Advanced Copper-Catalyzed Synthesis of Diaryl Sulfones for Scalable Pharmaceutical Manufacturing
Advanced Copper-Catalyzed Synthesis of Diaryl Sulfones for Scalable Pharmaceutical Manufacturing
The pharmaceutical and agrochemical industries continuously seek robust, scalable, and cost-effective methodologies for constructing privileged scaffolds such as diaryl sulfones. These structural motifs are ubiquitous in bioactive molecules, exhibiting potent antifungal, antibacterial, and antitumor activities, alongside applications in inhibiting HIV-1 reverse transcriptase. A groundbreaking approach detailed in patent CN111689883B introduces a highly efficient synthetic pathway utilizing a novel monovalent copper complex containing a diphosphine o-carborane ligand. This technology represents a paradigm shift from traditional harsh synthetic routes, enabling the self-coupling of aryl sulfonyl hydrazides under remarkably mild conditions. By leveraging air as a green oxidant and operating at ambient temperatures, this innovation addresses critical pain points in process chemistry, offering a reliable pharmaceutical intermediates supplier with a distinct competitive edge in sustainability and operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the diaryl sulfone backbone has been fraught with significant chemical and engineering challenges that hinder efficient commercial production. Traditional Friedel-Crafts reactions, while foundational, necessitate strongly acidic environments and extreme temperatures often exceeding 200°C, which poses severe safety risks and limits the tolerance of sensitive functional groups. Furthermore, these classical methods are restricted to electron-rich aromatic rings, severely curtailing the diversity of accessible substrates due to positioning effects. More modern transition metal-catalyzed cross-coupling strategies, although versatile, frequently rely on expensive noble metals like palladium and require rigorous exclusion of oxygen and moisture. The reliance on sodium arylsulfinate or halogenated aromatics as coupling partners often involves multi-step precursor synthesis, inflating raw material costs and generating substantial waste streams. Additionally, existing copper-catalyzed variants often demand high catalyst loadings and elevated temperatures, negating the economic benefits of using base metals.
The Novel Approach
The methodology disclosed in the patent data revolutionizes this landscape by introducing a specialized copper complex that facilitates direct self-coupling of aryl sulfonyl hydrazides. Unlike prior art requiring inert atmospheres, this novel system thrives in the presence of air, utilizing molecular oxygen as the terminal oxidant to drive the reaction forward. The unique architecture of the catalyst, featuring a rigid o-carborane diphosphine ligand, imparts exceptional stability to the copper center, allowing the reaction to proceed smoothly at room temperature. This eliminates the energy-intensive heating steps associated with legacy processes and removes the capital expenditure associated with nitrogen blanketing systems. The result is a streamlined protocol that achieves high conversion rates with minimal catalyst loading, typically in the range of 1 to 3 mol%, thereby drastically simplifying the workup and purification stages for cost reduction in pharmaceutical intermediates manufacturing.

Mechanistic Insights into Copper-Catalyzed Self-Coupling
The efficacy of this transformation is rooted in the sophisticated design of the catalyst precursor, which features a copper center coordinated by a bidentate diphosphine ligand derived from o-carborane. The o-carborane cage provides a unique electronic and steric environment that stabilizes the copper(I) species against disproportionation and oxidation, ensuring a long catalytic lifetime. Mechanistically, the reaction likely proceeds through the activation of the sulfonyl hydrazide by the copper center, followed by the extrusion of nitrogen to generate a reactive sulfonyl radical or copper-sulfonyl intermediate. The presence of air facilitates the re-oxidation of the reduced copper species, closing the catalytic cycle without the need for stoichiometric chemical oxidants. This elegant redox neutrality minimizes the formation of inorganic salt byproducts, leading to a cleaner reaction profile and higher atom economy.
From an impurity control perspective, the mild reaction conditions play a pivotal role in maintaining product integrity. High-temperature processes often promote side reactions such as desulfonylation or ring degradation, which complicate downstream purification and reduce overall yield. By operating at room temperature, this copper-catalyzed system suppresses thermal decomposition pathways, ensuring that the resulting diaryl sulfone retains high chemical purity. The wide substrate scope demonstrated in the patent data, accommodating both electron-deficient nitro groups and electron-rich methoxy substituents, suggests a robust mechanistic pathway that is insensitive to electronic variations on the aromatic ring. This versatility is crucial for process chemists aiming to synthesize diverse libraries of high-purity pharmaceutical intermediates without needing to re-optimize conditions for every new analog.
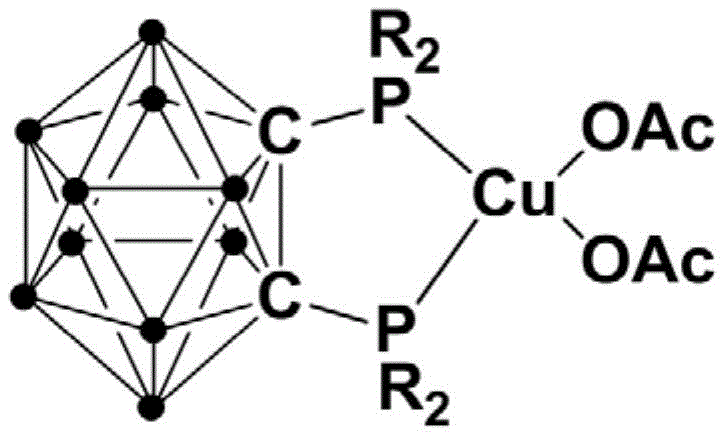
How to Synthesize Diaryl Sulfone Compounds Efficiently
The practical implementation of this technology involves a straightforward one-pot procedure that is amenable to standard laboratory and pilot plant equipment. The process begins with the preparation of the active copper complex, which can be synthesized via a sequential addition of n-butyllithium, o-carborane, and a chlorophosphine reagent, followed by complexation with copper acetate. Once the catalyst is prepared, the actual coupling reaction requires simply dissolving the aryl sulfonyl hydrazide and the catalyst in a common organic solvent such as toluene. The mixture is then stirred open to the atmosphere, allowing the reaction to reach completion within a few hours. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Dissolve the specific copper complex catalyst and aryl sulfonyl hydrazide substrate in an organic solvent such as toluene.
- Allow the reaction mixture to stir at room temperature using air as the sole oxidant for a duration of 2 to 5 hours.
- Concentrate the reaction solution and purify the crude product via column chromatography to isolate the target diaryl sulfone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed technology translates into tangible strategic benefits that extend beyond mere chemical yield. The shift from noble metal catalysts to a base metal copper system fundamentally alters the cost structure of diaryl sulfone production. By eliminating the dependency on volatile palladium markets and reducing the catalyst loading to trace levels, manufacturers can achieve substantial cost savings on raw materials. Furthermore, the ability to run reactions at room temperature significantly lowers utility consumption, reducing the carbon footprint and operational expenditures associated with heating and cooling large-scale reactors. The simplified workflow also means faster batch turnover times, enhancing overall plant throughput.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with a stable copper complex offers a direct avenue for lowering the Bill of Materials (BOM). Since the catalyst loading is low and the metal is abundant, the cost per kilogram of the final API intermediate is significantly optimized. Additionally, the use of air as an oxidant removes the need for purchasing and handling hazardous chemical oxidants, further reducing material costs and waste disposal fees associated with spent oxidant residues.
- Enhanced Supply Chain Reliability: Relying on copper, a globally abundant commodity, mitigates the supply risk associated with precious metals that are often subject to geopolitical instability and price spikes. The robustness of the catalyst, which remains stable in air, simplifies storage and logistics, removing the need for specialized inert atmosphere packaging. This stability ensures consistent quality and availability of the catalyst, preventing production delays caused by reagent degradation or supply shortages.
- Scalability and Environmental Compliance: The mild conditions and lack of toxic byproducts make this process inherently safer and easier to scale from gram to ton quantities. The absence of strong acids and high temperatures reduces the corrosion burden on reactor vessels and lowers the risk of thermal runaway incidents. From an environmental standpoint, the cleaner reaction profile minimizes the generation of hazardous waste, facilitating easier compliance with increasingly stringent environmental regulations and reducing the cost of effluent treatment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear picture of what potential partners can expect when integrating this technology into their manufacturing pipelines. Understanding these nuances is essential for making informed decisions about process adoption and vendor selection.
Q: What are the primary advantages of this copper complex catalyst over traditional palladium systems?
A: The copper complex described in patent CN111689883B eliminates the need for expensive noble metals like palladium. It operates efficiently at room temperature with air as an oxidant, significantly reducing energy costs and catalyst loading compared to conventional high-temperature or inert-gas methods.
Q: What is the substrate scope for this self-coupling reaction?
A: The method demonstrates a wide substrate range, successfully converting various aryl sulfonyl hydrazides including those with electron-donating groups (like methoxy and methyl) and electron-withdrawing groups (like nitro and chloro) into symmetric diaryl sulfones with high yields.
Q: Is nitrogen protection required for this synthesis?
A: No, nitrogen protection is not required. The reaction utilizes air as the oxidant, which simplifies the operational setup and enhances the safety profile for large-scale manufacturing by removing the need for inert gas purging systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaryl Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN111689883B for the production of high-value sulfone derivatives. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped to handle air-sensitive and exothermic reactions with precision, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for pharmaceutical applications.
We invite you to leverage our technical expertise to optimize your supply chain for diaryl sulfone intermediates. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our capabilities align with your strategic goals for cost-effective and sustainable manufacturing.
