Scalable Large-Process Synthesis of Erexib for Global Pharmaceutical Supply Chains
Scalable Large-Process Synthesis of Erexib for Global Pharmaceutical Supply Chains
The global demand for non-steroidal anti-inflammatory drugs (NSAIDs) continues to rise, driven by an aging population and the increasing prevalence of osteoarthritis. Erexib, also known chemically as Imrecoxib (1-n-propyl-3-(4-methylphenyl)-4-(4-methylsulfonylphenyl)-2,5-dihydro-1H-2-pyrrolidone), represents a significant advancement in COX-2 selective inhibition, offering potent pain relief with a favorable safety profile. However, the commercial viability of such high-value Active Pharmaceutical Ingredients (APIs) is frequently bottlenecked by complex synthetic routes that suffer from low yields, difficult purification, and the use of hazardous reagents. Patent CN111747879B addresses these critical industrial challenges by disclosing a robust, large-process synthesis method that fundamentally redesigns the construction of the pyrrolidone core. This technical insight report analyzes the strategic advantages of this novel pathway, demonstrating how it aligns with the rigorous demands of modern pharmaceutical manufacturing for purity, cost-efficiency, and supply continuity.
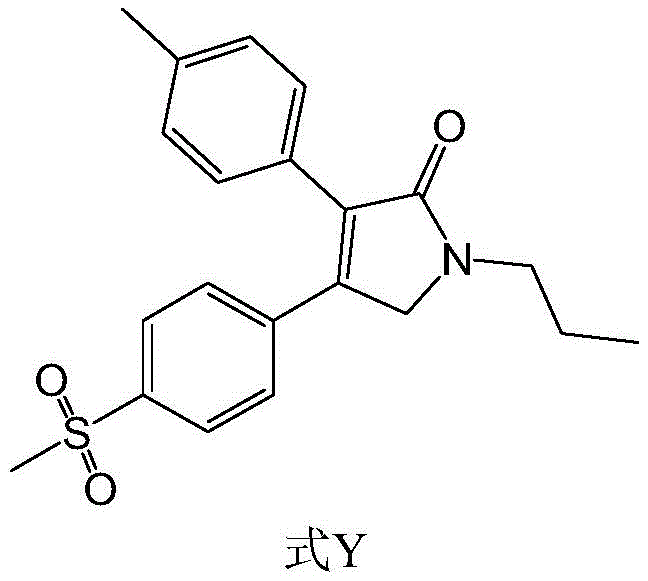
The structural complexity of Erexib, featuring a highly substituted dihydropyrrolidone ring flanked by electron-rich and electron-deficient aromatic systems, poses significant synthetic hurdles. Traditional approaches often struggle with regioselectivity and the stability of key intermediates. The methodology outlined in CN111747879B circumvents these issues through a clever manipulation of oxidation states and protecting group strategies. By shifting the synthetic logic away from unstable alpha-aminoketones towards stable acetal-protected amines, the process ensures that the molecular architecture is built upon a foundation of chemical robustness. This shift is not merely academic; it translates directly into operational excellence on the factory floor, reducing batch failure rates and ensuring that the final API meets the stringent purity specifications required by regulatory bodies worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes for Erexib and its analogues have been plagued by inherent chemical instabilities that compromise industrial scalability. A primary example, referenced in the background of the patent, involves the use of alpha-aminoketones as key building blocks. While theoretically sound, alpha-aminoketones are notoriously unstable under the alkaline conditions often required for subsequent cyclization or coupling reactions. This instability leads to rapid self-polymerization and degradation, resulting in a chaotic reaction mixture that is difficult to control. Furthermore, attempts to stabilize these intermediates by converting them into hydrochloride salts often necessitate the use of stronger bases in later steps to liberate the free amine, which in turn exacerbates side reactions and complicates the workup procedure. These factors collectively contribute to low overall yields, extensive waste generation, and a reliance on expensive chromatographic purification methods that are economically unfeasible for multi-ton production.
The Novel Approach
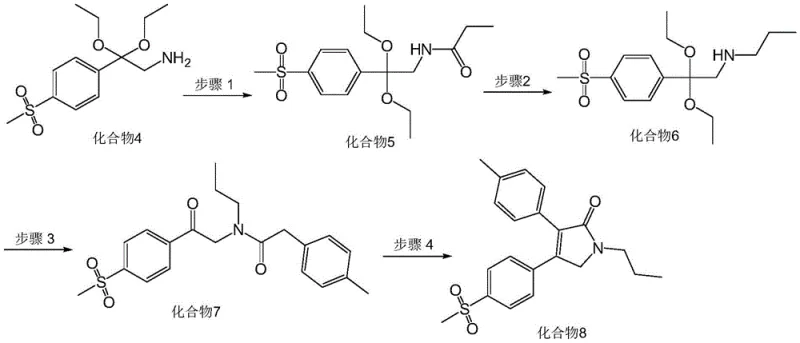
The innovative strategy presented in CN111747879B effectively neutralizes these risks by introducing a protective acyl group early in the sequence. Instead of battling the instability of a free alpha-aminoketone, the process begins with a stable amino-acetal intermediate (Compound 4). This precursor is first subjected to acylation with propionyl chloride, transforming the reactive amine into a robust amide (Compound 5). This protection step is crucial as it prevents unwanted nucleophilic attacks during subsequent transformations. Following this, a chemoselective reduction converts the amide back to an amine (Compound 6) under mild conditions, ready for coupling. This "protect-reduce-couple" sequence ensures that the reactive nitrogen center is only exposed when necessary, dramatically improving the cleanliness of the reaction profile. The final cyclization is achieved under basic conditions that are compatible with the fully assembled linear precursor, allowing for a smooth closure of the pyrrolidone ring without the decomposition issues seen in older methods.
Mechanistic Insights into Amide Reduction and Cyclization
The heart of this synthesis lies in the precise control of functional group interconversions, particularly the reduction of the intermediate amide. In Step 2 of the process, the conversion of Compound 5 to Compound 6 utilizes borane-tetrahydrofuran (BH3·THF) as the reducing agent. Unlike lithium aluminum hydride, which can be overly aggressive and reduce other sensitive functionalities like esters or sulfones, borane exhibits a high degree of chemoselectivity for amides. The mechanism involves the coordination of the boron atom to the carbonyl oxygen, followed by hydride transfer to the carbonyl carbon, eventually cleaving the C-O bond to yield the methylene group while preserving the N-C bond. This selectivity is paramount for maintaining the integrity of the methylsulfonyl group on the aromatic ring, which is essential for the biological activity of the final drug. The reaction is conducted under a nitrogen atmosphere to prevent the oxidation of the borane reagent, ensuring consistent stoichiometry and reaction kinetics throughout the batch.
Following the assembly of the linear chain via peptide-like coupling in Step 3, the final transformation (Step 4) is an intramolecular cyclization that constructs the core heterocycle. This step employs potassium carbonate in an ethanol solution, acting as a mild base to deprotonate the alpha-carbon adjacent to the ketone or activate the amide nitrogen for nucleophilic attack. The reaction conditions—heating to reflux between 80°C and 120°C—provide the necessary thermal energy to overcome the activation barrier for ring closure. Crucially, the use of ethanol as a solvent facilitates the solubility of the organic substrate while allowing the inorganic base to function effectively. The mechanism likely proceeds through an enolate intermediate that attacks the electrophilic center, followed by dehydration or elimination to establish the double bond within the dihydropyrrolidone ring. The robustness of this cyclization allows for simple isolation via cooling and filtration, bypassing the need for complex extraction protocols.
How to Synthesize Erexib Efficiently
The execution of this large-process synthesis requires strict adherence to the optimized parameters defined in the patent to maximize yield and purity. The route is designed to be telescoped where possible, minimizing the handling of intermediates and reducing solvent consumption. The initial acylation is exothermic and requires controlled addition of propionyl chloride to maintain temperature stability. The subsequent reduction step demands anhydrous conditions to preserve the activity of the borane reagent. Finally, the condensation and cyclization steps leverage standard coupling reagents (EDCI/HOBt) and inorganic bases that are readily available at commodity prices. For a detailed breakdown of the specific molar ratios, solvent volumes, and temperature profiles required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- Acylation: React the amino-acetal intermediate (Compound 4) with propionyl chloride and triethylamine in acetonitrile to form the protected amide (Compound 5).
- Reduction: Reduce the amide (Compound 5) using borane-tetrahydrofuran complex under nitrogen protection to yield the secondary amine (Compound 6).
- Condensation: Couple Compound 6 with p-methylphenylacetic acid using EDCI, HOBt, and DMAP in dichloromethane to form the linear precursor (Compound 7).
- Cyclization: Reflux Compound 7 with potassium carbonate in ethanol to induce intramolecular cyclization, yielding the final Erexib product (Compound 8).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from legacy synthesis methods to the protocol described in CN111747879B offers substantial strategic benefits. The primary advantage lies in the drastic simplification of the purification workflow. By avoiding the formation of polymeric byproducts associated with unstable aminoketones, the reaction mixtures are significantly cleaner. This cleanliness allows for purification via recrystallization rather than expensive and time-consuming column chromatography. Recrystallization is inherently more scalable, consumes less solvent, and generates less hazardous waste, directly contributing to cost reduction in pharmaceutical manufacturing. Furthermore, the use of common, non-proprietary reagents such as propionyl chloride, triethylamine, and potassium carbonate ensures that the supply chain is not vulnerable to the bottlenecks often associated with specialized catalysts or exotic ligands.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of costly purification steps and the improvement in overall yield. By stabilizing the intermediates, the process minimizes material loss due to degradation, ensuring that a higher percentage of raw materials are converted into the final valuable API. The avoidance of transition metal catalysts, which often require expensive removal steps to meet residual metal limits in pharmaceuticals, further lowers the cost of goods sold (COGS). Additionally, the ability to isolate intermediates as stable solids or oils that can be stored or transported without degradation provides flexibility in production scheduling, allowing manufacturers to optimize batch sizes and inventory levels without the risk of spoilage.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for uninterrupted API production. This synthesis route enhances reliability by utilizing starting materials that are commercially available in bulk quantities, such as 4-methylsulfonyl acetophenone and p-methylphenylacetic acid. The robustness of the chemical transformations means that the process is less sensitive to minor fluctuations in reaction conditions, leading to consistent batch-to-batch quality. This consistency reduces the likelihood of out-of-specification (OOS) results that can halt production lines. Moreover, the simplified workup procedures reduce the turnaround time between batches, enabling a faster response to market demand surges and ensuring a steady flow of material to downstream formulation partners.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this large-process method is superior to traditional routes. The reduction in solvent usage, particularly the avoidance of large volumes of chlorinated solvents for chromatography, aligns with green chemistry principles and reduces the burden on waste treatment facilities. The use of aqueous workups and ethanol-based recrystallizations simplifies solvent recovery and recycling. Scalability is inherently supported by the use of standard unit operations such as filtration, distillation, and crystallization, which are easily implemented in existing multipurpose reactors. This ease of scale-up from kilogram to metric ton scales ensures that the technology can grow with the commercial success of the drug, providing a long-term sustainable manufacturing solution that meets increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and mechanistic rationale provided in the patent documentation, offering clarity on the feasibility and advantages of the process for potential licensees and manufacturing partners.
Q: Why is the conventional alpha-aminoketone route problematic for Erexib synthesis?
A: Conventional methods often rely on alpha-aminoketones which are chemically unstable under alkaline conditions, leading to self-polymerization and low yields. The patented method avoids this by using a stable acetal-protected amine strategy.
Q: What are the key reagents used in the large-process synthesis of Erexib?
A: The process utilizes common industrial reagents including propionyl chloride for protection, borane-tetrahydrofuran for reduction, EDCI/HOBt for condensation, and potassium carbonate for the final cyclization step.
Q: How does this synthesis method improve supply chain reliability?
A: By eliminating the need for unstable intermediates and harsh purification steps like column chromatography, the method allows for robust crystallization-based purification, ensuring consistent quality and easier scale-up for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Erexib Supplier
The technological breakthroughs detailed in Patent CN111747879B represent a significant leap forward in the manufacturing of Erexib, yet translating laboratory success into commercial reality requires a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM stands at the forefront of this capability, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including the safe handling of borane reagents and the precise temperature control needed for the cyclization step. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of Erexib intermediate meets the highest international standards, providing our clients with the confidence needed to navigate the complex regulatory landscape of pharmaceutical approvals.
We invite global pharmaceutical companies and contract manufacturing organizations to collaborate with us to leverage this efficient synthesis route. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and supply chain constraints. Our technical team is ready to provide specific COA data and route feasibility assessments to demonstrate how this optimized process can enhance your bottom line. Contact our technical procurement team today to discuss how we can secure your supply of high-quality Erexib intermediates and drive your product development forward with speed and certainty.
