Advanced Biocatalytic Synthesis of Chiral Amino Alcohols for Commercial Scale-Up
The recent disclosure of patent CN112852894A introduces a groundbreaking advancement in the biocatalytic synthesis of chiral amino alcohol compounds, specifically targeting high-value pharmaceutical intermediates. This innovation centers on a novel amine dehydrogenase mutant derived from Sporosarcina psychrophila, engineered through precise site-directed mutagenesis at multiple critical positions to enhance catalytic efficiency. By leveraging this engineered biocatalyst, manufacturers can achieve stereoselectivity exceeding 99 percent ee, a metric that is paramount for the production of active pharmaceutical ingredients requiring strict chiral purity. The technology addresses long-standing challenges in the industry by offering a sustainable alternative to traditional chemical synthesis, which often relies on hazardous reagents and extreme reaction conditions. Consequently, this patent represents a significant leap forward for organizations seeking to optimize their supply chains for complex chiral building blocks while adhering to increasingly stringent environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional chemical methodologies for synthesizing chiral amino alcohols, such as (S)-2-amino-1-butanol, frequently necessitate the use of expensive transition metal catalysts like nickel or lithium aluminum hydride under high-pressure hydrogenation conditions. These processes are inherently fraught with safety risks due to the handling of pyrophoric reagents and the generation of significant hazardous waste streams that require costly disposal protocols. Furthermore, achieving high enantiomeric excess often demands complex resolution steps involving chiral resolving agents, which drastically reduces the overall atom economy and increases the final cost of goods. The reliance on harsh reaction environments also limits the compatibility with sensitive functional groups, thereby restricting the scope of substrates that can be effectively processed without degradation. Ultimately, these traditional pathways impose a substantial burden on both operational safety and environmental compliance, driving the urgent need for greener alternatives.
The Novel Approach
In stark contrast, the novel biocatalytic approach utilizing the SpAmDH mutant operates under mild aqueous conditions, typically at temperatures ranging from 20 to 35 degrees Celsius and atmospheric pressure. This enzymatic pathway eliminates the requirement for precious metal catalysts and toxic reducing agents, thereby simplifying the downstream purification process and significantly reducing the environmental footprint of the manufacturing operation. The mutant enzyme demonstrates exceptional substrate tolerance and catalytic activity, enabling the direct asymmetric amination of hydroxy ketones with conversion rates reaching up to 99 percent in optimized systems. By integrating this biocatalytic step, producers can streamline their synthesis routes, avoiding the multi-step protection and deprotection sequences often associated with chemical methods. This shift not only enhances process safety but also aligns with global sustainability goals, making it an attractive option for large-scale industrial applications.
Mechanistic Insights into SpAmDH-Catalyzed Asymmetric Reduction
The core of this technological breakthrough lies in the rational design of the SpAmDH protein, where specific amino acid residues at positions such as 40, 61, 111, and 294 have been mutated to optimize the active site geometry for substrate binding. These strategic modifications enhance the enzyme's affinity for hydroxy ketone substrates while simultaneously improving the stereochemical control during the hydride transfer from the NADH cofactor. The catalytic cycle involves the precise orientation of the substrate within the enzyme pocket, ensuring that the reduction occurs exclusively on one face of the carbonyl group to yield the desired chiral amine. This level of molecular precision is critical for minimizing the formation of unwanted enantiomers, which are often difficult and expensive to separate in later stages of drug manufacturing. The engineering of these specific sites allows for a tailored fit that accommodates the steric bulk of various substrates, thereby broadening the applicability of the enzyme beyond simple model compounds.
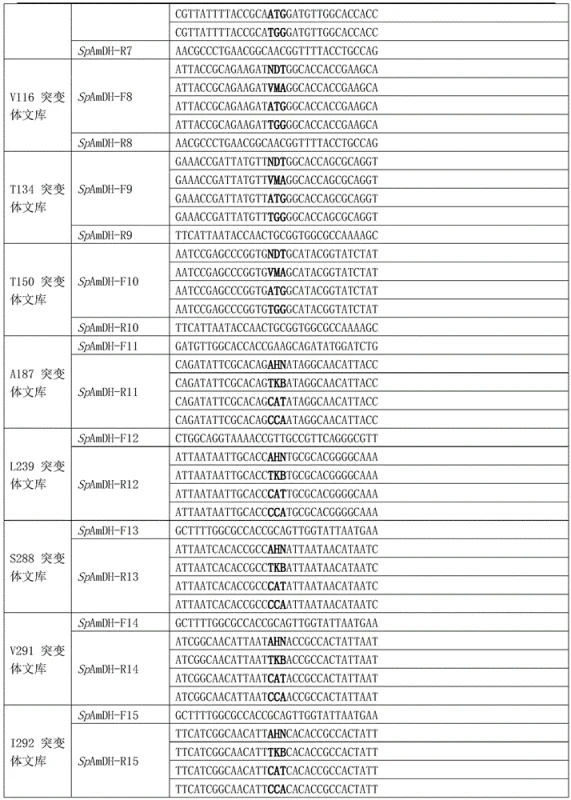
Regarding impurity control, the high stereoselectivity of the SpAmDH mutant inherently suppresses the generation of the opposite enantiomer, resulting in a product profile with greater than 99 percent enantiomeric excess. The use of whole-cell biocatalysts or lysates further simplifies the reaction matrix, as the cellular environment helps stabilize the enzyme and cofactor regeneration systems without the need for external additives. This robustness ensures consistent product quality across different batches, reducing the variability that often plagues chemical synthesis campaigns. Moreover, the absence of metal residues eliminates the need for rigorous metal scavenging steps, which are mandatory for pharmaceutical intermediates intended for human consumption. The streamlined purification process not only saves time but also reduces the consumption of organic solvents, contributing to a more sustainable and cost-effective manufacturing workflow for high-value chiral intermediates.
How to Synthesize Chiral Amino Alcohol Efficiently
The implementation of this biocatalytic route requires a systematic approach to strain engineering and fermentation optimization to ensure maximum enzyme expression and activity. Detailed protocols involve the construction of recombinant vectors, transformation into host cells like E. coli BL21(DE3), and subsequent induction under controlled conditions to produce the active biocatalyst. The standardized synthesis steps outlined below provide a comprehensive guide for replicating the high-yield production of chiral amino alcohols using this patented technology. Adhering to these parameters is essential for achieving the reported conversion rates and stereoselectivity metrics in a commercial setting. Careful attention to pH control and temperature regulation during the fermentation phase is critical to maintaining enzyme stability and preventing the formation of inactive aggregates.
- Construct recombinant vectors with SpAmDH mutant genes and transform into E. coli BL21(DE3) host cells.
- Induce enzyme expression under controlled fermentation conditions and prepare whole cells or lysates.
- Catalyze the reduction of hydroxy ketone substrates in ammonium chloride buffer with cofactor regeneration.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this SpAmDH mutant technology offers profound benefits for procurement and supply chain teams looking to mitigate risks associated with traditional chemical manufacturing. By shifting to a biocatalytic process, companies can decouple their production from the volatile pricing of precious metals and hazardous reagents, leading to more predictable cost structures. The simplified workflow reduces the number of unit operations required, which directly translates to lower capital expenditure and operational overheads for facility management. Furthermore, the enhanced safety profile minimizes the potential for workplace accidents and regulatory fines, safeguarding the company's reputation and financial stability.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in solvent usage significantly lower the raw material costs associated with production. This qualitative shift in the cost structure allows for substantial savings over the lifecycle of the product, especially when scaled to industrial volumes. The removal of metal scavenging steps further reduces the consumption of specialized resins and filtration media, contributing to overall process efficiency.
- Enhanced Supply Chain Reliability: The reliance on fermentation-based production ensures a consistent and scalable supply of the biocatalyst, independent of geopolitical tensions affecting metal mining. This stability is crucial for maintaining continuous manufacturing operations and meeting tight delivery schedules for downstream pharmaceutical clients. The robustness of the engineered strain also reduces the risk of batch failures, ensuring a steady flow of high-quality intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions facilitate easier scale-up from laboratory to commercial production without the need for specialized high-pressure equipment. This accessibility accelerates the time to market for new drug candidates while ensuring compliance with increasingly strict environmental regulations regarding waste disposal. The green chemistry credentials of this process also enhance the brand value of the final pharmaceutical product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this amine dehydrogenase technology in industrial settings. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and feasibility of adopting this biocatalytic route. Understanding these details is vital for decision-makers evaluating the potential integration of this technology into their existing manufacturing portfolios. Addressing these concerns proactively helps to mitigate risks associated with technology transfer and ensures a smoother adoption curve for production teams.
Q: What are the advantages of using SpAmDH mutants over chemical synthesis?
A: SpAmDH mutants offer higher stereoselectivity (>99% ee), milder reaction conditions, and eliminate the need for hazardous metal catalysts.
Q: Can this process be scaled for industrial production?
A: Yes, the fermentation-based production of the biocatalyst is highly scalable and suitable for commercial manufacturing volumes.
Q: What substrates are compatible with this enzyme technology?
A: The enzyme accepts various hydroxy ketones including 1-hydroxy-2-butanone and 4-hydroxy-2-butanone for chiral amino alcohol synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amino Alcohol Supplier
Partnering with NINGBO INNO PHARMCHEM provides access to extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to market. Our team possesses stringent purity specifications and rigorous QC labs to guarantee that every batch of chiral amino alcohol meets the highest industry standards for pharmaceutical applications. We are committed to delivering reliable pharmaceutical intermediate supplier services that align with your strategic goals for cost efficiency and supply security. Our infrastructure is designed to handle complex biocatalytic processes with the same level of precision and reliability as traditional chemical synthesis.
Request a Customized Cost-Saving Analysis from our technical procurement team to explore how this technology can optimize your specific supply chain. We invite you to contact us for specific COA data and route feasibility assessments tailored to your unique production requirements. Our experts are ready to assist you in navigating the complexities of biocatalytic manufacturing to achieve your business objectives. Engaging with us early in your development cycle allows for better planning and resource allocation.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
