Revolutionizing 3-Trifluoromethyl-1,2,4-Triazole Production: A Cost-Effective DMF-Based Strategy for Global Pharmaceutical Supply Chains
Revolutionizing 3-Trifluoromethyl-1,2,4-Triazole Production: A Cost-Effective DMF-Based Strategy for Global Pharmaceutical Supply Chains
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and economically viable synthetic routes for high-value heterocyclic scaffolds, particularly those incorporating fluorine motifs which are critical for enhancing metabolic stability and bioavailability. Patent CN114920707B, published in April 2023, introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds that fundamentally shifts the paradigm of carbon source utilization in heterocyclic synthesis. This technology leverages the ubiquitous organic solvent N,N-dimethylformamide (DMF) not merely as a reaction medium but as an active carbon synthon, thereby streamlining the synthetic pathway and reducing the reliance on specialized, costly reagents. The significance of this innovation cannot be overstated for R&D directors and procurement managers alike, as 1,2,4-triazole cores are foundational structures in numerous blockbuster drugs and bioactive inhibitors, ranging from anticonvulsants to Factor IXa inhibitors.
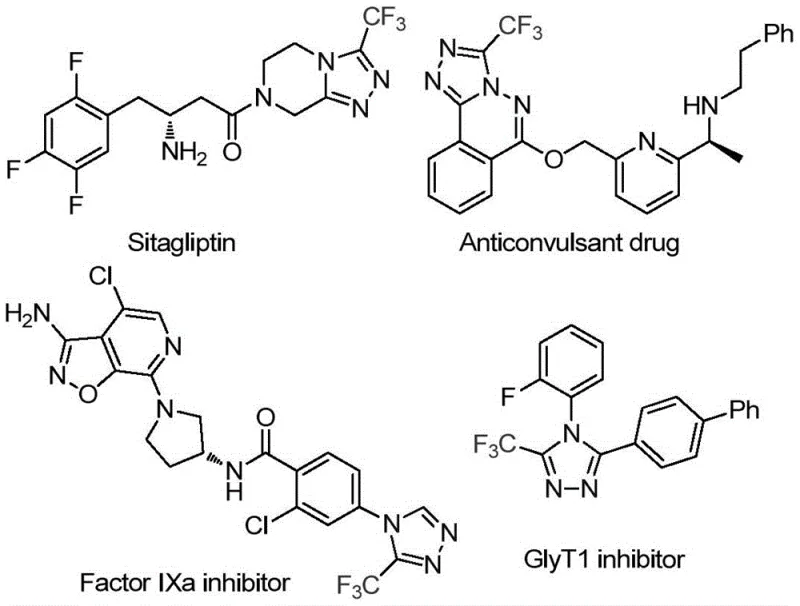
As illustrated in the structural diversity of bioactive molecules shown above, the 1,2,4-triazole ring system is a privileged scaffold found in widely used therapeutics such as Sitagliptin and various kinase inhibitors. The presence of the trifluoromethyl group on this ring further amplifies the pharmacological profile by modulating lipophilicity and electronic properties, making the efficient construction of this specific motif a high-priority target for process chemistry teams. Traditional methods for installing these groups often involve multi-step sequences, harsh reaction conditions, or the use of hazardous gases, creating bottlenecks in both development timelines and commercial manufacturing. The disclosed invention addresses these pain points directly by offering a one-pot, tandem cyclization strategy that operates under remarkably mild and practical conditions, utilizing molecular iodine as a promoter to activate the solvent itself. This approach not only simplifies the operational workflow but also opens new avenues for the rapid generation of diverse compound libraries for drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-trifluoromethyl-1,2,4-triazoles has been plagued by several intrinsic inefficiencies that hinder large-scale adoption and increase the overall cost of goods sold (COGS) for pharmaceutical intermediates. Conventional routes frequently necessitate the use of pre-functionalized starting materials that are expensive to procure and often unstable, requiring specialized storage and handling protocols that strain supply chain logistics. Furthermore, many existing methodologies rely on stoichiometric amounts of strong bases, toxic heavy metal catalysts, or gaseous reagents that pose significant safety risks and environmental compliance challenges in a production setting. The requirement for strictly anhydrous and anaerobic conditions in many traditional protocols adds another layer of complexity, demanding expensive inert gas lines and dried solvents, which drastically inflates the operational expenditure for contract development and manufacturing organizations (CDMOs). Additionally, the atom economy of these older methods is often poor, generating substantial quantities of chemical waste that require costly disposal procedures, thereby conflicting with modern green chemistry principles and sustainability goals.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the method described in patent CN114920707B presents a elegantly simple yet highly effective solution that utilizes DMF as a dual-purpose reagent, acting simultaneously as the solvent and the source of the carbon atom required for ring closure. This innovative strategy eliminates the need for external carbon sources or complex formylating agents, effectively collapsing multiple synthetic steps into a single, efficient transformation driven by molecular iodine. The reaction proceeds smoothly under an air atmosphere at temperatures between 110°C and 130°C, removing the necessity for glovebox techniques or rigorous exclusion of moisture and oxygen, which significantly lowers the barrier to entry for implementation in standard chemical plants. By employing readily available trifluoroethyliminohydrazide derivatives as the nitrogen source, the process ensures a high degree of modularity, allowing for the easy introduction of various substituents on the aromatic ring without compromising yield or purity. This streamlined approach not only accelerates the timeline from bench-scale discovery to pilot plant production but also enhances the overall safety profile of the manufacturing process by avoiding hazardous reagents.
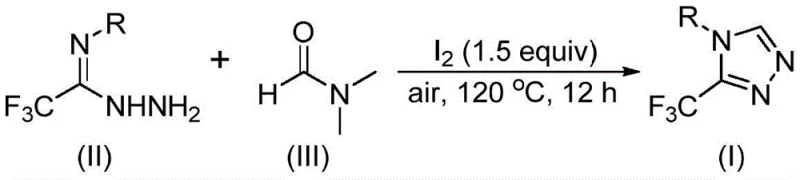
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic elegance of this transformation lies in the unique ability of molecular iodine to activate the robust DMF molecule, facilitating its cleavage and subsequent incorporation into the triazole framework through a tandem cyclization sequence. Detailed analysis suggests that the reaction can proceed via two potential pathways depending on which fragment of the DMF molecule acts as the carbon source: either the formyl group or the N-methyl group. When the formyl group serves as the carbon source, it undergoes a condensation reaction with the hydrazide moiety of the starting material to generate a hydrazone intermediate, which then cyclizes intramolecularly with the elimination of dimethylamine to yield the final triazole product. Alternatively, if the N-methyl group is utilized, the iodine first reacts with DMF to generate an reactive amine salt species, which subsequently undergoes nucleophilic addition with the hydrazide followed by elimination and oxidative aromatization steps. This dual-pathway capability underscores the versatility of the method and highlights the critical role of iodine not just as an oxidant but as a Lewis acid activator that lowers the energy barrier for C-N and C-C bond formation.
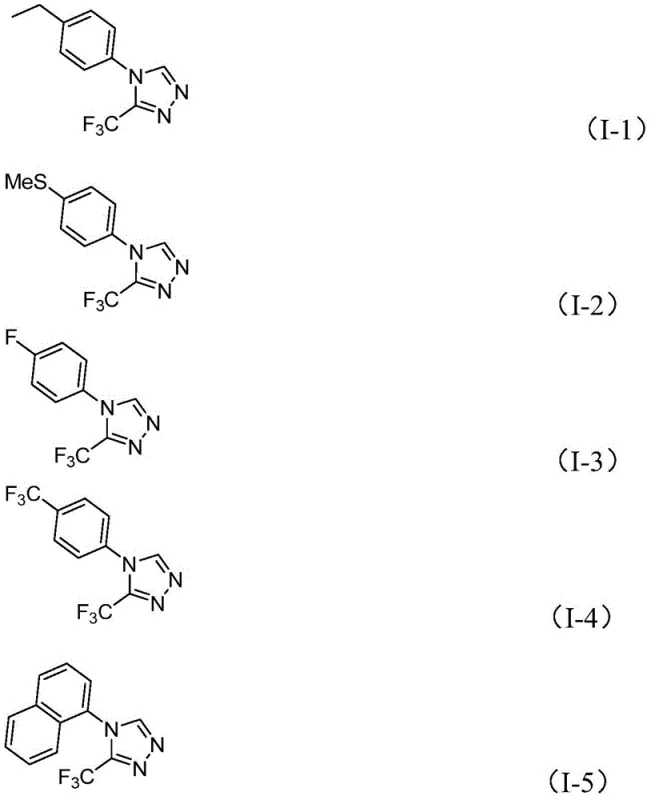
From an impurity control perspective, this mechanism offers distinct advantages for ensuring high product quality, which is paramount for regulatory compliance in pharmaceutical manufacturing. The specificity of the iodine-mediated activation minimizes side reactions such as over-oxidation or polymerization that are common in radical-based cyclizations, leading to cleaner reaction profiles and simplified downstream purification. The broad substrate scope demonstrated in the patent data indicates that the electronic nature of the substituent R on the aromatic ring has a manageable impact on the reaction efficiency, with yields remaining robust across electron-rich and electron-deficient systems. For instance, substrates bearing electron-withdrawing groups like trifluoromethyl or halogens, as well as electron-donating groups like methoxy or alkyl chains, all successfully convert to the desired triazole products with good to excellent yields. This tolerance allows medicinal chemists to explore a wide chemical space around the triazole core without needing to re-optimize reaction conditions for each new analog, thereby accelerating lead optimization campaigns.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and ensure reproducibility, leveraging the simplicity of the reagent setup while maintaining control over the reaction kinetics. The process begins with the precise weighing of trifluoroethyliminohydrazide and molecular iodine, typically in a molar ratio of 1:1.5, which are then dissolved in DMF, serving as both the reaction medium and the carbon donor. The mixture is heated to a controlled temperature range of 110°C to 130°C and stirred under ambient air pressure for a duration of 10 to 15 hours, allowing sufficient time for the tandem cyclization and aromatization steps to reach completion. Following the reaction period, the crude mixture undergoes a straightforward workup procedure involving filtration to remove inorganic salts, followed by silica gel column chromatography to isolate the pure 3-trifluoromethyl-1,2,4-triazole compound.
- Combine trifluoroethyliminohydrazide, molecular iodine (1.5 equivalents), and DMF solvent in a reaction vessel under air atmosphere.
- Heat the reaction mixture to a temperature range of 110-130°C and maintain stirring for 10 to 15 hours to ensure complete conversion.
- Upon completion, perform standard post-treatment including filtration, washing, and silica gel column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DMF-based synthetic route represents a strategic opportunity to optimize raw material costs and enhance supply chain resilience for critical pharmaceutical intermediates. The primary economic driver here is the substitution of expensive, specialized carbon sources with DMF, a commodity chemical that is produced on a massive global scale and is available from numerous suppliers at very low cost. This shift significantly reduces the direct material cost per kilogram of the final product, contributing to substantial margin improvements for high-volume manufacturing campaigns. Furthermore, the elimination of the need for anhydrous solvents and inert gas protection simplifies the infrastructure requirements for production facilities, allowing for the utilization of standard stainless steel reactors without the need for specialized lining or drying systems. This reduction in capital expenditure and operational complexity translates directly into faster turnaround times and increased production capacity, enabling suppliers to respond more agilely to fluctuating market demands.
- Cost Reduction in Manufacturing: The utilization of DMF as a dual-function reagent eliminates the procurement of separate formylating agents or C1 synthons, which are often priced at a premium due to their specialized nature and lower production volumes. By consolidating the solvent and reactant roles into a single, cheap commodity chemical, the overall bill of materials is drastically reduced, leading to significant cost savings that can be passed down the supply chain or retained as improved gross margins. Additionally, the use of molecular iodine as a promoter is advantageous because it is relatively inexpensive compared to transition metal catalysts like palladium or rhodium, and it does not require complex ligand systems that add further cost and purification burdens. The simplified workup procedure, which avoids extensive aqueous extractions or complex quenching steps, also reduces the consumption of auxiliary chemicals and utilities such as water and energy, further driving down the variable costs associated with production.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved by the reliance on universally available starting materials like DMF and simple aromatic amines, which are not subject to the same supply constraints or geopolitical risks as rare earth metals or exotic reagents. This abundance ensures a stable and continuous supply of raw materials, minimizing the risk of production stoppages due to vendor shortages or logistics disruptions. The robustness of the reaction conditions, which tolerate air and moisture, also means that the quality of incoming raw materials does not need to be held to excessively stringent specifications regarding water content, allowing for the use of technical grade solvents where appropriate. This flexibility in raw material specification broadens the supplier base and increases negotiating power for procurement teams, ensuring long-term supply security for critical intermediates used in API manufacturing.
- Scalability and Environmental Compliance: The scalability of this process is inherently high due to the absence of hazardous gases or highly exothermic steps that typically limit batch sizes in traditional heterocyclic synthesis. The ability to run the reaction under air atmosphere simplifies the engineering controls required for scale-up, reducing the need for complex pressure vessels or explosion-proof equipment. From an environmental standpoint, the atom economy is improved by incorporating the solvent into the product, reducing the volume of organic waste generated per unit of product. While DMF itself requires careful handling and recovery, the overall waste profile is cleaner compared to multi-step routes involving chlorinated solvents or heavy metal residues, facilitating easier compliance with increasingly stringent environmental regulations and reducing the costs associated with waste treatment and disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel triazole synthesis method, providing clarity for stakeholders evaluating its potential integration into their existing workflows. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these nuances is essential for assessing the feasibility of adopting this technology for specific project requirements and for anticipating any potential challenges during the technology transfer phase.
Q: What is the primary advantage of using DMF in this triazole synthesis?
A: DMF serves a dual function as both the reaction solvent and the essential carbon source (C1 synthon), eliminating the need for separate, expensive formylating agents and significantly simplifying the raw material procurement process.
Q: Does this method require strict anhydrous or anaerobic conditions?
A: No, one of the key operational benefits of this patented process is that it proceeds efficiently under standard air atmosphere without the need for rigorous anhydrous or oxygen-free environments, reducing equipment and operational costs.
Q: What is the substrate scope for the R-group in this reaction?
A: The method demonstrates broad compatibility with various substituted aryl groups, including those with electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens and trifluoromethyl, allowing for diverse derivative synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this DMF-mediated cyclization technology for the efficient production of high-value pharmaceutical intermediates and are fully equipped to leverage these advancements for our global clientele. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and compliant with international quality standards. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-trifluoromethyl-1,2,4-triazole delivered meets the exacting requirements of modern drug development pipelines. Our commitment to process excellence means we can rapidly adapt this novel synthetic route to meet your specific volume needs while maintaining the highest levels of safety and environmental stewardship.
We invite you to engage with our technical procurement team to discuss how this cost-effective synthesis method can be tailored to your specific project goals and to request a Customized Cost-Saving Analysis for your target molecules. By partnering with us, you gain access to specific COA data and route feasibility assessments that demonstrate the tangible benefits of this technology in terms of yield, purity, and overall cost efficiency. Let us help you optimize your supply chain and accelerate your time-to-market with our reliable supply of high-purity 3-trifluoromethyl-1,2,4-triazole intermediates, backed by our proven track record in process innovation and customer-centric service.
