Scalable Production of High-Purity Vinylaromatic Intermediates via Green Microwave Chemistry
Scalable Production of High-Purity Vinylaromatic Intermediates via Green Microwave Chemistry
The chemical industry is currently witnessing a paradigm shift towards sustainable and high-efficiency synthetic methodologies, particularly in the realm of fine chemical and pharmaceutical intermediate production. A pivotal development in this sector is documented in Chinese Patent CN102010279A, which discloses a novel preparation method for aryl ethylene derivatives. This technology leverages the synergistic effects of focused microwave radiation and polyethylene glycol (PEG-400) as a green solvent to facilitate palladium-catalyzed cross-coupling reactions. Unlike traditional thermal processes that often suffer from prolonged reaction times and hazardous solvent usage, this innovation achieves completion within a mere 10 to 15 minutes. For R&D directors and process chemists, this represents a significant breakthrough in reaction engineering, offering a pathway to drastically reduce energy consumption while maintaining high product yields of up to 95%. The ability to synthesize complex vinylaromatic scaffolds rapidly positions this technology as a critical asset for reliable pharmaceutical intermediate supplier networks aiming to optimize their manufacturing pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl ethylene derivatives via the Heck cross-coupling reaction has been plagued by several inherent inefficiencies that hinder large-scale commercial viability. Traditional protocols frequently rely on expensive and toxic phosphine or nitrogen-based ligands to stabilize the palladium catalyst, which not only inflates raw material costs but also introduces complex impurity profiles that are difficult to remove during purification. Furthermore, conventional methods typically utilize polar aprotic solvents such as dimethylformamide (DMF) or dimethylacetamide (DMA), which pose significant environmental and safety hazards due to their toxicity and difficulty in disposal. From a process engineering perspective, these legacy methods often require extended reaction times ranging from 11 to 37 hours to reach acceptable conversion rates, creating bottlenecks in production scheduling and limiting the overall throughput of manufacturing facilities. The combination of high catalyst loading, expensive ligands, and toxic solvents results in a process that is economically burdensome and environmentally unsustainable for modern green chemistry standards.
The Novel Approach
In stark contrast to these legacy issues, the methodology outlined in patent CN102010279A introduces a ligand-free palladium chloride catalytic system operating within a PEG-400 solvent matrix under focused microwave irradiation. This approach fundamentally alters the reaction kinetics by utilizing the dielectric heating properties of PEG-400 to achieve rapid and uniform heating of the reaction mixture, thereby accelerating the oxidative addition and reductive elimination steps of the catalytic cycle. The elimination of phosphine ligands simplifies the reaction mixture, reducing the formation of phosphine oxide byproducts and streamlining the downstream purification process. Moreover, the use of PEG-400, a non-toxic and biodegradable polymer, replaces hazardous organic solvents, aligning the synthesis with stringent environmental regulations and reducing the cost associated with solvent recovery and waste treatment. This novel technique demonstrates exceptional versatility, successfully coupling various iodoarenes with olefins such as acrylic acid and acrylonitrile to produce high-value intermediates with remarkable speed and efficiency.
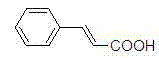
Mechanistic Insights into PdCl2-Catalyzed Microwave Heck Coupling
The mechanistic superiority of this process lies in the unique interaction between the microwave field and the polar PEG-400 solvent, which creates a superheated environment that promotes the formation of the active Pd(0) species from the PdCl2 precursor without the need for external reducing agents or stabilizing ligands. In this ligand-free system, the palladium catalyst undergoes a standard Heck catalytic cycle involving oxidative addition of the aryl iodide, migratory insertion of the olefin, and beta-hydride elimination to release the vinylaromatic product. The microwave irradiation specifically enhances the rate of the oxidative addition step, which is often the rate-determining step in palladium-catalyzed cross-couplings, by providing the necessary activation energy instantaneously throughout the bulk solution. This results in a dramatic reduction in reaction time from hours to minutes, allowing for precise control over the reaction progress and minimizing the potential for thermal degradation of sensitive functional groups. The stability of the catalyst in the PEG-400 medium also suggests a potential for catalyst recycling, further enhancing the atom economy and sustainability of the process for industrial applications.
From an impurity control perspective, the absence of bulky ligands and the use of a high-boiling, non-volatile solvent like PEG-400 significantly simplify the isolation of the target molecule. The workup procedure described involves simple acidification to pH 1-3 followed by ether extraction, which effectively separates the organic product from the aqueous PEG phase and inorganic salts. This phase separation behavior is crucial for maintaining high purity levels, as it prevents the co-elution of catalyst residues and solvent impurities that are common in DMF-based systems. The resulting products, such as cinnamic acid derivatives and stilbenes, exhibit clean spectral data with minimal side products, indicating a highly selective transformation. This level of purity is essential for downstream applications in pharmaceutical synthesis, where strict regulatory limits on heavy metal residues and organic impurities must be met to ensure patient safety and drug efficacy.
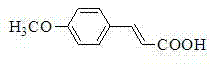
How to Synthesize Cinnamic Acid Derivatives Efficiently
The synthesis of cinnamic acid derivatives and related vinylaromatic compounds using this microwave-assisted protocol offers a robust and scalable route for process chemists looking to optimize their production workflows. The procedure is characterized by its operational simplicity, requiring only the mixing of readily available starting materials—iodoarenes and olefins—with a catalytic amount of palladium chloride and a base in PEG-400. The reaction is then driven to completion in a dedicated microwave reactor equipped with temperature and power control, ensuring reproducibility and safety. Following the brief irradiation period, the reaction mixture undergoes a straightforward acidic workup and extraction sequence to isolate the crude product, which can be further purified by standard silica gel chromatography if necessary. This streamlined approach eliminates the need for inert atmosphere techniques often required for sensitive ligand-based systems, making it accessible for a wide range of laboratory and pilot plant settings.
- Combine iodoarene, olefin compound, PdCl2 catalyst (1-5 mol%), and potassium carbonate base in PEG-400 solvent.
- Subject the reaction mixture to focused microwave radiation at 100-130°C and 8-10W power for 10-15 minutes.
- Acidify the mixture to pH 1-3 with HCl, extract with ether, dry over magnesium sulfate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this microwave-assisted synthesis technology presents a compelling value proposition centered on cost reduction and supply reliability. The primary economic driver is the elimination of expensive phosphine ligands, which are often subject to volatile market pricing and supply chain disruptions, thereby stabilizing the raw material cost structure. Additionally, the drastic reduction in reaction time from nearly two days to merely 15 minutes translates into a massive increase in equipment utilization rates, allowing existing reactor infrastructure to produce significantly higher volumes of intermediates without capital expenditure on new hardware. The use of PEG-400 as a solvent not only reduces hazardous waste disposal costs but also simplifies the logistics of solvent handling and storage, as it is non-flammable and less toxic than traditional alternatives. These factors collectively contribute to a more resilient and cost-effective supply chain capable of meeting the demanding delivery schedules of global pharmaceutical and agrochemical clients.
- Cost Reduction in Manufacturing: The transition to a ligand-free catalytic system removes one of the most significant cost components in traditional Heck coupling reactions, as high-purity phosphine ligands are notoriously expensive and often required in stoichiometric or near-stoichiometric amounts. By utilizing inexpensive palladium chloride in low loadings (1-5 mol%) and eliminating the ligand entirely, the direct material cost per kilogram of product is substantially decreased. Furthermore, the recyclability of the PEG-400 solvent and the potential for catalyst recovery mean that the consumption of consumables is minimized, leading to long-term operational savings that compound over large production runs. This economic efficiency makes the process highly competitive for the commercial scale-up of complex pharmaceutical intermediates where margin pressure is intense.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as iodoarenes, acrylic acid, and potassium carbonate ensures a stable and diversified supply base, reducing the risk of production stoppages due to single-source supplier failures. The robustness of the reaction conditions, which tolerate a variety of functional groups and do not require stringent exclusion of moisture or oxygen, further enhances operational reliability by reducing the sensitivity of the process to minor variations in raw material quality. This resilience is critical for maintaining continuous supply to downstream customers, particularly in the event of global logistical disruptions or raw material shortages. The ability to rapidly switch between different aryl ethylene derivatives by simply changing the iodoarene input adds flexibility to the manufacturing schedule, allowing for agile responses to changing market demands.
- Scalability and Environmental Compliance: The green chemistry credentials of this process, specifically the use of non-toxic PEG-400 and the absence of hazardous volatile organic compounds (VOCs), facilitate easier regulatory approval and permitting for new manufacturing lines. As environmental regulations become increasingly stringent globally, adopting a solvent system that is biodegradable and safe for workers provides a strategic advantage in maintaining social license to operate. The scalability of microwave technology has advanced significantly, with continuous flow microwave reactors now available for ton-scale production, ensuring that the efficiency gains observed at the gram scale can be translated to industrial quantities. This alignment with sustainability goals not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer among eco-conscious partners and stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this microwave-assisted Heck coupling technology in industrial settings. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a transparent view of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into their existing production portfolios.
Q: What are the primary advantages of using PEG-400 over traditional solvents like DMF in this Heck coupling?
A: PEG-400 serves as a non-toxic, biodegradable, and microwave-absorbent solvent that eliminates the environmental hazards associated with polar aprotic solvents like DMF or DMA, while simultaneously simplifying the post-reaction workup process.
Q: Is the palladium catalyst reusable in this microwave-assisted protocol?
A: Yes, the patent explicitly states that the palladium chloride catalyst system is recyclable, which significantly reduces the consumption of precious metals and lowers the overall cost of goods for large-scale manufacturing.
Q: How does the reaction time compare to conventional thermal heating methods?
A: The microwave-assisted method completes the reaction in merely 10 to 15 minutes, whereas conventional ligand-free or phosphine-ligand methods typically require heating for 11 to 37 hours, representing a drastic improvement in throughput efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cinnamic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the microwave-assisted Heck coupling described in CN102010279A for producing high-value vinylaromatic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiency and purity benefits of this novel chemistry are fully realized at an industrial level. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of cinnamic acid derivatives or related intermediates meets the exacting standards required by the global pharmaceutical industry. We are committed to leveraging our technical expertise to optimize these green processes, delivering cost-effective and sustainable solutions that drive value for our partners.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits of switching to this ligand-free, microwave-driven protocol. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance your supply chain resilience and accelerate your time to market.
