Revolutionizing Beta-Alanine Production: Advanced Enzyme Engineering for Commercial Scale-Up
The global demand for high-purity beta-alanine continues to surge, driven by its critical role as a precursor in pharmaceutical synthesis, particularly for vitamins and cardiovascular drugs, as well as its expanding use in sports nutrition. However, traditional biocatalytic routes often face bottlenecks related to enzyme efficiency and stability. Patent CN113444712B introduces a groundbreaking solution by engineering specific mutants of L-aspartate-alpha-decarboxylase (PanD). This technology addresses the fundamental limitation of wild-type PanD enzymes, which often exist in an inactive precursor state, by utilizing directed evolution to enhance self-cleavage efficiency. For procurement leaders and R&D directors seeking a reliable beta-alanine supplier, this innovation represents a pivotal shift towards more economical and robust manufacturing processes that can be seamlessly integrated into existing fermentation infrastructure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the biosynthesis of beta-alanine using wild-type L-aspartate-alpha-decarboxylase has been hampered by the enzyme's complex activation mechanism. In many prokaryotic systems, such as Escherichia coli, the PanD enzyme is initially expressed as an inactive zymogen known as the pi-protein. This precursor requires a specific self-cleavage event, often dependent on auxiliary activators like PanZ or specific cellular conditions, to split into the active alpha and beta subunits. Without efficient cleavage, a significant portion of the expressed protein remains catalytically inert, leading to suboptimal substrate conversion rates and lower final titers of beta-alanine. This inefficiency translates directly into higher production costs, as larger fermentation volumes and longer processing times are required to achieve target yields, creating a substantial burden on supply chain logistics and manufacturing overheads for fine chemical producers.
The Novel Approach
The technology disclosed in CN113444712B overcomes these inherent biological constraints through precise protein engineering. By identifying and mutating specific amino acid residues at positions 14, 44, and 85 of the EcoPanD sequence, the inventors have created variants (EcoPanD-K14T, EcoPanD-I44V, and EcoPanD-V85L) that exhibit drastically improved self-cleavage capabilities. Unlike the wild-type enzyme which struggles to activate, these mutants spontaneously and efficiently convert from the inactive pi-form to the active heterodimeric structure without heavy reliance on external activators. This structural optimization ensures that a much higher percentage of the expressed protein is functionally active during fermentation. Consequently, this novel approach not only simplifies the metabolic burden on the host strain but also establishes a foundation for cost reduction in pharmaceutical intermediate manufacturing by maximizing the catalytic output per unit of biomass.
Mechanistic Insights into Site-Directed Mutagenesis of PanD
The core of this technological advancement lies in the molecular understanding of the PanD activation pathway. The wild-type enzyme undergoes an N-to-O acyl transfer reaction at the Gly24-Ser25 bond to initiate self-cleavage. However, evolutionary analysis revealed that certain sites in the Class II PanD family were under positive selection pressure, suggesting they play a critical role in functional differentiation. The patent identifies residues Lys14, Ile44, and Val85 as key determinants of this activity. By substituting these residues with Threonine, Valine, and Leucine respectively, the local steric environment and electrostatic interactions around the cleavage site are optimized. This modification lowers the energy barrier for the intramolecular nucleophilic attack required for cleavage, effectively 'unlocking' the enzyme's potential. The result is a catalytic machine that spends less time in the inactive precursor state and more time converting L-aspartic acid into beta-alanine and carbon dioxide.
Furthermore, the stability of the resulting active subunits is crucial for sustained fermentation performance. The mutations do not merely accelerate cleavage; they also appear to stabilize the resulting alpha and beta chains, preventing rapid degradation or aggregation within the cytoplasm of the host E. coli W3110 cells. This is evidenced by SDS-PAGE analysis which shows a marked decrease in the proportion of the pi-band and a corresponding increase in the intensity of the alpha and beta bands in the mutant strains compared to the wild type.  . This shift in protein profile confirms that the genetic modifications successfully alter the post-translational processing of the enzyme. For R&D teams, this implies a more predictable and controllable biocatalytic system where enzyme activity correlates directly with expression levels, reducing the variability often seen in wild-type fermentations.
. This shift in protein profile confirms that the genetic modifications successfully alter the post-translational processing of the enzyme. For R&D teams, this implies a more predictable and controllable biocatalytic system where enzyme activity correlates directly with expression levels, reducing the variability often seen in wild-type fermentations.
How to Synthesize Beta-Alanine Efficiently
Implementing this patented technology involves a straightforward genetic engineering workflow compatible with standard molecular biology protocols. The process begins with the construction of recombinant plasmids where the mutated panD genes are cloned into the pTrc99A vector, followed by transformation into E. coli W3110. The detailed standardized synthesis steps see the guide below.
- Construct recombinant plasmids by inserting the mutated EcopanD gene (K14T, I44V, or V85L) into the pTrc99A vector using whole-plasmid PCR and one-step cloning.
- Transform the engineered plasmids into E. coli W3110 competent cells and select positive clones on kanamycin-resistant LB agar plates.
- Conduct high-density fermentation with IPTG induction at optimized temperatures (28-30°C) to maximize the conversion of L-aspartic acid to beta-alanine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to these engineered PanD mutants offers tangible strategic benefits beyond mere technical novelty. The primary advantage is the significant enhancement in volumetric productivity. Data from the patent indicates that strains expressing the I44V mutant achieved beta-alanine yields of 0.360 g/L, a substantial improvement over the 0.215 g/L observed with the wild-type strain under identical conditions. 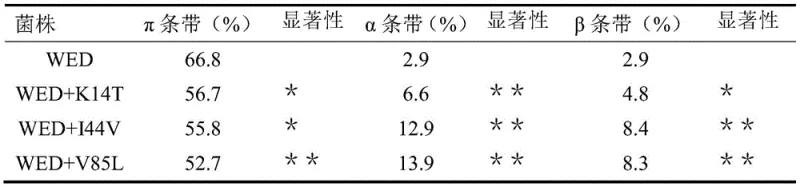 . This increase in titer means that manufacturers can produce the same amount of product using smaller fermentation tanks or shorter batch cycles, directly impacting capital expenditure and operational efficiency. Such improvements are critical for maintaining competitiveness in the volatile market of high-purity pharmaceutical intermediates.
. This increase in titer means that manufacturers can produce the same amount of product using smaller fermentation tanks or shorter batch cycles, directly impacting capital expenditure and operational efficiency. Such improvements are critical for maintaining competitiveness in the volatile market of high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The enhanced catalytic efficiency of the mutant enzymes leads to a more complete conversion of the L-aspartic acid substrate. In traditional processes, incomplete conversion often necessitates expensive downstream purification steps to separate unreacted starting materials from the product. By driving the reaction closer to completion, this technology minimizes waste and reduces the load on purification columns and crystallization units. Furthermore, the reduced reliance on auxiliary activators like PanZ simplifies the medium formulation, potentially lowering raw material costs. The elimination of bottlenecks associated with enzyme activation allows for higher cell density fermentations without the risk of accumulating inactive protein aggregates, ensuring that every gram of biomass contributes effectively to production.
- Enhanced Supply Chain Reliability: Consistency is paramount in the supply of fine chemicals. Wild-type enzymatic processes can suffer from batch-to-batch variability due to fluctuations in the expression of activation factors. The self-activating nature of the K14T, I44V, and V85L mutants provides a more robust and stable production platform. This reliability translates to more predictable lead times for customers, as the risk of failed batches or sub-par yields is significantly mitigated. For global buyers, this means a secure source of beta-alanine that can withstand the pressures of scaling up from pilot studies to multi-ton annual commercial production without compromising on quality or delivery schedules.
- Scalability and Environmental Compliance: The process utilizes E. coli W3110, a well-characterized and safe host strain widely accepted in industrial biotechnology. The fermentation media described in the patent consists of common, inexpensive components such as ammonium sulfate, glucose, and yeast extract, avoiding the need for exotic or hazardous reagents. This simplicity facilitates easy scale-up from laboratory flasks to industrial bioreactors. Additionally, the higher conversion efficiency reduces the environmental footprint by minimizing the discharge of unreacted organic acids in the effluent. This aligns with increasingly stringent environmental regulations, making the process not only economically viable but also sustainable for long-term operation in regulated jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzyme engineering technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity for stakeholders evaluating this route for their own production needs.
Q: How does the K14T/I44V/V85L mutation improve PanD enzyme activity?
A: These specific mutations target the self-cleavage site of the PanD precursor protein. By altering amino acids at positions 14, 44, and 85, the mutant enzymes exhibit significantly enhanced spontaneous cleavage from the inactive pi-protein form into the active alpha and beta subunits, thereby increasing overall catalytic turnover.
Q: What are the primary industrial applications of the beta-alanine produced via this method?
A: The high-purity beta-alanine generated serves as a critical precursor for synthesizing pantothenic acid (Vitamin B5), carnosine, and various pharmaceutical intermediates like balsalazide. It is also widely used in the food industry as a flavor enhancer and nutritional supplement.
Q: Is this enzymatic process scalable for commercial manufacturing?
A: Yes, the patent demonstrates successful fermentation in E. coli W3110 hosts using standard industrial vectors like pTrc99A. The significant increase in yield per liter of fermentation broth indicates strong potential for cost-effective scale-up from pilot to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Alanine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzyme engineering strategies outlined in CN113444712B. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the precise fermentation controls required for these high-efficiency mutant strains, ensuring stringent purity specifications are met for every batch. With our rigorous QC labs and deep expertise in biocatalysis, we are uniquely positioned to help you leverage this technology to secure a competitive edge in the beta-alanine market.
We invite you to collaborate with us to explore the full commercial viability of this process for your specific applications. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can optimize your supply chain and reduce your overall cost of goods sold.
