Advanced Synthesis of Bioactive Ursolic Acid Derivatives for Commercial Pharmaceutical Manufacturing
Advanced Synthesis of Bioactive Ursolic Acid Derivatives for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks novel scaffolds with enhanced therapeutic indices, and natural product modification remains a cornerstone of drug discovery. Patent CN103626828A presents a significant advancement in the structural diversification of ursolic acid, a pentacyclic triterpenoid known for its broad pharmacological profile. This technology focuses on systematic chemical modifications at the C-3, C-2, and C-28 positions to generate a library of derivatives exhibiting potent antitumor activity against cell lines such as Hela, HepG2, and BGC-823. By transforming the native hydroxyl and carboxyl groups into oximes, esters, amides, and halogenated species, the patent establishes a robust platform for developing next-generation anticancer agents. For R&D teams and procurement specialists, understanding these synthetic pathways is crucial for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials for preclinical and clinical evaluation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to modifying triterpenoids often suffer from poor regioselectivity and harsh reaction conditions that degrade the sensitive steroid-like backbone. Conventional esterification or amidation at the C-28 position frequently requires high temperatures or strong coupling agents that can lead to epimerization or elimination side reactions, particularly given the steric hindrance around the E-ring. Furthermore, direct functionalization of the A-ring, specifically at the C-2 position adjacent to the C-3 hydroxyl, is notoriously difficult without protecting group strategies that add unnecessary steps and reduce overall atom economy. These inefficiencies result in low yields, complex purification profiles, and inconsistent batch-to-batch quality, posing significant risks for cost reduction in API manufacturing and delaying the timeline for biological screening campaigns.
The Novel Approach
The methodology outlined in the patent overcomes these hurdles through a modular strategy that prioritizes chemoselectivity and operational simplicity. By first oxidizing the C-3 hydroxyl to a ketone using Jones reagent, the synthesis creates a versatile handle for subsequent oxime formation or A-ring functionalization. The activation of the C-28 carboxyl group via oxalyl chloride allows for mild coupling with a wide array of nucleophiles, including benzyl alcohol, aniline, and amino alcohols, under controlled pH conditions. This approach not only preserves the integrity of the pentacyclic core but also enables the rapid generation of diverse libraries, such as compounds I through XIX, facilitating comprehensive structure-activity relationship (SAR) studies. 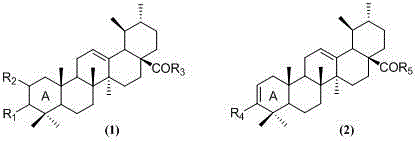
Mechanistic Insights into Oxalyl Chloride Activation and Vilsmeier Formylation
The core of this synthetic innovation lies in the precise activation of inert functional groups to enable downstream diversification. The conversion of the C-28 carboxylic acid to an acid chloride using oxalyl chloride proceeds via a nucleophilic acyl substitution mechanism, where the carbonyl oxygen attacks the electrophilic carbon of the reagent, displacing chloride and forming a reactive mixed anhydride intermediate that collapses to the acid chloride with the evolution of CO and CO2 gases. This activated species is highly susceptible to nucleophilic attack by amines or alcohols, forming stable amide or ester bonds respectively. In parallel, the introduction of functionality at the C-2 position utilizes the Vilsmeier-Haack reaction, where phosphorus oxychloride reacts with DMF to generate a chloroiminium ion. This electrophile attacks the enolizable alpha-position of the 3-keto ursolic acid derivative, ultimately yielding the 2-formyl compound (Compound VI) after hydrolysis, a transformation that is critical for introducing polar handles for further conjugation.
Impurity control is paramount in the production of high-purity pharmaceutical intermediates, and this process incorporates specific measures to mitigate side reactions. The use of pyridine as a solvent and base during oxime formation helps to scavenge generated HCl, preventing acid-catalyzed dehydration or rearrangement of the triterpene skeleton. Additionally, the purification protocols described, such as silica gel column chromatography using petroleum ether and ethyl acetate gradients, are optimized to separate closely related stereoisomers and unreacted starting materials. For instance, the synthesis of Compound VI involves a workup that includes washing with saturated brine and drying over anhydrous sodium sulfate to remove residual DMF and phosphorus byproducts, ensuring the final material meets stringent purity specifications required for biological testing and potential scale-up.
How to Synthesize Ursolic Acid Derivatives Efficiently
The synthesis of these bioactive modifiers follows a logical sequence starting from the isolation of ursolic acid from natural sources like loquat leaves, followed by oxidation and functional group interconversion. The process is designed to be adaptable, allowing chemists to swap out nucleophiles at the C-28 position or vary the reagents at the C-3 position to access different regions of chemical space. Detailed standard operating procedures for these transformations, including specific molar ratios, temperature controls, and workup techniques, are essential for reproducibility.
- Oxidize ursolic acid at the C-3 position using Jones reagent to form 3-oxoursolic acid, followed by oximation with hydroxylamine hydrochloride.
- Activate the C-28 carboxyl group using oxalyl chloride to facilitate amidation or esterification with various amines and alcohols.
- Perform A-ring modifications such as Vilsmeier formylation at C-2 or chlorination to generate diverse structural analogs like compounds VI and XV.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits regarding cost efficiency and supply security. The reliance on naturally abundant ursolic acid as a starting material ensures a stable feedstock supply, reducing the volatility associated with fully synthetic starting materials. Moreover, the use of common industrial reagents like oxalyl chloride, thionyl chloride, and standard solvents such as dichloromethane and ethanol simplifies the sourcing of raw materials, eliminating the need for exotic or highly regulated catalysts that often bottleneck production schedules.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive transition metal catalysts or precious metal complexes, which significantly lowers the raw material cost per kilogram. By utilizing straightforward activation strategies like acid chloride formation, the process avoids multi-step protection and deprotection sequences, thereby reducing solvent consumption and waste disposal costs. The ability to perform reactions at ambient or moderate temperatures (e.g., room temperature stirring for 20 hours) rather than requiring cryogenic conditions or extreme high-pressure equipment further contributes to substantial cost savings in utility and infrastructure expenditure.
- Enhanced Supply Chain Reliability: The robustness of the chemistry ensures high batch consistency, which is critical for maintaining long-term supply contracts with pharmaceutical partners. Since the starting material is derived from widespread botanical sources and the reagents are commodity chemicals, the risk of supply disruption due to geopolitical or logistical issues is minimized. This stability allows for better inventory planning and reduces the lead time for high-purity pharmaceutical intermediates, enabling faster progression from discovery to development phases without waiting for custom synthesis slots.
- Scalability and Environmental Compliance: The processes described, such as the reflux in DMAC at 168°C for A-ring modification, are amenable to scale-up in standard stainless steel reactors without requiring specialized lining or pressure vessels. The workup procedures involve standard aqueous washes and crystallization steps, which generate waste streams that are easier to treat compared to those containing heavy metals. This alignment with green chemistry principles facilitates regulatory compliance and simplifies the environmental impact assessment, making the commercial scale-up of complex pharmaceutical intermediates more feasible and sustainable.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis and application of these ursolic acid derivatives, based on the detailed experimental data provided in the patent documentation. Understanding these nuances helps R&D teams evaluate the feasibility of integrating these intermediates into their own drug discovery pipelines.
Q: What are the key structural modifications described in patent CN103626828A?
A: The patent details modifications at the C-3 position (oxidation to carbonyl or conversion to oximes), the C-2 position (introduction of hydroxymethylene groups), and the C-28 carboxyl group (formation of esters, amides, and hydrazides).
Q: Which reagents are critical for the C-28 activation step?
A: Oxalyl chloride is primarily used to activate the C-28 carboxyl group into an acid chloride intermediate, which then reacts with nucleophiles like amines or alcohols under mild conditions.
Q: How does the Vilsmeier reaction contribute to this synthesis?
A: The Vilsmeier-Haack reaction, utilizing phosphorus oxychloride and DMF, is employed to introduce a formyl group at the C-2 position of the 3-oxo derivative, expanding the chemical diversity of the A-ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ursolic Acid Derivatives Supplier
As a leading CDMO partner, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless. Our rigorous QC labs and stringent purity specifications guarantee that every batch of ursolic acid derivatives meets the exacting standards required for oncology research and development. We understand the critical nature of timeline adherence in drug discovery and are equipped to handle the complex purification challenges associated with triterpenoid chemistry, delivering materials that accelerate your project milestones.
We invite you to contact our technical procurement team to discuss your specific requirements for these antitumor intermediates. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can reduce your overall project costs. We are ready to provide specific COA data and route feasibility assessments to support your decision-making, ensuring a partnership built on transparency, quality, and scientific excellence.
