Scalable Green Synthesis of 3,5-Disubstituted Isoxazoles Using Water-Soluble Vitamin E Derivatives
Introduction to Green Isoxazole Synthesis Technology
The pharmaceutical and agrochemical industries are increasingly demanding sustainable manufacturing processes that minimize environmental impact while maintaining high product quality. Patent CN113582937B introduces a groundbreaking methodology for the synthesis of isoxazole compounds, a privileged scaffold found in numerous bioactive molecules ranging from antibiotics to hormone regulators. This technology leverages the unique properties of water-soluble vitamin E derivatives, specifically tocopherol methoxy polyethylene glycol succinate (TPGS-750-M), to facilitate efficient 1,3-dipolar cycloaddition reactions in an aqueous medium. By shifting away from traditional organic solvents and precious metal catalysts, this approach addresses critical pain points regarding waste generation and toxicity. The innovation lies in the formation of micellar nanoreactors that concentrate hydrophobic reactants, enabling high-yield transformations under mild conditions. For procurement and supply chain leaders, this represents a pivotal shift towards greener, more cost-effective pharmaceutical intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the isoxazole ring has relied heavily on homogeneous metal catalysis or harsh chemical conditions that pose significant operational and environmental challenges. Traditional protocols often utilize expensive transition metals such as Palladium, Copper, or Gold to catalyze coupling reactions or cycloadditions. These methods frequently require anhydrous organic solvents, strict temperature controls, and inert atmospheres, which drive up energy consumption and infrastructure costs. Furthermore, the presence of residual heavy metals in the final product necessitates complex and costly purification steps to meet stringent regulatory limits for high-purity pharmaceutical intermediates. The generation of hazardous waste streams from organic solvents and metal salts further complicates disposal and compliance, creating bottlenecks in the commercial scale-up of complex heterocyclic compounds.
The Novel Approach
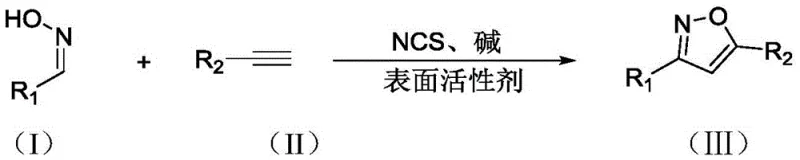
In stark contrast, the method described in CN113582937B utilizes a surfactant-mediated aqueous system that operates at room temperature, eliminating the need for external heating and specialized equipment. The core of this innovation is the use of TPGS-750-M, which self-assembles into micelles in water, creating hydrophobic pockets that solubilize organic substrates like aldoximes and alkynes. This "on-water" effect significantly accelerates reaction kinetics without the use of toxic organic co-solvents. The process employs N-chlorosuccinimide (NCS) as a mild oxidant to generate nitrile oxides in situ, which then undergo cycloaddition with alkynes. This metal-free strategy not only simplifies the reaction setup but also ensures that the final product is free from heavy metal contamination, thereby reducing lead time for high-purity isoxazole derivatives and streamlining the downstream purification workflow.
Mechanistic Insights into Micellar Catalyzed 1,3-Dipolar Cycloaddition
The mechanistic pathway of this transformation is driven by the synergistic interaction between the oxidant, the base, and the micellar environment. Initially, the aldoxime substrate is oxidized by N-chlorosuccinimide (NCS) in the presence of a base, such as triethylamine, to generate a reactive nitrile oxide intermediate. In conventional organic solvents, nitrile oxides are prone to dimerization, leading to unwanted furoxan byproducts. However, within the confined space of the TPGS-750-M micelles, the local concentration of the alkyne dipolarophile is significantly enhanced relative to the nitrile oxide. This proximity effect favors the intermolecular 1,3-dipolar cycloaddition over dimerization, resulting in superior regioselectivity and yield. The hydrophobic interior of the micelle acts as a nanoreactor, shielding the reactive intermediates from bulk water while facilitating the necessary orbital overlap for ring closure.
From an impurity control perspective, this mechanism offers distinct advantages. The mild reaction conditions (room temperature) prevent thermal degradation of sensitive functional groups often present in complex drug candidates. Additionally, the absence of transition metals eliminates the risk of metal-catalyzed side reactions such as homocoupling of alkynes. The surfactant itself can be recovered and recycled from the aqueous phase after product extraction, contributing to a circular economy model. This level of control over the reaction environment ensures a clean impurity profile, which is critical for meeting the rigorous specifications required for API intermediate manufacturing. The robustness of the micellar system allows for a broad substrate scope, accommodating various electron-withdrawing and electron-donating groups on both the aldoxime and alkyne components without compromising efficiency.
How to Synthesize 3,5-Disubstituted Isoxazoles Efficiently
Implementing this green synthesis route requires careful attention to the preparation of the micellar medium and the sequential addition of reagents to maximize the formation of the nitrile oxide intermediate. The process is designed to be operationally simple, avoiding the need for gloveboxes or cryogenic cooling. Operators should ensure the TPGS-750-M solution is freshly prepared to maintain optimal micelle integrity. The reaction progress can be monitored via TLC or HPLC, with typical completion times ranging from 6 to 16 hours depending on the steric hindrance of the substrates. The detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Prepare a 2wt.% aqueous solution of the surfactant TPGS-750-M and add the aldoxime substrate and N-chlorosuccinimide (NCS).
- Stir the mixture at room temperature for approximately 4 hours to allow initial activation.
- Add the base (triethylamine) and the alkyne substrate, continuing stirring at room temperature for 6-16 hours to complete the cycloaddition.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this aqueous micellar catalysis technology offers transformative benefits for the supply chain, primarily driven by the elimination of costly inputs and the simplification of waste management. By replacing precious metal catalysts with inexpensive organic oxidants and reusable surfactants, manufacturers can achieve cost reduction in pharmaceutical intermediate manufacturing without sacrificing quality. The reliance on water as the primary solvent removes the volatility and flammability risks associated with organic solvents, lowering insurance and storage costs while enhancing facility safety. Furthermore, the simplified workup procedure, which involves basic extraction and chromatography, reduces the man-hours required for production, thereby increasing overall throughput capacity.
- Cost Reduction in Manufacturing: The most significant economic driver is the complete removal of palladium, copper, or gold catalysts, which are subject to volatile market pricing and supply constraints. Eliminating these metals also removes the expensive downstream processing steps required to reduce metal residues to ppm levels, which often involve specialized scavengers or repeated crystallizations. Additionally, the ability to recycle the TPGS-750-M surfactant from the aqueous layer further decreases the raw material cost per kilogram of product. This cumulative effect results in substantial cost savings that improve the margin profile for high-volume fine chemical intermediates.
- Enhanced Supply Chain Reliability: The starting materials for this process, including aldoximes, terminal alkynes, and NCS, are commodity chemicals with robust global supply chains, reducing the risk of production delays due to raw material shortages. Unlike specialized ligands or air-sensitive catalysts that require cold chain logistics, the reagents used here are stable and easy to transport. This stability ensures consistent production schedules and reliable delivery timelines for clients. The process tolerance to moisture and oxygen also means that manufacturing can proceed without the need for highly controlled inert environments, further de-risking the supply chain against equipment failures.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage is straightforward because heat transfer and mixing are more efficient in aqueous systems compared to viscous organic media. The process generates minimal hazardous waste, aligning with increasingly strict environmental regulations regarding VOC emissions and heavy metal discharge. This compliance advantage prevents potential regulatory shutdowns and fines, ensuring long-term business continuity. The green nature of the process also enhances the brand value of the final pharmaceutical product, appealing to end-users who prioritize sustainability in their sourcing decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this micellar catalysis technology. These answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on yield expectations, substrate compatibility, and environmental impact. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this route into existing production lines.
Q: How does this method eliminate heavy metal contamination?
A: Unlike traditional methods requiring Palladium, Copper, or Gold catalysts, this protocol utilizes N-chlorosuccinimide (NCS) as an oxidant in a micellar system, completely removing the need for expensive transition metals and subsequent purification steps.
Q: What are the solvent benefits of using TPGS-750-M?
A: The process uses water as the primary solvent instead of volatile organic compounds (VOCs). The surfactant forms nanoreactors that solubilize organic reactants, allowing for zero solvent discharge and significantly reduced environmental impact.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the reaction operates at room temperature with simple workup procedures (extraction and chromatography). The absence of sensitive metal catalysts and the use of water make it highly robust and scalable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoxazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting sustainable and efficient synthetic routes like the one described in CN113582937B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of isoxazole intermediate meets the highest global standards. We are committed to leveraging our technical expertise to optimize this green chemistry platform for your specific portfolio needs.
We invite you to engage with our technical procurement team to discuss how this technology can be tailored to your project requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this metal-free aqueous process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both your product quality and your bottom line.
