Advanced Liquid Phase Total Synthesis of Plecanatide Using Recyclable BDK Auxiliary Groups for Commercial Scale-up
The pharmaceutical industry is constantly seeking more efficient and sustainable methods for the production of complex peptide therapeutics like Plecanatide, a guanylate cyclase-C agonist used for treating chronic idiopathic constipation. A significant breakthrough in this domain is detailed in patent CN111285921A, which introduces a novel liquid-phase total synthesis method utilizing a specialized BDK (4,4'-bis(diphenylphosphinyloxy)diphenyl ketone) auxiliary group. This technology represents a paradigm shift from traditional solid-phase peptide synthesis (SPPS) and biological extraction methods, offering a robust pathway for high-purity API manufacturing. By replacing consumable solid-phase resins with a recyclable small-molecule phosphate ester carrier, this method addresses critical bottlenecks in scalability and waste management. The innovation lies in the unique physicochemical properties of the BDK group, which facilitates easy purification through crystallization, thereby streamlining the production workflow for multinational pharmaceutical enterprises seeking reliable supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional production of Plecanatide has largely relied on biological extraction or solid-phase peptide synthesis (SPPS), both of which present substantial challenges for large-scale commercial manufacturing. Biological extraction is inherently limited by finite biological resources and often results in low yields, cumbersome purification processes, and difficulties in achieving the high purity levels required for regulatory approval. On the other hand, while SPPS revolutionized peptide synthesis, it suffers from significant drawbacks when scaled up. The process typically requires a massive excess of protected amino acids and coupling reagents, often 3 to 5 times the stoichiometric amount, to drive reactions to completion in a heterogeneous system. Furthermore, the solid-phase resins and protecting groups used are destroyed during the cleavage process, generating vast amounts of hazardous chemical waste and driving up raw material costs. The reliance on high-boiling or toxic solvents like DMF and NMP further complicates environmental compliance and worker safety, making these conventional routes less attractive for modern green chemistry initiatives.
The Novel Approach
The liquid-phase synthesis method described in the patent overcomes these limitations by employing a soluble BDK auxiliary group that mimics the benefits of solid-phase synthesis while retaining the advantages of solution-phase chemistry. This approach allows for the construction of the Plecanatide amino acid sequence through iterative coupling cycles where each intermediate can be rigorously purified. Unlike solid resins, the BDK carrier enables the peptide intermediates to crystallize out of solution upon the addition of low-polarity solvents such as alkanes or ethers. This crystallization-driven purification effectively removes unreacted reagents and by-products without the need for complex chromatography at every step. Moreover, the BDK carrier itself is not consumed; it can be recovered from the mother liquor, regenerated, and reused, drastically reducing material waste. This method combines the high efficiency of automated synthesis with the scalability and purification ease of liquid-phase chemistry, offering a superior alternative for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into BDK-Assisted Liquid Phase Peptide Synthesis
The core of this technology lies in the chemical structure and behavior of the BDK auxiliary group, specifically the 4,4'-bis(diphenylphosphinyloxy)diphenyl ketone derivative. Mechanistically, the synthesis begins with the coupling of the BDK carrier to the C-terminus of an N-terminal Fmoc-protected amino acid, typically Leucine or its enantiomer, using standard dehydration coupling agents like EDC·HCl and DMAP. The resulting Fmoc-Xaa1-BDK intermediate possesses distinct solubility characteristics that are exploited for purification. In polar solvents, the intermediate remains soluble for reaction, but upon the introduction of non-polar solvents, it precipitates as high-purity crystals. This property is maintained throughout the chain elongation process. As amino acids are sequentially coupled and the N-terminal Fmoc group is removed using mild bases like DEA, the growing peptide chain remains anchored to the BDK group. This anchoring ensures that the entire molecule retains its crystallization capability, allowing for the removal of racemization by-products and deletion sequences that often plague peptide synthesis. The phosphate ester linkages are stable under coupling conditions but can be cleanly cleaved under acidic conditions alongside side-chain protecting groups.
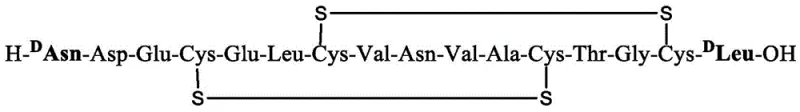
Following the assembly of the linear peptide chain, the method employs a sophisticated orthogonal strategy for disulfide bond formation, which is critical for the bioactivity of Plecanatide. The patent describes a two-step oxidation process where specific cysteine residues are selectively deprotected and oxidized to form the correct intramolecular disulfide bridges (Cys4-Cys12 and Cys7-Cys15). Initially, milder oxidizing conditions are used to form one set of bonds, followed by iodine-mediated oxidation for the second set. This controlled cyclization minimizes the formation of intermolecular polymers or incorrect disulfide isomers, ensuring the final product matches the native conformation of the hormone. The final step involves a cocktail of trifluoroacetic acid (TFA), thioanisole, EDT, and water to simultaneously remove all remaining side-chain protecting groups (such as tBu, Trt, and Acm) and cleave the peptide from the BDK carrier. The free peptide then precipitates due to low solubility in the organic medium, while the BDK carrier remains in the solution phase for recovery, completing the cycle of efficient synthesis and material conservation.
How to Synthesize Plecanatide Efficiently
The synthesis of Plecanatide using this BDK-assisted liquid phase method involves a systematic series of coupling, purification, and deprotection steps that ensure high fidelity of the amino acid sequence. The process begins with the activation of the BDK carrier and its conjugation to the first amino acid, followed by iterative cycles of Fmoc deprotection and amino acid coupling. Each cycle is punctuated by a crystallization step that purifies the growing peptide chain, eliminating the need for extensive column chromatography until the final stages. This streamlined workflow significantly reduces processing time and solvent consumption compared to traditional methods. For detailed operational parameters, including specific solvent ratios, reaction temperatures, and stoichiometry, operators should refer to the standardized protocols derived from the patent data. The following guide outlines the critical phases of this advanced synthesis route.
- Couple the BDK auxiliary group with Fmoc-protected amino acids using dehydration coupling agents to form the initial intermediate.
- Perform separation and purification by inducing crystallization of the BDK-peptide intermediate in low-polarity solvents, filtering out impurities.
- Remove N-terminal Fmoc protection and repeat coupling cycles to extend the peptide chain, followed by side-chain deprotection and oxidative cyclization to form disulfide bonds.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this BDK-based liquid phase synthesis offers transformative economic and logistical benefits. The primary advantage stems from the drastic reduction in raw material waste. In traditional SPPS, the resin support is single-use, representing a significant recurring cost that scales linearly with production volume. In contrast, the BDK carrier is recyclable; after the peptide is cleaved, the carrier residue can be precipitated, purified, and reintroduced into the synthesis cycle. This circular economy approach within the manufacturing process leads to substantial cost savings on high-value starting materials. Furthermore, the ability to purify intermediates via crystallization rather than preparative HPLC at every step reduces the consumption of expensive chromatography columns and solvents, lowering the overall cost of goods sold (COGS) for the final API.

- Cost Reduction in Manufacturing: The elimination of solid-phase resin waste and the recovery of the BDK auxiliary group directly translate to lower material costs. Additionally, the process operates under milder conditions and utilizes simpler workup procedures like filtration and washing, which reduces energy consumption and labor hours associated with complex purification tasks. The reduction in solvent usage, particularly the avoidance of large volumes of DMF and NMP required for swelling resins, further decreases waste disposal costs and regulatory burdens related to volatile organic compound (VOC) emissions.
- Enhanced Supply Chain Reliability: Liquid-phase synthesis is inherently more scalable than solid-phase methods, which often face mass transfer limitations in large reactors. This technology allows for the production of Plecanatide in larger batch sizes without compromising quality, ensuring a more stable and continuous supply for downstream formulation. The robustness of the crystallization purification steps also means that the process is less sensitive to minor fluctuations in reagent quality, leading to more consistent batch-to-batch yields and reducing the risk of supply disruptions caused by failed production runs.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns with green chemistry principles by minimizing hazardous waste generation. The recyclability of the phosphate ester carrier significantly reduces the chemical footprint of the manufacturing process. This is increasingly important for pharmaceutical companies facing stricter environmental regulations and corporate sustainability goals. The ability to scale up without a proportional increase in waste disposal requirements makes this technology a future-proof solution for long-term commercial production of complex peptides.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this BDK-assisted synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for R&D teams evaluating process transfer and for procurement teams assessing supplier capabilities.
Q: What are the advantages of the BDK auxiliary group over traditional solid-phase resins?
A: The BDK auxiliary group allows for liquid-phase synthesis where intermediates can be purified via simple crystallization and filtration, unlike solid-phase resins which are consumed and generate significant waste. Additionally, the BDK carrier can be recovered and recycled, reducing raw material costs.
Q: How does this method improve the purity of Plecanatide?
A: By utilizing the unique crystallization properties of the phosphate ester carrier in different solvent systems, impurities are effectively separated at each coupling step. This iterative purification ensures high purity of the final peptide before the final global deprotection and cyclization steps.
Q: Is the BDK carrier reusable in this synthesis process?
A: Yes, a key feature of this invention is the recyclability of the BDK carrier. After the peptide is cleaved, the residual BDK derivative can be precipitated, filtered, regenerated, and reused in subsequent synthesis batches, significantly enhancing process sustainability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Plecanatide Supplier
The technological advancements detailed in patent CN111285921A highlight the immense potential of liquid-phase peptide synthesis for the commercial production of high-value APIs like Plecanatide. At NINGBO INNO PHARMCHEM, we pride ourselves on staying at the forefront of such innovative manufacturing technologies. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and high-quality supply of critical pharmaceutical intermediates. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the exacting standards required for global regulatory submissions. We understand the complexities of peptide chemistry and are dedicated to optimizing these advanced synthetic routes for maximum efficiency and yield.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized processes can reduce your overall procurement costs. Please contact us to request specific COA data and route feasibility assessments for Plecanatide and its analogs. Let us help you secure a sustainable and cost-effective supply of this vital therapeutic agent.
