Revolutionizing 1,2,3,6-Tetrahydrophthalimide Production with Novel Low-Temperature Catalytic Technology
The chemical industry is currently witnessing a significant paradigm shift in the synthesis of critical heterocyclic intermediates, driven by the urgent need for greener, more efficient, and higher-purity manufacturing processes. A prime example of this technological evolution is found in the preparation of 1,2,3,6-tetrahydrophthalimide, a vital building block extensively utilized in the production of pharmaceutical agents, agrochemicals, and advanced polymer additives. The traditional methods for synthesizing this compound have long been plagued by issues such as low selectivity, harsh reaction conditions, and significant environmental burdens. However, the emergence of patent CN110642774A introduces a groundbreaking fractional step method that fundamentally redefines the production landscape. This innovative approach utilizes a controlled low-temperature ammonolysis followed by a precise dehydration ring-closing reaction in a specialized solvent system. By decoupling the ring-opening and ring-closing stages, this technology effectively mitigates the formation of viscous colloidal byproducts that typically occur in the 80-110°C range during conventional one-step processes. For R&D Directors and Technical Procurement Managers seeking reliable partners for complex intermediate synthesis, understanding the mechanistic superiority of this patent is crucial for securing a competitive supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 1,2,3,6-tetrahydrophthalimide has relied heavily on three primary methodologies, each carrying distinct and substantial drawbacks that hinder large-scale efficiency and product quality. The urea method, while common, suffers from severe limitations due to the use of organic solvents like DMF which complicate downstream purification and lead to significant product loss during imine formation. Furthermore, urea is prone to self-polymerization side reactions at the required high temperatures, generating a plethora of by-products that drastically lower selectivity and overall yield, often capping efficiency below optimal levels. The ammonia water method presents a different set of challenges, primarily revolving around excessive energy consumption required for dehydration in the later stages of the reaction. The exothermic nature of the reaction between ammonia water and phthalic anhydride necessitates complex batch feeding and cooling systems, increasing both equipment costs and labor intensity. Additionally, the high water content in ammonia water promotes the hydrolysis of phthalic anhydride at elevated temperatures, which is detrimental to imine generation and results in lower selectivity. Finally, the hydrogenation method, although capable of decent yields, introduces significant safety hazards associated with high-pressure hydrogen gas and the use of pyrophoric catalysts like Raney nickel, making it less desirable for modern, safety-conscious manufacturing facilities.
The Novel Approach
In stark contrast to these legacy techniques, the novel fractional step method outlined in the patent data offers a sophisticated solution that addresses the root causes of inefficiency and impurity. By initiating the reaction at a controlled low temperature between 0-30°C, the process ensures the formation of a stable reaction intermediate without triggering the thermal degradation or side reactions seen in traditional high-heat methods. This initial step allows for precise control over the stoichiometry of the ammonia source, whether it be ammonia gas, aqueous ammonia, or ammonium bicarbonate, ensuring minimal waste. The subsequent transfer of this intermediate into a preheated solvent system, such as xylene or trimethylbenzene, facilitates a clean dehydration ring-closing reaction. Crucially, this method avoids the problematic temperature window of 80-110°C where viscous colloidal byproducts typically form, thereby maintaining a high level of selectivity throughout the synthesis. The result is a streamlined process that not only simplifies operation and control but also significantly enhances the environmental profile of the manufacturing cycle by enabling the recycling of excess ammonia and minimizing waste gas emissions.
Mechanistic Insights into Low-Temperature Ammonolysis and Dehydration Cyclization
The core innovation of this synthesis route lies in its meticulous separation of the ring-opening and ring-closing mechanisms, a strategy that provides unparalleled control over the reaction pathway and impurity profile. In the first stage, the 1,2,3,6-tetrahydrophthalic anhydride undergoes a nucleophilic attack by the ammonia source under strictly regulated low-temperature conditions. This thermal restraint is critical; it prevents the rapid, uncontrolled exothermic reactions that often lead to the hydrolysis of the anhydride moiety, a common failure point in the ammonia water method. By maintaining the system between 0-30°C, the reaction kinetics favor the formation of the desired amide-acid intermediate while suppressing competing pathways. The use of ammonia gas in an anhydrous environment further elevates the selectivity to nearly 99.9%, as it eliminates water-induced side reactions entirely. This precise control ensures that the intermediate entering the second stage is of exceptional quality, setting the foundation for a high-yield final product.
The second stage involves the dehydration ring-closing reaction, which is executed in a high-boiling aromatic solvent system under vacuum conditions. The choice of solvent, specifically xylene or trimethylbenzene, is not arbitrary; these solvents provide the necessary thermal stability and solubility characteristics to facilitate the removal of water generated during cyclization. By preheating the solvent to 130-180°C before introducing the intermediate, the process bypasses the dangerous 80-110°C zone where polymeric gums and tars are known to form. The continuous removal of water under a vacuum of -0.01 MPa to -0.04 MPa drives the equilibrium towards the completion of the ring closure, ensuring full conversion of the intermediate. This mechanistic precision results in a comprehensive two-step yield exceeding 95% and a final product purity that consistently surpasses 99%, meeting the stringent requirements of high-end pharmaceutical and electronic chemical applications.
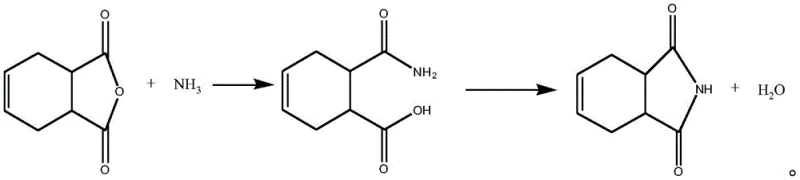
How to Synthesize 1,2,3,6-Tetrahydrophthalimide Efficiently
Implementing this advanced synthesis route requires a disciplined adherence to the specific operational parameters defined in the patent to ensure reproducibility and safety at scale. The process begins with the careful charging of 1,2,3,6-tetrahydrophthalic anhydride into a reaction vessel equipped with efficient cooling capabilities to maintain the critical 0-30°C range during ammonolysis. Once the stable intermediate is confirmed via sampling, typically when residual anhydride drops below 0.5 wt%, the material is transferred to the dehydration reactor. This second reactor must be pre-charged with the appropriate volume of xylene or trimethylbenzene and heated to the target cyclization temperature prior to feeding. The intermediate is added in batches over a period of 20 to 40 minutes to prevent local overheating and ensure uniform reaction kinetics. Throughout the holding period of over 2 hours, the system operates under vacuum to continuously strip out the water of reaction, driving the equilibrium forward. Detailed standardized synthetic steps see the guide below.
- Mix 1,2,3,6-tetrahydrophthalic anhydride with an ammonia source at 0-30°C to form a stable reaction intermediate.
- Transfer the intermediate into a preheated solvent system (xylene or trimethylbenzene) at 130-180°C.
- Maintain temperature for over 2 hours while removing generated water under vacuum to complete dehydration ring-closing.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis technology translates directly into tangible strategic advantages regarding cost stability, supply reliability, and regulatory compliance. The elimination of hazardous reagents and the simplification of the reaction workflow significantly reduce the operational complexity associated with manufacturing this key intermediate. Unlike the hydrogenation method which requires specialized high-pressure equipment and rigorous safety protocols, this ambient pressure or mild vacuum process can be executed in standard glass-lined or stainless steel reactors, lowering capital expenditure barriers for suppliers. Furthermore, the ability to recycle excess ammonia gas and the absence of toxic waste gas generation drastically cut down on environmental treatment costs, which are becoming an increasingly significant portion of the total cost of ownership in the chemical sector. These factors combine to create a more resilient supply chain that is less susceptible to regulatory shutdowns and raw material volatility.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the drastic simplification of the purification workflow and the elimination of expensive catalysts. By avoiding the use of transition metal catalysts like Raney nickel, manufacturers remove the need for costly and time-consuming heavy metal removal steps, which often require specialized adsorbents or filtration media. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-remove by-products, thereby increasing the effective yield of the final crystalline product without the need for extensive recrystallization cycles. The recovery and reuse of ammonia gas further contribute to raw material efficiency, ensuring that input costs remain optimized even during periods of market fluctuation. This qualitative improvement in process efficiency leads to substantial cost savings that can be passed down the supply chain.
- Enhanced Supply Chain Reliability: From a logistics and planning perspective, the robustness of this synthesis method offers superior predictability compared to traditional routes. The reaction conditions are mild and easy to control, reducing the risk of batch failures due to thermal runaways or equipment malfunctions which are common in high-temperature urea processes. The use of commercially available and stable solvents like xylene ensures that raw material sourcing is straightforward and not subject to the geopolitical or supply constraints often associated with specialized reagents. Moreover, the shorter reaction times and simplified work-up procedures allow for faster turnaround times between batches, enabling suppliers to respond more agilely to sudden spikes in demand from downstream pharmaceutical or agrochemical clients.
- Scalability and Environmental Compliance: As global regulations on industrial emissions tighten, the environmental profile of a chemical process becomes a critical factor in vendor selection. This method generates no waste gas during the reaction phase (except when using ammonium bicarbonate, where CO2 is manageable), and the solvent system allows for the recycling of distilled water, aligning perfectly with green chemistry principles. The absence of viscous colloidal byproducts means that reactor fouling is minimized, allowing for longer campaign runs and easier cleaning between batches, which is essential for multi-purpose plants. This scalability ensures that the transition from pilot scale to commercial production of hundreds of tons is seamless, providing buyers with confidence in the long-term continuity of supply.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1,2,3,6-tetrahydrophthalimide using this advanced methodology. These insights are derived directly from the comparative data and technical specifications outlined in the patent documentation, providing a clear understanding of why this route is superior for high-specification applications. Understanding these nuances helps stakeholders make informed decisions about supplier qualification and process validation.
Q: How does this new method improve product purity compared to the urea method?
A: The novel fractional step method separates ring-opening and ring-closing processes, effectively avoiding side reactions like self-polymerization common in high-temperature urea methods, resulting in purity exceeding 99%.
Q: Is the ammonia source recyclable in this synthesis process?
A: Yes, when using ammonia gas as the source, excess ammonia can be recovered and recycled, significantly reducing environmental protection costs and raw material waste.
Q: What are the safety advantages over the hydrogenation method?
A: This method eliminates the need for high-pressure hydrogen and dangerous catalysts like Raney nickel, operating under safer atmospheric or mild vacuum conditions without explosive risks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,3,6-Tetrahydrophthalimide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into practical, commercial reality to truly benefit our partners. As a leading CDMO expert, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity and yield demonstrated in the lab are maintained at an industrial level. Our facilities are equipped with state-of-the-art rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 1,2,3,6-tetrahydrophthalimide meets the exacting standards required for pharmaceutical and fine chemical synthesis. We understand that consistency is key, and our robust quality management systems are designed to detect and mitigate any potential variance before it impacts your production line.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the specific economic benefits tailored to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate the superior quality and reliability of our manufacturing capabilities firsthand. Let us collaborate to drive efficiency and innovation in your chemical projects.
