Advanced Two-Step Synthesis of 1,2,3,6-Tetrahydrophthalimide for Commercial Scale-Up
The chemical landscape for producing high-value cyclic imides is undergoing a significant transformation, driven by the need for greener processes and higher purity standards. A pivotal advancement in this domain is detailed in patent CN110642774B, which outlines a novel preparation method for 1,2,3,6-tetrahydrophthalimide. This specific intermediate is critical for various downstream applications, ranging from pharmaceutical synthesis to specialized polymer additives. The core innovation lies in a fractional step approach that fundamentally alters the reaction kinetics compared to traditional one-pot systems. By decoupling the initial ammonolysis from the subsequent dehydration cyclization, the process effectively mitigates the formation of viscous colloidal byproducts that typically plague conventional heating methods. This technical breakthrough not only enhances the selectivity of the reaction but also establishes a robust foundation for industrial scalability, addressing key pain points for R&D directors and supply chain managers alike who seek reliable [pharmaceutical intermediates] with consistent quality profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing of 1,2,3,6-tetrahydrophthalimide has relied on three primary methodologies, each fraught with significant operational and economic inefficiencies. The urea method, for instance, necessitates heating reactants to approximately 144°C in DMF, a process that frequently triggers self-polymerization of urea and generates substantial amounts of difficult-to-remove byproducts, resulting in yields hovering around 80%. Similarly, the ammonia water method suffers from high energy consumption due to the extensive dehydration required in the later stages, alongside the risk of hydrolyzing the phthalic anhydride at elevated temperatures, which severely compromises selectivity. Furthermore, the hydrogenation method, while effective, introduces severe safety hazards associated with high-pressure hydrogen and Raney nickel catalysts, alongside prolonged reaction times of 10 to 16 hours that bottleneck production throughput. These legacy processes collectively contribute to elevated waste treatment costs and inconsistent product quality, creating a pressing need for [cost reduction in fine chemical intermediates manufacturing] through process innovation.
The Novel Approach
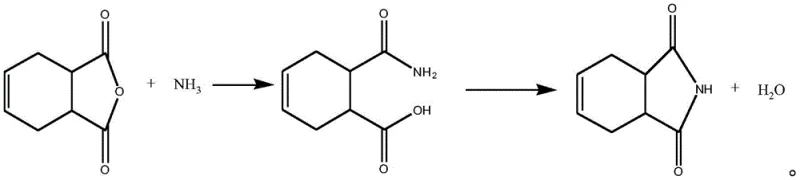
In stark contrast to these legacy systems, the novel approach delineated in the patent data introduces a sophisticated two-step protocol that optimizes both thermodynamic and kinetic parameters. The first stage involves a low-temperature reaction between 1,2,3,6-tetrahydrophthalic anhydride and an ammonia source, strictly maintained between 0-30°C to form a stable intermediate without triggering premature side reactions. This intermediate is then transferred to a solvent system, such as xylene or trimethylbenzene, preheated to 130-180°C for dehydration ring-closing. This separation of reaction phases effectively bypasses the critical temperature window of 80-110°C where viscous colloidal byproducts typically form, thereby ensuring a much cleaner reaction profile. The result is a comprehensive two-step yield that consistently exceeds 95%, with product purity reaching upwards of 99.5%, demonstrating a clear pathway for [commercial scale-up of complex pharmaceutical intermediates] that prioritizes both efficiency and environmental compliance.
Mechanistic Insights into Low-Temperature Ammonolysis and Dehydration Cyclization
The mechanistic superiority of this synthesis route is rooted in the precise control of the ring-opening and ring-closing events. In the initial low-temperature phase, the ammonia source attacks the carbonyl groups of the tetrahydrophthalic anhydride to form an ammonium carboxylate intermediate. By keeping the temperature below 30°C, the system prevents the thermal degradation of the ammonia source and avoids the hydrolysis of the anhydride ring, which is a common failure mode in aqueous systems. The reaction is monitored until the residual anhydride content drops below 0.5 wt%, ensuring near-quantitative conversion before the second stage begins. This meticulous control over the intermediate stability is crucial, as it sets the stage for a clean cyclization process that is free from the polymeric impurities often associated with direct high-temperature ammonolysis.
Following the formation of the stable intermediate, the dehydration ring-closing reaction is initiated in a high-boiling solvent. The choice of solvent, specifically xylene or trimethylbenzene, is not arbitrary; these solvents facilitate azeotropic distillation, allowing for the continuous removal of water generated during the imide formation. This continuous removal of water drives the equilibrium towards the product side, significantly enhancing the conversion rate. Moreover, the batch feeding of the intermediate into the hot solvent prevents localized concentration spikes that could lead to side reactions. The process effectively avoids the formation of viscous byproducts in the 80-110°C range, leading to a final product with exceptional purity and a white acicular solid appearance, which is highly desirable for downstream [high-purity pharmaceutical intermediates] applications.
How to Synthesize 1,2,3,6-Tetrahydrophthalimide Efficiently
The operational execution of this synthesis route requires precise adherence to the temperature and feeding protocols established in the patent data to ensure reproducibility and safety. The process begins with the careful addition of the ammonia source to the anhydride under strict thermal control, followed by a controlled transfer to the dehydration vessel. This structured approach minimizes operator error and maximizes batch consistency, which is essential for meeting the rigorous specifications demanded by global supply chains. For a detailed breakdown of the specific reagent quantities, stirring rates, and vacuum parameters required to replicate this high-yield process, please refer to the standardized synthesis guide provided below.
- Mix 1,2,3,6-tetrahydrophthalic anhydride with an ammonia source at 0-30°C to form a stable reaction intermediate.
- Transfer the intermediate into a preheated solvent system (xylene or trimethylbenzene) at 130-180°C.
- Maintain temperature for over 2 hours while removing water under vacuum, then cool and recrystallize to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this novel synthesis method offers substantial advantages that extend beyond mere chemical yield. The elimination of dangerous high-pressure hydrogenation steps and the removal of heavy metal catalysts like Raney nickel significantly reduce the regulatory burden and safety infrastructure costs associated with production. This simplification of the process flow translates directly into [cost reduction in pharmaceutical intermediates manufacturing] by lowering capital expenditure on specialized safety equipment and reducing the complexity of waste stream management. Furthermore, the ability to recycle excess ammonia gas and reuse reaction-generated water creates a closed-loop system that minimizes raw material waste, offering a compelling value proposition for organizations focused on sustainability and operational efficiency.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive transition metal catalysts and the subsequent purification steps required to remove metal residues, which are often costly and time-consuming. By utilizing readily available solvents like xylene and avoiding the high energy consumption associated with evaporating large volumes of water in the ammonia water method, the overall production cost is significantly optimized. This efficiency allows for a more competitive pricing structure without compromising on the stringent quality standards required for [high-purity pharmaceutical intermediates], ensuring that the final cost per kilogram is favorable for large-volume contracts.
- Enhanced Supply Chain Reliability: The robustness of this two-step method ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply chains for downstream drug manufacturers. The use of stable intermediates and controlled reaction conditions reduces the risk of batch failures, thereby [reducing lead time for high-purity pharmaceutical intermediates] by minimizing the need for re-processing or rejection. Additionally, the raw materials required, such as tetrahydrophthalic anhydride and ammonia, are commodity chemicals with stable global availability, mitigating the risk of supply disruptions that can occur with specialized or hazardous reagents.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that are easily controlled in large-scale reactors without the safety risks associated with high-pressure hydrogenation. The absence of waste gas generation and the ability to recycle byproducts align with increasingly strict environmental regulations, reducing the liability and cost associated with environmental compliance. This green chemistry approach not only future-proofs the manufacturing process against regulatory changes but also enhances the corporate social responsibility profile of the supply chain, making it an attractive option for [reliable pharmaceutical intermediates supplier] partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1,2,3,6-tetrahydrophthalimide. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent data, providing clarity on the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their own synthesis pipelines or for procurement officers assessing supplier capabilities.
Q: How does this method improve purity compared to the urea method?
A: By separating the ring-opening and ring-closing steps, this method avoids the high-temperature side reactions and self-polymerization common in the urea method, achieving purity levels exceeding 99%.
Q: Is the process safe for large-scale manufacturing?
A: Yes, unlike hydrogenation methods that require high-pressure hydrogen and dangerous catalysts, this process operates at atmospheric pressure with recyclable ammonia, significantly enhancing operational safety.
Q: What are the environmental benefits of this synthesis route?
A: The process generates no waste gas during the reaction phase, allows for the recycling of excess ammonia, and produces water that can be reused, drastically reducing environmental treatment costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,3,6-Tetrahydrophthalimide Supplier
The technical potential of this two-step synthesis route represents a significant opportunity for optimizing the production of 1,2,3,6-tetrahydrophthalimide, and NINGBO INNO PHARMCHEM is uniquely positioned to capitalize on this innovation. As a seasoned CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale patent data to industrial reality is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 1,2,3,6-tetrahydrophthalimide meets the exacting standards required by global R&D directors and procurement managers.
We invite you to initiate a dialogue regarding your specific supply chain requirements and explore how our optimized manufacturing processes can drive value for your organization. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our production efficiencies can translate into tangible economic benefits for your projects. We encourage you to contact our technical procurement team to obtain specific COA data and route feasibility assessments, ensuring that your partnership with us is built on a foundation of transparency, technical excellence, and mutual success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
