Advanced Catalytic Hydrogenation Technology for High-Purity Aminoanisole Production
The chemical industry is currently witnessing a significant paradigm shift towards greener and more efficient synthesis routes, particularly in the production of critical intermediates like aminoanisole. Patent CN100475772C introduces a groundbreaking process for preparing amino phenylmether, commonly known as anisidine, through the catalytic hydrogenation of a nitroanisole mixture. This technology represents a substantial departure from traditional reduction methods, addressing long-standing issues related to environmental pollution and production costs. By utilizing methanol as a solvent and employing robust catalysts such as Raney-Ni or Pd-C, the process achieves a continuous or intermittent catalytic hydrogenating reduction under controlled conditions. The innovation lies not just in the reaction itself but in the integrated system of catalyst recovery, decolorization, and precise separation, which collectively ensure a high-yield, high-purity output suitable for demanding downstream applications in the pharmaceutical and agrochemical sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 2-anisidine and 4-anisidine has relied heavily on reduction techniques using sodium sulfite or iron powder. These conventional methodologies are fraught with significant operational and environmental drawbacks that hinder modern manufacturing efficiency. The most pressing issue is the massive generation of waste; iron powder reduction, for instance, produces substantial quantities of iron sludge and wastewater, creating a severe burden on waste treatment facilities and posing serious risks of secondary pollution. Furthermore, these older processes often suffer from high raw material consumption and corrosive conditions that degrade equipment longevity, leading to increased maintenance costs and potential safety hazards. The resulting product quality from these methods is frequently inconsistent, often requiring extensive and costly post-processing to meet the rigorous purity standards demanded by high-end fine chemical applications, thereby inflating the overall cost of goods sold.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages catalytic hydrogenation to overcome these inherent limitations, offering a clean and highly efficient alternative. By reacting a mixture of 2-Nitroanisole and 4-Nitroanisole with hydrogen in the presence of a catalyst within a methanol solvent system, the process achieves a transformation that is both atom-economical and environmentally benign. The use of methanol serves a dual purpose: it acts as an effective solvent that reduces the viscosity of the reaction system, thereby improving the dispersion state of the catalyst and enhancing heat transfer processes. This improved thermal management allows for precise control over reaction temperatures between 130°C and 160°C and pressures ranging from 4.8MPa to 6.0MPa, ensuring optimal conversion rates. The result is a streamlined production flow that minimizes waste discharge, eliminates the need for corrosive reagents, and delivers a superior quality product with significantly reduced downstream processing requirements.
Mechanistic Insights into Catalytic Hydrogenation of Nitroanisole
The core of this technological advancement lies in the mechanistic efficiency of the catalytic hydrogenation reaction, where the nitro group is selectively reduced to an amino group without compromising the integrity of the methoxy substituent. The reaction proceeds on the surface of the heterogeneous catalyst, whether it be Raney-Ni or Pd-C, where hydrogen molecules are activated and transferred to the nitroanisole substrate. The choice of a three-phase fixed bed, fluidized bed, or stirred-tank reactor facilitates intimate contact between the gaseous hydrogen, liquid substrate, and solid catalyst, maximizing the reaction kinetics. This setup ensures that the per pass conversion can reach more than 85%, a testament to the high activity of the catalytic system. Moreover, the specific operating parameters are tuned to prevent over-reduction or side reactions, which is critical for maintaining the structural fidelity of the anisidine molecule required for subsequent synthetic steps in drug or pesticide manufacturing.
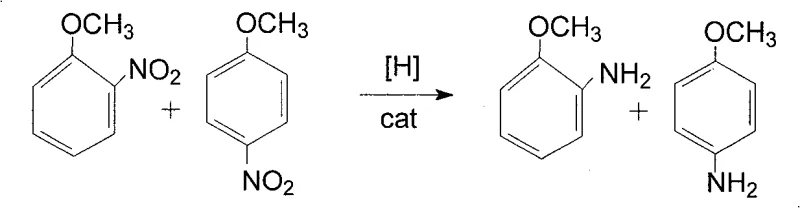
Beyond the primary reduction mechanism, the process incorporates sophisticated impurity control strategies that are vital for achieving pharmaceutical-grade purity. Following the hydrogenation step, the reaction mass undergoes a rigorous decolorization and impurity removal operation using adsorbents such as activated carbon, diatomite, or molecular sieves. This step is crucial for removing trace colored impurities and by-products that could otherwise compromise the quality of the final crystal. The subsequent separation and refining stage utilizes fractional crystallization, exploiting the solubility differences of 2-anisidine and 4-anisidine in methanol at varying temperatures. By carefully controlling the cooling profile—first to 10°C to precipitate 4-anisidine and then to 0°C for 2-anisidine—the process effectively isolates each isomer with a purity exceeding 98%. This level of control over the impurity profile is essential for R&D directors who require consistent and reliable intermediates for complex synthesis pathways.
How to Synthesize Aminoanisole Efficiently
The synthesis of high-purity aminoanisole via this catalytic route involves a sequence of precisely controlled unit operations designed to maximize yield and minimize waste. The process begins with the preparation of the reaction mixture, where the nitroanisole feedstock is dissolved in methanol and introduced into the reactor along with the catalyst. Hydrogen is then fed into the system under elevated pressure and temperature to drive the reduction to completion. Once the reaction is finished, indicated by the cessation of hydrogen consumption, the mixture is cooled and depressurized to facilitate the separation of the solid catalyst. The detailed standardized synthesis steps, including specific ratios, temperature ramps, and filtration protocols, are outlined in the comprehensive guide below to ensure reproducibility and safety in a commercial setting.
- Conduct catalytic hydrogenation reduction of nitroanisole mixture in methanol solvent using Raney-Ni or Pd-C catalyst at 130-160°C and 4.8-6.0 MPa pressure.
- Perform solid-liquid separation to recover the catalyst for recycling, followed by adsorption bleaching to remove impurities.
- Separate and refine the liquid phase through crystallization to obtain high-purity 2-anisidine and 4-anisidine products.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic hydrogenation technology translates into tangible strategic advantages that extend far beyond simple chemical conversion. The shift away from iron powder and sodium sulfite reduction fundamentally alters the cost structure of anisidine manufacturing by eliminating the heavy logistical and financial burdens associated with hazardous waste disposal. The ability to recycle the catalyst with an organic efficiency reaching 99.5% means that the consumption of expensive noble metals or nickel is drastically reduced, leading to significant long-term cost savings. Furthermore, the use of methanol as a solvent allows for efficient recovery and reuse through distillation, further lowering the variable costs of production. These factors combine to create a more resilient and cost-effective supply chain that is less vulnerable to fluctuations in raw material prices and regulatory changes regarding environmental compliance.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of costly waste treatment procedures and the high efficiency of raw material utilization. Unlike traditional methods that generate tons of iron sludge requiring expensive disposal, this catalytic route produces minimal waste, thereby reducing the operational expenditure related to environmental management. Additionally, the recyclability of the catalyst and the solvent recovery system ensure that the input costs per kilogram of product are significantly lower than those of conventional batch processes. This efficiency allows manufacturers to offer more competitive pricing without sacrificing margin, providing a distinct advantage in price-sensitive markets.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this continuous or semi-continuous process ensures a steady and reliable flow of high-quality intermediates. The equipment used, such as three-phase fixed bed reactors, is designed for durability and consistent performance, minimizing unplanned downtime and production delays. The availability of raw materials like methanol and hydrogen is generally stable on a global scale, reducing the risk of supply disruptions that can plague more specialized reagent-dependent processes. This reliability is critical for downstream customers who depend on just-in-time delivery schedules to maintain their own production lines, making this technology a preferred choice for long-term supply agreements.
- Scalability and Environmental Compliance: The process is inherently scalable, capable of transitioning from pilot-scale batches to large-scale industrial production with minimal modification to the core chemistry. The use of standard industrial reactors and filtration systems means that capacity can be expanded to meet growing market demand without the need for exotic or hard-to-source equipment. Moreover, the clean nature of the process ensures strict adherence to increasingly stringent environmental regulations, mitigating the risk of fines or shutdowns due to non-compliance. This alignment with green chemistry principles not only protects the manufacturer's license to operate but also enhances the brand value for customers seeking sustainable supply chain partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of aminoanisole using this advanced catalytic hydrogenation method. These answers are derived directly from the patent data and practical implementation experiences, providing clarity on the process capabilities and limitations. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios or sourcing strategies.
Q: What are the primary advantages of catalytic hydrogenation over iron powder reduction for anisidine?
A: Catalytic hydrogenation eliminates the generation of large amounts of iron sludge and wastewater associated with traditional iron powder reduction, significantly reducing environmental pollution and waste treatment costs while improving product purity.
Q: Can the catalyst be reused in this process?
A: Yes, the process utilizes efficient filtration devices to recover the Raney-Ni or Pd-C catalyst, allowing it to be recycled with an organic efficiency reaching up to 99.5%, which drastically lowers raw material consumption.
Q: What purity levels can be achieved with this synthesis method?
A: Through optimized crystallization and purification steps, the process consistently yields 2-anisidine and 4-anisidine with purity levels exceeding 98%, meeting stringent requirements for pharmaceutical and agrochemical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aminoanisole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final products, which is why we have invested heavily in mastering advanced synthesis technologies like the one described in CN100475772C. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We operate under stringent purity specifications and utilize rigorous QC labs to verify that every batch of aminoaniside meets the highest international standards, giving you confidence in the reliability of your supply chain. Our commitment to excellence extends beyond mere production; we act as a strategic partner dedicated to optimizing your chemical sourcing strategy.
We invite you to engage with our technical procurement team to discuss how our capabilities can align with your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how switching to our catalytically produced anisidine can reduce your overall manufacturing costs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that will demonstrate the tangible benefits of partnering with a leader in fine chemical innovation. Let us help you secure a sustainable and efficient supply of critical intermediates for your future growth.
