Advanced Copper Complex Catalysis for Efficient Diaryl Sulfone Manufacturing and Commercial Scale-Up
Advanced Copper Complex Catalysis for Efficient Diaryl Sulfone Manufacturing and Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways for synthesizing critical intermediates, particularly diaryl sulfone compounds which serve as pivotal scaffolds in drug discovery and crop protection agents. A groundbreaking technological advancement detailed in patent CN111689883B introduces a novel synthetic methodology that leverages a specialized monovalent copper complex containing a diphosphine o-carborane ligand to catalyze the self-coupling of aryl sulfonyl hydrazides. This innovation represents a significant paradigm shift from traditional high-energy processes, enabling the production of high-purity diaryl sulfones under remarkably mild conditions, specifically at room temperature and using ambient air as the sole oxidant. For R&D directors and procurement strategists, this development offers a compelling value proposition by combining exceptional reaction selectivity with operational simplicity, thereby addressing long-standing challenges related to substrate limitations and harsh reaction environments that have historically plagued sulfone synthesis.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of diaryl sulfone compounds has been dominated by the Friedel-Crafts reaction between aryl sulfonyl chlorides and aromatic hydrocarbons, a process fraught with significant technical and economic drawbacks that hinder modern sustainable manufacturing goals. This classical approach necessitates the use of strongly acidic conditions and extremely high reaction temperatures often exceeding 200°C, which not only imposes severe demands on reactor materials and safety protocols but also drastically limits the scope of applicable substrates to only electron-rich aromatic rings. Furthermore, the regioselectivity in these traditional methods is heavily influenced by the positioning effects of existing substituent groups, leading to complex mixtures of isomers that require costly and time-consuming purification steps to achieve the necessary purity standards for pharmaceutical applications. Alternative transition metal-catalyzed strategies, such as those employing palladium, have emerged but introduce their own set of challenges, including the prohibitive cost of noble metal catalysts and the frequent requirement for inert atmospheres and specialized ligands that complicate supply chain management and increase the overall cost of goods sold.
The Novel Approach
In stark contrast to these legacy technologies, the novel approach disclosed in the patent utilizes a uniquely designed copper complex that facilitates the direct self-coupling of aryl sulfonyl hydrazides with unprecedented efficiency and environmental compatibility. By employing a diphosphine o-carborane ligand system, the catalyst achieves remarkable stability in air, allowing the reaction to proceed smoothly at room temperature without the need for nitrogen protection or expensive oxidants. This method effectively bypasses the electronic limitations of Friedel-Crafts chemistry, demonstrating a wide substrate tolerance that includes various functional groups such as methyl, methoxy, nitro, and chloro substituents, all of which are converted to the corresponding diaryl sulfones with yields reaching as high as 96%. The operational simplicity of this new route, characterized by convenient product separation and the elimination of harsh acidic waste streams, positions it as a superior alternative for the reliable diaryl sulfone supplier seeking to optimize both technical performance and commercial viability.
Mechanistic Insights into Copper-o-Carborane Catalyzed Self-Coupling
The core of this technological breakthrough lies in the sophisticated design of the copper catalyst, where the o-carborane cage acts as a robust scaffold for the diphosphine ligands, creating a sterically and electronically optimized environment for the copper center. As illustrated in the general catalyst structure, the rigid three-dimensional architecture of the carborane cluster prevents ligand dissociation and catalyst deactivation, which are common failure modes in homogeneous copper catalysis. This structural integrity allows the monovalent copper species to effectively activate the aryl sulfonyl hydrazide substrate, facilitating the extrusion of nitrogen and the subsequent formation of the sulfur-carbon bond through a radical or organometallic pathway that is highly selective for the desired symmetric coupling product. The ability of this complex to utilize molecular oxygen from air as the terminal oxidant further underscores its mechanistic elegance, as it closes the catalytic cycle by regenerating the active copper species without generating stoichiometric amounts of toxic heavy metal waste or requiring external chemical oxidants.
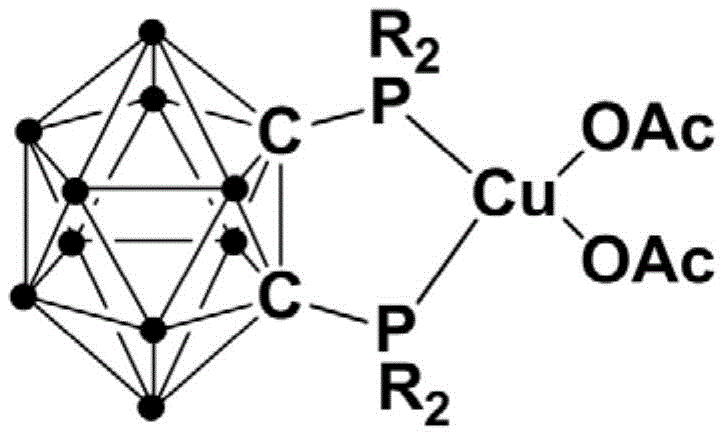
From an impurity control perspective, the high selectivity of this catalytic system is paramount for meeting the stringent purity specifications required in the production of API intermediates and fine chemicals. The specific coordination geometry imposed by the o-carborane ligand minimizes side reactions such as over-oxidation of the sulfone group or non-selective C-H activation on the aromatic ring, which are prevalent issues in less controlled oxidative coupling reactions. This precision in bond formation ensures that the resulting crude product contains minimal byproduct contamination, significantly reducing the burden on downstream purification units and enhancing the overall mass balance of the process. For quality assurance teams, this means a more consistent impurity profile across different batches, which is critical for regulatory compliance and maintaining the reliability of the supply chain for high-value pharmaceutical intermediates.
How to Synthesize Diaryl Sulfone Compounds Efficiently
The practical implementation of this synthesis involves a straightforward protocol that begins with the preparation of the specialized copper complex catalyst, followed by its application in the coupling reaction under ambient conditions. The catalyst itself is synthesized via a one-pot reaction involving the lithiation of ortho-carborane, followed by phosphorylation and complexation with copper acetate, a process that yields the active species in high purity and stability. Once the catalyst is secured, the coupling reaction is initiated by simply dissolving the aryl sulfonyl hydrazide and the catalyst in a solvent like toluene and stirring the mixture open to the air, a procedure that dramatically simplifies the operational requirements compared to glovebox techniques. Detailed standardized synthetic steps for the preparation of the catalyst and the subsequent coupling reaction are provided in the guide below to ensure reproducibility and optimal yield.
- Dissolve the specific copper complex catalyst (0.01-0.03 molar equivalent) and the aryl sulfonyl hydrazide substrate (1.0 molar equivalent) in an organic solvent such as toluene.
- Allow the reaction mixture to stir at room temperature under an air atmosphere, utilizing oxygen as the green oxidant for a duration of 2 to 5 hours.
- Upon completion, concentrate the reaction solution and purify the crude product via column chromatography using a petroleum ether and dichloromethane eluent system to isolate the high-purity diaryl sulfone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed technology translates into tangible strategic benefits that directly impact the bottom line and operational resilience of the manufacturing organization. The shift away from noble metal catalysts like palladium to abundant and inexpensive copper salts represents a fundamental reduction in raw material costs, while the elimination of high-temperature and high-pressure requirements leads to substantial energy savings and reduced wear on production equipment. Furthermore, the use of air as an oxidant removes the need for purchasing, storing, and handling hazardous chemical oxidants, thereby simplifying inventory management and reducing the regulatory burden associated with dangerous goods. These factors collectively contribute to a more robust and cost-efficient supply chain capable of responding rapidly to market demands for diaryl sulfone derivatives.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with a stable copper complex significantly lowers the direct material costs associated with catalytic loading, which is particularly impactful given the low catalyst loading ratios of 0.01 to 0.03 equivalents demonstrated in the patent data. Additionally, the ability to run reactions at room temperature eliminates the capital and operational expenditures related to heating systems and thermal insulation, while the simplified workup procedure reduces solvent consumption and labor hours required for purification. This holistic reduction in processing costs enhances the competitiveness of the final product in the global market for pharmaceutical and agrochemical intermediates.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as aryl sulfonyl hydrazides and common solvents like toluene ensures a stable and secure supply base that is less susceptible to the geopolitical and logistical volatility often seen with specialized reagents. The stability of the copper catalyst in air further mitigates supply chain risks by removing the need for specialized shipping and storage conditions, such as cold chains or inert gas blanketing, which can be prone to disruption. This ease of handling facilitates smoother logistics and inventory turnover, ensuring continuous production capabilities even during periods of market fluctuation.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of corrosive acids or toxic heavy metal waste streams make this process inherently safer and easier to scale from laboratory benchtop to multi-ton commercial production facilities. The environmental footprint is significantly reduced due to the use of air as a green oxidant and the generation of benign byproducts, aligning with increasingly stringent global environmental regulations and corporate sustainability goals. This compliance advantage not only avoids potential fines and remediation costs but also enhances the brand reputation of the manufacturer as a responsible and eco-friendly partner in the chemical value chain.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this technology into their existing portfolios, we have compiled a set of frequently asked questions based on the specific technical disclosures and experimental data found in the patent literature. These inquiries address critical aspects regarding catalyst stability, substrate scope, and process safety, providing a clear understanding of the operational parameters and potential limitations. The answers provided below are derived directly from the documented examples and comparative analysis within the intellectual property, ensuring accuracy and relevance for industrial application assessments.
Q: What are the primary advantages of this copper-catalyzed method over traditional Friedel-Crafts sulfonation?
A: Unlike conventional Friedel-Crafts reactions that require strongly acidic conditions and temperatures exceeding 200°C, this novel method operates at room temperature with air as the oxidant. It eliminates the need for harsh reagents, significantly reducing energy consumption and equipment corrosion risks while expanding the substrate scope to include electron-deficient aromatic rings.
Q: How does the o-carborane ligand contribute to the catalyst's performance?
A: The diphosphine o-carborane ligand provides exceptional stability to the monovalent copper center, allowing the complex to exist stably in air. This structural feature enables the catalyst to efficiently promote the self-coupling of aryl sulfonyl hydrazides with high selectivity and yield, avoiding the decomposition issues common in less stable transition metal complexes.
Q: Is this process suitable for large-scale industrial production of symmetric diaryl sulfones?
A: Yes, the process is highly amenable to scale-up due to its mild conditions and simple workup procedures. The use of inexpensive copper salts instead of noble metals like palladium, combined with the ability to use air as a free oxidant, drastically lowers the cost of goods sold (COGS) and simplifies supply chain logistics for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaryl Sulfone Supplier
As a premier CDMO and fine chemical manufacturer, NINGBO INNO PHARMCHEM is uniquely positioned to leverage this advanced copper-catalyzed technology to deliver high-quality diaryl sulfone compounds to the global market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of our pharmaceutical intermediates meets the highest international standards, providing our partners with the confidence and consistency required for successful drug development and commercialization.
We invite forward-thinking R&D and procurement leaders to collaborate with us to explore how this novel synthetic route can optimize your specific project requirements and drive down overall manufacturing costs. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume needs, along with specific COA data and route feasibility assessments that demonstrate the tangible benefits of partnering with a supplier at the forefront of chemical innovation.
