Advanced Synthesis of Water-Soluble Aggregation-Induced Emission Quinoxaline Compounds for High-Performance Optoelectronics
Advanced Synthesis of Water-Soluble Aggregation-Induced Emission Quinoxaline Compounds for High-Performance Optoelectronics
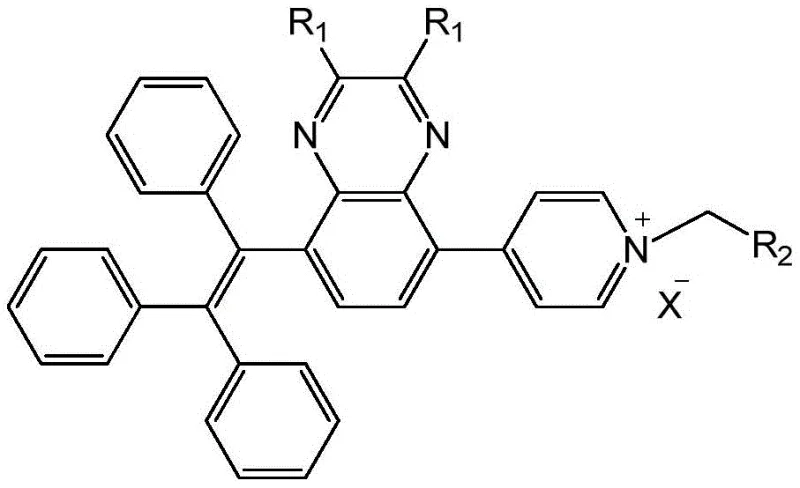
The landscape of functional organic materials is undergoing a significant transformation, driven by the demand for high-efficiency luminescent compounds that can operate in diverse environments. Patent CN108530423B introduces a groundbreaking methodology for synthesizing water-soluble aggregation-induced emission (AIE) quinoxaline compounds, addressing a critical bottleneck in the field of fluorescent probes and solid-state lighting. Traditionally, AIE materials suffer from poor solubility in aqueous media, limiting their utility in biological imaging and specific optoelectronic applications where polar processing is required. This patent discloses a sophisticated multi-step synthetic route that successfully integrates a hydrophobic AIE-active triphenylethylene unit with a hydrophilic pyridinium salt functionality onto a quinoxaline core. By leveraging sequential Suzuki-Miyaura cross-coupling reactions followed by a quaternization step, the inventors have created a versatile platform for generating materials that exhibit strong fluorescence in the solid state while maintaining excellent solubility in water-organic mixed solvents. This technological leap represents a substantial advancement for manufacturers seeking reliable electronic chemical suppliers capable of delivering next-generation luminescent intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of quinoxaline-based fluorophores has been hindered by the inherent trade-off between luminescence efficiency and solubility. Conventional synthesis routes often focus on extending conjugation to red-shift emission, but this frequently leads to planar structures prone to Aggregation-Caused Quenching (ACQ), where fluorescence is killed upon aggregation due to pi-pi stacking interactions. Furthermore, existing AIE molecules, while solving the quenching issue, are predominantly hydrophobic, restricting their application to organic phases. The inability to dissolve these materials in high percentages of water prevents their use in physiological conditions or green chemistry manufacturing processes. Traditional methods also struggle with the precise installation of functional groups at the 2,3,5,8-positions of the quinoxaline ring without causing side reactions or requiring harsh conditions that degrade the sensitive fluorophore. This lack of modularity and solubility has created a significant gap in the market for high-purity OLED material precursors that can be processed in environmentally friendly solvent systems.
The Novel Approach
The methodology outlined in CN108530423B offers a transformative solution by decoupling the AIE mechanism from solubility constraints through a modular design. The core innovation lies in the strategic placement of a triphenylethylene group at the 5-position and a pyridine moiety at the 8-position of the quinoxaline scaffold. This architecture ensures that the molecule retains its propeller-shaped conformation necessary for AIE, while the subsequent conversion of the pyridine nitrogen into a pyridinium cation imparts ionic character and water solubility. The synthesis utilizes a stepwise Suzuki coupling strategy, allowing for the independent optimization of each functional group attachment. This approach not only simplifies the purification process—often allowing for isolation via precipitation rather than tedious chromatography—but also enables the fine-tuning of photophysical properties by varying the substituents on the quinoxaline ring. For procurement teams, this translates to cost reduction in electronic chemical manufacturing, as the robust nature of the Suzuki reaction ensures high reproducibility and yield consistency across batches.
Mechanistic Insights into Pd-Catalyzed Suzuki Coupling and Quaternization
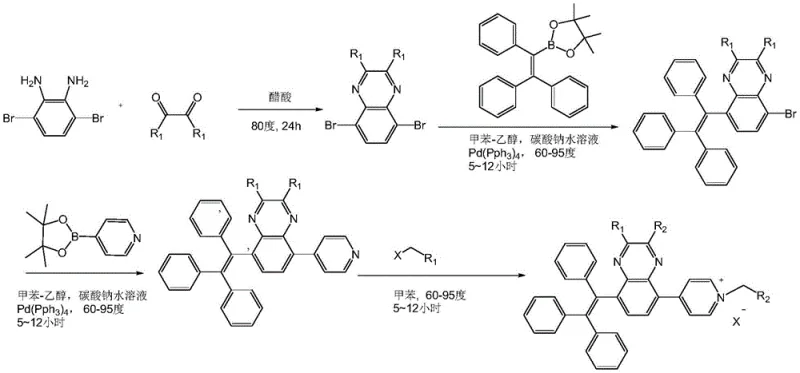
The heart of this synthetic protocol is the dual application of the Suzuki-Miyaura cross-coupling reaction, a palladium-catalyzed process renowned for its tolerance of functional groups and mild reaction conditions. In the first coupling stage, the 5,8-dibromo-quinoxaline intermediate reacts with triphenylethylene borate. The mechanism involves the oxidative addition of the aryl bromide to the Pd(0) catalyst, followed by transmetallation with the organoboron species in the presence of a base, and finally reductive elimination to forge the carbon-carbon bond. This step is critical for installing the AIE-active unit without disrupting the electron-deficient nature of the quinoxaline core. The second coupling introduces the pyridine ring, which serves as the handle for the final solubilizing modification. The choice of catalyst, such as Pd(PPh3)4, and the careful control of temperature between 60°C and 105°C are essential to minimize homocoupling byproducts and ensure high regioselectivity. This precision is vital for R&D directors focused on purity and杂质谱 (impurity profiles), as residual palladium or unreacted halides can act as quenchers in the final optoelectronic device.
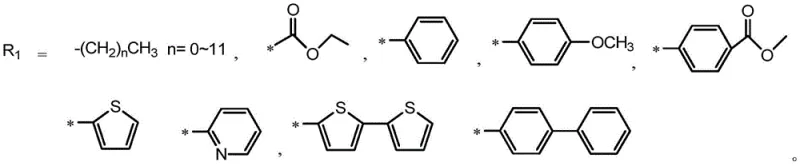
Following the construction of the neutral precursor, the final step involves a nucleophilic substitution reaction to generate the pyridinium salt. By reacting the pyridine nitrogen with an electrophile containing an active methylene group or a simple alkyl halide, the molecule undergoes quaternization. This transformation fundamentally alters the physicochemical properties of the compound, converting it from a lipophilic neutral species into a hydrophilic ionic salt. The resulting electrostatic repulsion between the cationic head groups prevents the formation of tight, non-emissive aggregates, thereby preserving the AIE effect even in highly polar environments. Additionally, the variability of the R1 and R2 groups, as depicted in the structural diagrams, allows chemists to modulate the HOMO-LUMO energy levels, thereby tuning the emission color from green to red. This level of mechanistic control underscores the value of partnering with a specialized supplier who understands the nuances of heterocyclic functionalization.
How to Synthesize Water-Soluble AIE Quinoxaline Efficiently
The synthesis of these advanced fluorophores requires strict adherence to the patented protocol to ensure optimal photophysical performance and solubility characteristics. The process begins with the condensation of diamines and diketones, followed by the critical palladium-catalyzed coupling steps that build molecular complexity. Each stage demands precise stoichiometric control and inert atmosphere conditions to prevent catalyst deactivation. The detailed standardized synthesis steps below outline the specific reaction parameters, workup procedures, and purification techniques required to achieve the high yields reported in the patent examples.
- Condense 1,4-dibromo-2,3-diaminobenzene with an o-diketone in glacial acetic acid under reflux to form the dibromo-quinoxaline core.
- Perform a Suzuki coupling reaction with triphenylethylene borate using a palladium catalyst to introduce the AIE-active propeller unit.
- Execute a second Suzuki coupling with pyridine boronic acid to attach the solubilizing pyridine moiety.
- React the neutral intermediate with an active methylene nucleophile or alkyl halide in toluene to form the final water-soluble pyridinium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this synthetic route offers distinct logistical and economic benefits over traditional fluorophore production. The reliance on Suzuki coupling, a widely industrialized reaction, means that the necessary catalysts and boronic acid building blocks are readily available from global chemical suppliers, reducing the risk of raw material shortages. Furthermore, the modular nature of the synthesis allows for the production of a library of derivatives from a common intermediate, streamlining inventory management and reducing the need for multiple dedicated production lines. The ability to isolate the final product through simple filtration or precipitation, as demonstrated in the patent examples, significantly reduces the consumption of silica gel and organic solvents associated with column chromatography. This simplification of downstream processing directly contributes to substantial cost savings and a reduced environmental footprint, aligning with modern green chemistry mandates.
- Cost Reduction in Manufacturing: The elimination of complex purification steps typically required for planar fluorophores leads to a more streamlined production workflow. By utilizing precipitation as a primary isolation method for the final pyridinium salts, manufacturers can avoid the high operational costs associated with large-scale chromatographic separation. Additionally, the high atom economy of the Suzuki reaction minimizes waste generation, further lowering disposal costs and enhancing the overall economic viability of producing high-purity OLED material intermediates.
- Enhanced Supply Chain Reliability: The starting materials, including 1,4-dibromo-2,3-diaminobenzene and various substituted diketones, are commodity chemicals with stable supply chains. This reduces dependency on exotic or single-source reagents that often cause bottlenecks in fine chemical manufacturing. The robustness of the reaction conditions, which tolerate a range of temperatures and bases, ensures that production can continue reliably even with minor fluctuations in utility supplies, guaranteeing consistent delivery schedules for downstream clients.
- Scalability and Environmental Compliance: The transition from laboratory scale to commercial production is facilitated by the use of standard reactor setups compatible with heating and inert gas purging. The process avoids the use of highly toxic reagents or extreme pressures, simplifying safety compliance and permitting. Moreover, the water solubility of the final product opens up opportunities for aqueous-based processing in device fabrication, potentially replacing volatile organic compounds (VOCs) in the manufacturing of displays and sensors, thus supporting broader corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these water-soluble quinoxaline derivatives. Understanding these aspects is crucial for integrating this technology into existing product pipelines and ensuring successful scale-up.
Q: What distinguishes this quinoxaline synthesis from traditional AIE material production?
A: Unlike traditional methods that often yield hydrophobic products requiring complex organic solvents, this patented route integrates a pyridinium salt functionality directly via quaternization. This structural modification drastically enhances water solubility, allowing the material to function effectively in aqueous biological environments or polar processing streams without losing its aggregation-induced emission properties.
Q: How does the triphenylethylene unit contribute to the material's performance?
A: The triphenylethylene unit acts as a classic 'propeller-shaped' AIEgen. In solution, its phenyl rings rotate freely, dissipating excited state energy non-radiatively. However, in the aggregated or solid state, this intramolecular rotation is restricted (RIR), blocking non-radiative pathways and forcing the molecule to emit strong fluorescence, which is critical for solid-state lighting and sensing applications.
Q: Is this synthesis scalable for industrial production of OLED intermediates?
A: Yes, the process relies on robust Suzuki-Miyaura coupling reactions which are well-established in industrial settings. The use of standard palladium catalysts and the ability to isolate intermediates via filtration or simple extraction suggests a high degree of scalability, making it suitable for commercial scale-up of complex electronic chemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoxaline Derivatives Supplier
As the demand for advanced luminescent materials continues to surge in the fields of bio-imaging and organic electronics, having a partner with deep technical expertise is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the precise palladium-catalyzed reactions and sensitive quaternization steps required for synthesizing these complex AIE materials. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of quinoxaline intermediate meets the exacting standards required for high-performance optoelectronic applications. Our commitment to quality assurance ensures that your downstream devices perform consistently, free from the defects caused by trace impurities.
We invite you to collaborate with us to unlock the full potential of water-soluble AIE technology for your specific applications. Whether you require custom synthesis of novel derivatives or bulk supply of established intermediates, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability.
