Advanced Enzymatic Synthesis of Chiral Beta-Hydroxy Amino Acid Esters for Carbapenem Antibiotic Manufacturing
The pharmaceutical industry continuously seeks robust and scalable pathways for the production of critical antibiotic precursors, particularly for the carbapenem class which represents the frontline defense against resistant bacterial strains. Patent CN112941043B introduces a groundbreaking advancement in this domain by disclosing a specifically engineered carbonyl reductase mutant capable of synthesizing chiral beta'-hydroxy-beta-amino acid esters with unprecedented efficiency. This biocatalytic innovation addresses the long-standing challenges associated with traditional chemical synthesis, offering a route that combines high stereoselectivity with industrial feasibility. The technology leverages site-directed mutagenesis on a wild-type enzyme derived from Sporobolomyces salmonicolor, resulting in a biocatalyst that exhibits significantly enhanced catalytic activity and stability under process-relevant conditions. For R&D directors and procurement specialists, this patent signifies a pivotal shift towards greener, more cost-effective manufacturing of high-purity pharmaceutical intermediates. The ability to achieve high substrate loading and exceptional yield positions this technology as a cornerstone for reliable supply chain strategies in the competitive antibiotic market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral beta-hydroxy-beta-amino acid esters has relied heavily on transition metal-catalyzed dynamic kinetic resolution, which presents substantial drawbacks for large-scale commercial production. These conventional chemical methods often necessitate the use of expensive noble metal complexes and sophisticated chiral ligands that require multi-step synthesis themselves, driving up the raw material costs significantly. Furthermore, the reaction conditions for these metal-catalyzed processes are frequently harsh, involving organic solvents and extreme temperatures that pose safety risks and environmental compliance challenges. A critical bottleneck in these traditional routes is the difficulty in removing trace metal residues from the final product, which is unacceptable for pharmaceutical applications requiring stringent purity specifications. The formation of unwanted diastereomers is also a common issue, complicating downstream purification and reducing the overall process yield. Consequently, manufacturers face high production costs and extended lead times, making the supply of these key intermediates vulnerable to market fluctuations and regulatory scrutiny.
The Novel Approach
In stark contrast, the novel biocatalytic approach detailed in the patent utilizes a genetically modified carbonyl reductase that operates under mild, aqueous conditions, effectively circumventing the limitations of metal catalysis. This enzymatic route eliminates the need for toxic heavy metals and complex ligand systems, thereby simplifying the purification workflow and reducing the environmental footprint of the manufacturing process. The mutant enzyme demonstrates remarkable tolerance to high substrate concentrations, enabling efficient conversion rates that are essential for cost reduction in pharmaceutical intermediate manufacturing. By employing a phthalimide protecting group strategy, the process ensures stability during the reduction phase while allowing for controlled deprotection later in the synthesis sequence. This method not only enhances the stereoselectivity towards the desired (2S,3R) configuration but also minimizes the generation of by-products that would otherwise require resource-intensive separation steps. The shift to a biological catalyst thus represents a strategic upgrade in process chemistry, aligning with modern green chemistry principles while delivering superior economic and operational performance.
Mechanistic Insights into Carbonyl Reductase Mutant Catalysis
The core of this technological breakthrough lies in the precise engineering of the carbonyl reductase active site through site-directed mutagenesis, specifically targeting amino acid residues that interact directly with the substrate. The patent identifies key mutation sites such as 242 and 245, where substitutions like M242V and Q245S drastically alter the enzyme's conformational dynamics to favor the binding of the beta'-carbonyl-beta-(phthalimide) methylbutyrate substrate. These modifications enhance the hydride transfer efficiency from the cofactor NADPH to the carbonyl group, resulting in a significant increase in specific activity compared to the wild-type enzyme. The structural adjustments create a more rigid and complementary binding pocket that enforces the correct orientation of the substrate, thereby ensuring high stereoselectivity for the (2S,3R) isomer. This mechanistic precision is crucial for R&D teams aiming to minimize impurity profiles and streamline the synthesis of complex chiral molecules. The enzyme's ability to function effectively in the presence of co-solvents like DMSO further expands its applicability in processing substrates with lower aqueous solubility.
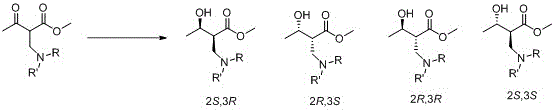
Control of the impurity profile is achieved through the enzyme's inherent ability to discriminate between the pro-chiral faces of the ketone substrate, effectively suppressing the formation of the (2S,3S), (2R,3S), and (2R,3R) diastereomers. The patent data indicates that the mutant variants can achieve diastereomeric excess (de) values of 98% or more, which is a critical parameter for ensuring the quality of the final antibiotic API. This high level of stereocontrol reduces the burden on downstream crystallization and chromatography steps, leading to substantial cost savings and improved throughput. The stability of the phthalimide protecting group under the enzymatic reaction conditions prevents premature hydrolysis, which is a common issue with benzoyl protection in aqueous biocatalysis. By maintaining the integrity of the protecting group, the process ensures that the amino functionality remains masked until the appropriate stage of the synthesis, preventing side reactions that could compromise yield. This robust mechanism underscores the reliability of the biocatalytic route for producing high-purity pharmaceutical intermediates at a commercial scale.
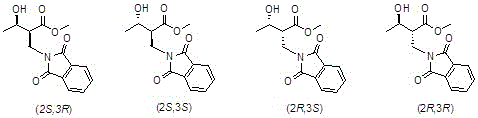
How to Synthesize (2S,3R)-3-hydroxy-2-(phthalimide) methylbutyrate Efficiently
The implementation of this synthesis route involves a streamlined workflow that begins with the construction of the recombinant expression system and culminates in the biocatalytic conversion of the ketone substrate to the chiral alcohol. Detailed standard operating procedures for the expression of the mutant enzyme and the optimization of reaction parameters such as pH and temperature are essential for replicating the high yields reported in the patent. The process leverages the high substrate tolerance of the mutant, allowing for concentrations up to 250 g/L, which is a key factor in achieving economic viability for bulk manufacturing. Operators must ensure precise control of the cofactor regeneration system, typically utilizing glucose dehydrogenase to recycle NADP+, to maintain continuous catalytic turnover. The following guide outlines the critical steps required to transition this laboratory-scale innovation into a robust industrial process.
- Construct recombinant plasmids containing the mutated carbonyl reductase gene (e.g., M242V/Q245S) and transform into E. coli expression hosts.
- Culture the engineered strains in LB medium with IPTG induction to express the mutant enzyme, then harvest cells via centrifugation.
- Perform biocatalytic reduction using wet cells, substrate (250 g/L), NADP+, and glucose dehydrogenase in buffer at 30°C to achieve >93.5% yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic technology offers transformative benefits that extend beyond mere technical performance, directly impacting the bottom line and operational resilience. The elimination of transition metal catalysts removes a significant cost center associated with both the purchase of expensive metals and the validation of their removal from the final product. This simplification of the manufacturing process leads to a drastically simplified supply chain, reducing the dependency on specialized metal suppliers and mitigating risks associated with raw material volatility. Furthermore, the high yield and substrate loading capabilities of the mutant enzyme mean that less raw material is wasted, enhancing the overall material efficiency of the production line. These factors combine to create a more predictable and cost-effective sourcing strategy for critical antibiotic intermediates, ensuring continuity of supply for downstream API manufacturers.
- Cost Reduction in Manufacturing: The transition from metal-catalyzed chemistry to biocatalysis eliminates the need for costly noble metals and complex chiral ligands, which are significant expense drivers in traditional synthesis. By removing the requirement for extensive metal scavenging and purification steps, the process reduces both material costs and processing time, leading to substantial cost savings in API intermediate manufacturing. The high catalytic efficiency of the mutant enzyme allows for lower enzyme loading relative to the substrate, further optimizing the cost structure of the reaction. Additionally, the use of aqueous buffers instead of organic solvents reduces waste disposal costs and aligns with environmental regulations, avoiding potential fines or compliance fees. This holistic reduction in operational expenses makes the biocatalytic route a financially superior choice for long-term production contracts.
- Enhanced Supply Chain Reliability: The reliance on fermentation-derived enzymes ensures a sustainable and scalable source of catalyst that is not subject to the geopolitical and mining constraints often associated with precious metals. The robustness of the engineered strain allows for consistent production of the biocatalyst, minimizing the risk of batch-to-batch variability that can disrupt manufacturing schedules. High substrate tolerance means that production capacity can be increased without proportional increases in reactor volume, effectively debottlenecking the supply chain for high-purity pharmaceutical intermediates. This scalability ensures that suppliers can meet surging demand for carbapenem precursors without compromising on quality or delivery timelines. Consequently, partners can rely on a stable supply of intermediates that supports their own production planning and inventory management strategies.
- Scalability and Environmental Compliance: The process operates under mild conditions in aqueous media, significantly reducing the generation of hazardous organic waste and lowering the energy consumption required for heating and cooling. This green chemistry profile simplifies environmental compliance and reduces the burden on waste treatment facilities, making it easier to scale up from pilot to commercial production without regulatory hurdles. The high stereoselectivity minimizes the formation of isomeric impurities, reducing the need for energy-intensive separation processes like preparative chromatography. This efficiency in resource utilization supports the industry's move towards sustainable manufacturing practices while maintaining high throughput. The combination of environmental safety and operational scalability makes this technology an ideal candidate for modern, compliant chemical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this carbonyl reductase mutant technology in industrial settings. These answers are derived directly from the patent data and provide clarity on the performance capabilities and operational requirements of the new biocatalytic route. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing infrastructure. The information provided here serves as a foundational reference for discussions on process optimization and quality assurance.
Q: How does the carbonyl reductase mutant improve stereoselectivity compared to wild-type enzymes?
A: The mutant features specific amino acid substitutions (e.g., at positions 242 and 245) that reshape the active site pocket, enforcing strict binding orientation for the (2S,3R) isomer while rejecting others, achieving >99% ee.
Q: What are the substrate concentration limits for this enzymatic process?
A: The engineered mutant demonstrates robust activity at high substrate loading, successfully converting concentrations up to 250 g/L while maintaining yields above 93.5%, which is critical for industrial throughput.
Q: Does this biocatalytic route eliminate the need for transition metal catalysts?
A: Yes, the process relies entirely on biological catalysis in aqueous buffer, removing the requirement for expensive metal complexes and the subsequent costly purification steps needed to remove metal residues.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2S,3R)-3-hydroxy-2-(phthalimide) methylbutyrate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced patent technologies like CN112941043B into commercial reality, offering partners a secure pathway to high-quality chiral intermediates. As a specialized CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial volume is seamless and efficient. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of (2S,3R)-3-hydroxy-2-(phthalimide) methylbutyrate meets the exacting standards required for carbapenem antibiotic synthesis. We understand the critical nature of these intermediates in the global supply chain and are committed to delivering consistency, reliability, and technical excellence in every shipment.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this enzymatic route can optimize your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this biocatalytic process for your production lines. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project requirements. Our team is ready to support your journey towards more sustainable and cost-effective manufacturing of complex pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
