Advanced Synthesis of Sugammadex Sodium: Overcoming Impurity Challenges for Commercial Scale-up
Advanced Synthesis of Sugammadex Sodium: Overcoming Impurity Challenges for Commercial Scale-up
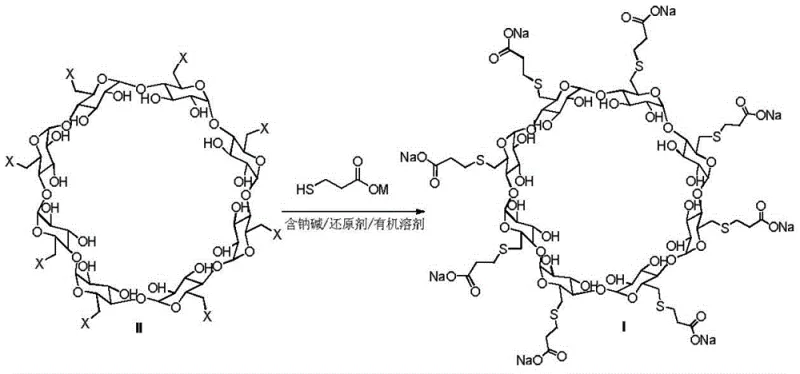
The pharmaceutical industry continuously seeks robust manufacturing routes for critical active pharmaceutical ingredients (APIs) and their intermediates, particularly for complex molecules like Sugammadex Sodium. Patent CN109021148B, published in late 2020, introduces a transformative methodology for preparing this selective muscle relaxant antagonist. The core innovation lies in the strategic integration of a reducing agent within the substitution reaction system, effectively mitigating the formation of structurally similar impurities that have historically plagued production yields. By utilizing perhalogenated gamma-cyclodextrin and 3-mercaptopropionic acid in a specialized sodium-containing alkali-reducing agent-organic solvent system, this process achieves superior control over the reaction pathway. This technical breakthrough not only enhances the purity profile but also simplifies the downstream processing requirements, positioning it as a highly attractive route for reliable API intermediate suppliers aiming to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sugammadex Sodium has been fraught with significant technical hurdles that impact both economic viability and supply chain stability. Early methodologies, such as those disclosed in U.S. Pat. No. 6,670,340, relied on a Vilsmeier-Hack reaction followed by substitution using sodium hydride in DMF. However, this conventional approach suffered from abysmal single-step reaction yields, often reported as low as 43%, with unreported purity levels that necessitated rigorous and costly purification protocols. Subsequent attempts to optimize this route, such as replacing sodium hydride with alkali metal alkoxides or amino bases, yielded marginal improvements but introduced new complexities, including the requirement for massive quantities of activated carbon or expensive liquid phase preparation instruments. These traditional processes are inherently inefficient, generating substantial waste and requiring energy-intensive purification steps like repeated dialysis or preparative HPLC, which are ill-suited for the demands of modern industrial batch production.
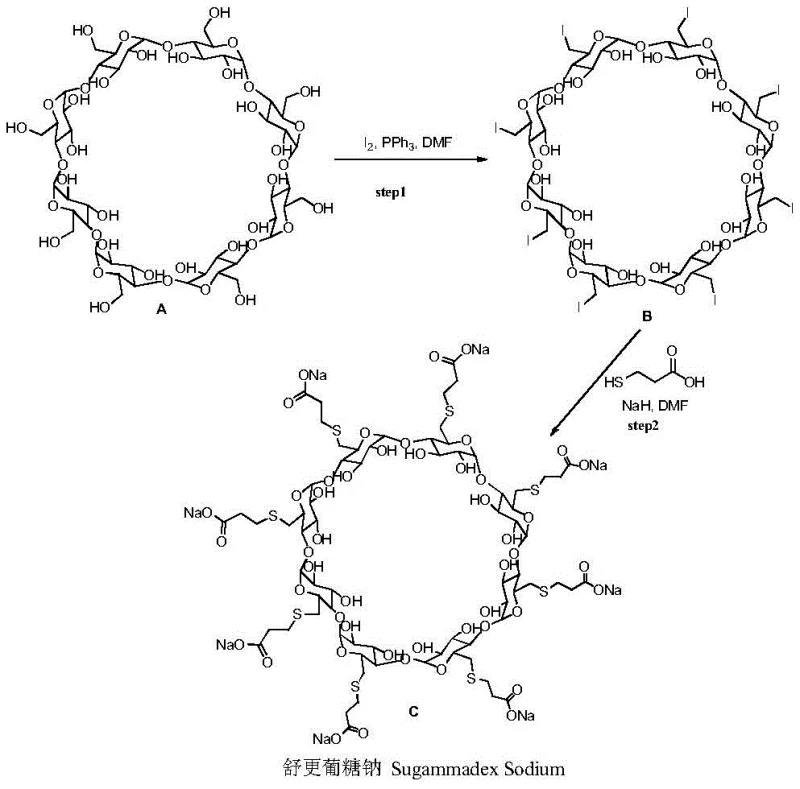
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in patent CN109021148B fundamentally reengineers the substitution step to maximize efficiency and minimize waste. The process employs perhalogenated gamma-cyclodextrin as the starting material, reacting it with 3-mercaptopropionic acid or its sodium salt in the presence of a specifically selected reducing agent. This system, operating within solvents like DMF, DMI, or DMAc at controlled temperatures of 65-75°C, effectively suppresses the formation of unwanted byproducts. The inclusion of reducing agents such as phosphines, zinc powder, or indium iodide creates a chemical environment that favors the target substitution while actively preventing oxidative degradation. This strategic modification allows for the direct isolation of crude products with yields exceeding 88% and purities approaching 90% through simple filtration and crystallization, eliminating the need for complex chromatographic separations and drastically reducing the operational burden on manufacturing facilities.
Mechanistic Insights into Reductive Substitution and Impurity Control
The mechanistic superiority of this patented process stems from its ability to dynamically manage the redox state of sulfur species throughout the reaction and purification phases. In conventional syntheses, the thioether groups and hydroxyl groups on the cyclodextrin scaffold are highly susceptible to oxidation, leading to the formation of disulfide impurities (Formula III and Formula V) and mercaptan impurities (Formula IV). These byproducts possess physicochemical properties strikingly similar to the target molecule, making their removal via standard crystallization nearly impossible. The introduction of a reducing agent disrupts this degradation pathway by maintaining a reducing environment that stabilizes the thiol intermediates. Furthermore, the process leverages a unique "waste-to-value" mechanism in the subsequent purification steps, where residual disulfide impurities are chemically reduced back to reactive thiols. These regenerated thiols then undergo a Michael addition reaction with added acrylic acid or sodium acrylate, effectively converting potential waste materials directly into the desired Sugammadex Sodium structure.
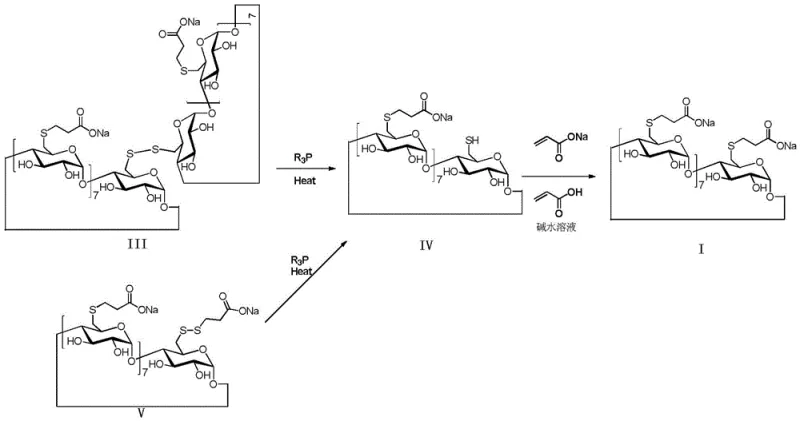
This sophisticated control over the impurity profile is critical for R&D directors focused on regulatory compliance and product consistency. The patent elucidates that the reducing agent facilitates the conversion of disulfide impurities into thiol impurities, which are subsequently trapped by the acrylic acid scavenger. This dual-action mechanism—prevention of oxidation during substitution and active conversion of impurities during purification—ensures that the final product meets stringent purity specifications without the need for excessive reprocessing. By understanding and controlling these specific mechanistic pathways, manufacturers can achieve a level of process robustness that translates directly into higher batch success rates and more predictable quality outcomes, essential factors for maintaining a reliable supply of high-purity pharmaceutical intermediates in a competitive global market.
How to Synthesize Sugammadex Sodium Efficiently
The synthesis protocol outlined in the patent provides a clear, scalable roadmap for producing Sugammadex Sodium with industrial-grade efficiency. The procedure begins with the careful preparation of the reaction mixture under inert atmosphere, followed by a controlled temperature ramp to facilitate the nucleophilic substitution. The subsequent steps involve a targeted purification strategy that utilizes the chemical reactivity of the impurities themselves to enhance overall yield. This approach minimizes the number of unit operations required, streamlining the transition from raw materials to finished API intermediate. For technical teams looking to implement this methodology, the detailed standardized synthesis steps provided below offer a comprehensive guide to replicating these high-yield results in a pilot or production setting.
- Perform nucleophilic substitution of perhalogenated gamma-cyclodextrin with 3-mercaptopropionic acid in the presence of a sodium base and a reducing agent (e.g., phosphine or zinc) in an organic solvent like DMF at 65-75°C.
- Treat the crude product with a reducing agent and acrylic acid/sodium acrylate in a polar solvent-water system at 50-70°C to convert disulfide and thiol impurities back into the target product.
- Purify the resulting crude material via recrystallization in a reducing agent-polar solvent system to obtain high-purity Sugammadex Sodium suitable for pharmaceutical use.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers profound strategic advantages that extend far beyond simple chemical yield improvements. The elimination of expensive and complex purification technologies, such as preparative HPLC or extensive activated carbon treatments, translates directly into significant cost reduction in pharmaceutical manufacturing. By simplifying the post-treatment process to basic filtration and crystallization, facilities can reduce their reliance on specialized equipment and lower their operational expenditure on consumables. Furthermore, the robust nature of the reaction conditions, which tolerate a variety of solvents and reducing agents, provides supply chain teams with greater flexibility in sourcing raw materials, thereby mitigating the risk of disruptions caused by single-source dependencies or volatile market prices for specific reagents.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by removing the need for costly purification steps like repeated dialysis or large-scale chromatography. The high single-step yield of over 88% means less raw material is wasted, and the ability to convert impurities back into the product maximizes the utility of every kilogram of input. This efficiency gain lowers the cost of goods sold (COGS) significantly, allowing for more competitive pricing strategies in the global API market without compromising on quality standards or profit margins.
- Enhanced Supply Chain Reliability: The simplified workflow reduces the overall production cycle time, enabling faster turnaround from order to delivery. Because the process does not rely on hard-to-source catalysts or complex instrumentation, it is less vulnerable to supply chain bottlenecks. The use of common industrial solvents like DMF and readily available reducing agents ensures that production can continue uninterrupted even during periods of raw material scarcity, providing a stable and dependable source of Sugammadex Sodium for downstream drug manufacturers.
- Scalability and Environmental Compliance: The patent explicitly demonstrates successful scale-up to 10kg batches, proving the feasibility of commercial amplification. The reduction in chemical waste, particularly the avoidance of massive amounts of spent activated carbon and solvent-heavy chromatography fractions, aligns with increasingly strict environmental regulations. This greener manufacturing footprint not only reduces waste disposal costs but also enhances the sustainability profile of the supply chain, a key metric for modern pharmaceutical partnerships and regulatory approvals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis method. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for stakeholders evaluating the feasibility of adopting this technology for their own production lines or sourcing strategies.
Q: How does the addition of a reducing agent improve the yield of Sugammadex Sodium?
A: The reducing agent prevents the oxidation of thioether groups and reduces formed disulfide impurities back to thiols, which can then react further to form the target product, significantly boosting overall yield compared to conventional methods.
Q: What are the critical reaction conditions for the substitution step?
A: The reaction typically requires a sodium-containing base (like NaH or sodium alkoxide), an organic solvent such as DMF or DMI, and temperatures maintained between 65-75°C under nitrogen protection to ensure complete substitution without degradation.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent demonstrates successful scale-up to 10kg batches with simplified post-treatment steps like filtration and crystallization, avoiding complex chromatography or excessive activated carbon usage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sugammadex Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for complex pharmaceutical intermediates like Sugammadex Sodium. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN109021148B are fully realized in practical, industrial applications. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch adheres to the highest international standards. Our capability to implement advanced reducing agent technologies allows us to offer a superior quality product with a consistent impurity profile, essential for the safety and efficacy of the final drug formulation.
We invite potential partners to engage with our technical procurement team to discuss how our optimized manufacturing capabilities can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to our supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most reliable and cost-effective partner in the industry.
