solution transforms a complex separation problem into a straightforward unit operation.
How to Synthesize 1-(4-Fluorophenyl)-1,3-dihydro-5-isobenzofurancarbonitrile Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the efficacy of the purification step. The process initiates with the preparation of the Grignard reagent, followed by its controlled addition to the benzofuranone substrate at low temperatures to minimize exothermic risks. Following the Grignard reaction, the mixture is quenched and subjected to reduction using aqueous sodium borohydride, a step that must be monitored to ensure complete conversion of the ketone to the diol without degrading the nitrile group. The subsequent cyclization is performed in a biphasic system to facilitate heat transfer and product isolation. Finally, the critical washing step with isopropanol or methyl tert-butyl ether is executed to strip away the residual impurities, yielding the high-purity intermediate ready for the final alkylation.
- React 5-cyano-2-benzofuranone with 4-fluorophenylmagnesium bromide in THF at low temperature (-10 to 0°C) to form the ketone intermediate.
- Reduce the crude ketone mixture using aqueous sodium borohydride (NaBH4) and sodium hydroxide at temperatures below 15°C to generate the diol.
- Perform cyclization by treating the diol residue with 60% phosphoric acid in a two-phase system (water/ethyl acetate) under reflux conditions.
- Purify the crude isobenzofuran product by washing with isopropanol or methyl tert-butyl ether, exploiting the high solubility of the bis-aryl impurity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of the process described in Patent CN1761658A offers tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the downstream processing requirements. By eliminating the need for resource-intensive purification methods such as column chromatography or multiple recrystallizations from exotic solvent systems, the overall production cycle time is significantly compressed. This efficiency gain translates directly into improved throughput capabilities, allowing manufacturers to respond more agilely to market fluctuations and urgent demand spikes without compromising on the stringent quality standards required for active pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The substitution of hazardous and expensive reducing agents like lithium aluminum hydride with sodium borohydride represents a significant opportunity for cost optimization. Sodium borohydride is not only more economical on a per-kilogram basis but also reduces the overhead costs associated with safety infrastructure, waste disposal, and specialized training. Furthermore, the high selectivity of the solubility-based purification minimizes product loss, ensuring that a greater proportion of the raw material input is converted into saleable high-value output, thereby enhancing the overall economic viability of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes to a more resilient supply chain by reducing the dependency on hard-to-source reagents and complex processing equipment. The use of common industrial solvents like ethyl acetate, isopropanol, and tetrahydrofuran ensures that raw material availability is not a bottleneck. Additionally, the ability to consistently produce intermediates with impurity levels below 0.5% reduces the risk of batch rejection during quality control testing, ensuring a steady flow of compliant materials to downstream formulation partners and mitigating the risk of production stoppages.
- Scalability and Environmental Compliance: From an environmental perspective, the process aligns well with green chemistry principles by avoiding the generation of heavy metal waste and reducing the volume of organic solvent waste associated with extensive purification steps. The aqueous workup procedures and the use of safer reagents simplify the effluent treatment process, lowering the environmental footprint of the facility. This compliance with increasingly strict environmental regulations future-proofs the manufacturing asset, ensuring long-term operational continuity and reducing the liability associated with hazardous chemical handling and disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this patented synthesis route. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this process for commercial production. The answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: How does this process remove the difficult 1,1-bis(4-fluorophenyl) impurity?
A: The process utilizes a specific solubility difference where the target intermediate is practically insoluble in solvents like isopropanol or methyl tert-butyl ether, while the bis-aryl impurity remains highly soluble, allowing for effective removal via simple filtration.
Q: Why is sodium borohydride preferred over lithium aluminum hydride in this synthesis?
A: Sodium borohydride allows for the reduction to be conducted safely in an aqueous alkaline medium at mild temperatures (below 15°C), eliminating the safety hazards and strict anhydrous conditions associated with pyrophoric lithium aluminum hydride.
Q: What is the expected purity of the intermediate using this method?
A: By implementing the selective crystallization step, the process consistently yields the intermediate with a purity greater than 98%, with the critical bis-aryl impurity reduced to levels below 0.5%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(4-Fluorophenyl)-1,3-dihydro-5-isobenzofurancarbonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just laboratory-scale success; it demands a partner with deep expertise in process engineering and scale-up. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 1-(4-fluorophenyl)-1,3-dihydro-5-isobenzofurancarbonitrile meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a stable, high-quality supply of this critical antidepressant intermediate.
