Revolutionizing Colchicine Production: A Scalable Asymmetric Synthesis Strategy for Global Pharma
Revolutionizing Colchicine Production: A Scalable Asymmetric Synthesis Strategy for Global Pharma
The pharmaceutical industry has long relied on the extraction of colchicine from Colchicum autumnale, a process fraught with inefficiencies, low yields, and supply chain volatility. However, a groundbreaking patent, CN110963937B, introduces a paradigm shift towards a fully synthetic, asymmetric approach that promises to redefine the availability of this critical anti-gout agent. This novel methodology leverages inexpensive commercial isovanillin as the starting material, bypassing the agricultural bottlenecks of traditional sourcing. By integrating a metal iridium-catalyzed asymmetric allylic amination as the stereochemistry-determining step, the process achieves exceptional enantiomeric excess while maintaining operational simplicity. For R&D directors and procurement strategists, this represents a significant opportunity to secure a reliable colchicine intermediate supplier capable of delivering high-purity materials with consistent quality attributes, independent of harvest seasons or geopolitical agricultural constraints.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the global supply of colchicine has been tethered to the extraction from bulbous plants, a method that is inherently unstable and economically inefficient. The extraction process is not only time-consuming and labor-intensive but also yields very low quantities of the active alkaloid relative to the biomass processed. Furthermore, the biological source material is subject to significant variability in alkaloid content due to environmental factors, leading to batch-to-batch inconsistencies that are unacceptable for modern Good Manufacturing Practice (GMP) standards. From a supply chain perspective, reliance on plant extraction creates a fragile dependency on agricultural outputs, making it difficult to scale production rapidly in response to surging demand for gout treatments or new oncology applications. Additionally, the structural modification of naturally extracted colchicine to create safer analogs like allocolchicine is chemically arduous, limiting the ability of medicinal chemists to explore structure-activity relationships effectively for next-generation drug development.
The Novel Approach
In stark contrast, the synthetic route disclosed in CN110963937B offers a robust, linear progression from simple aromatic precursors to the complex tricyclic colchicine scaffold. The strategy initiates with the conversion of isovanillin into an allylic alcohol, which then undergoes a highly stereoselective iridium-catalyzed transformation to install the crucial chiral center with >99% ee. This is followed by a Suzuki coupling reaction to assemble the biaryl framework, setting the stage for a rapid intramolecular oxidative coupling that constructs the seven-membered ring characteristic of allocolchicine. The synthesis culminates in a biomimetic cyclopropane ring-opening strategy that elegantly installs the tropolone ring system found in the final colchicine structure. This modular approach not only simplifies the synthesis but also allows for the potential production of various derivatives by modifying the starting materials, thereby offering a versatile platform for pharmaceutical innovation.
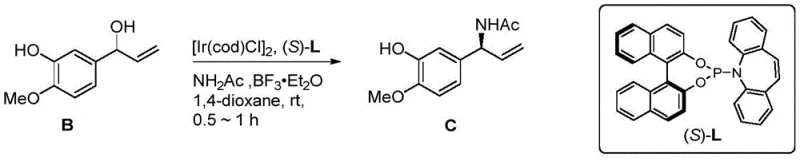
Mechanistic Insights into Ir-Catalyzed Asymmetric Allylic Amination and Oxidative Cyclization
The cornerstone of this synthetic achievement is the iridium-catalyzed asymmetric allylic amination, which serves as the primary source of chirality for the entire molecule. In this mechanistic sequence, the iridium catalyst, coordinated with the chiral phosphine ligand (S)-L, activates the allylic alcohol substrate through the formation of a pi-allyl iridium complex. The nucleophilic attack by acetamide occurs with high regio- and enantioselectivity, driven by the steric environment created by the bulky ligand framework. This step is critical because it establishes the absolute configuration at the benzylic position early in the synthesis, ensuring that all subsequent transformations proceed with the correct stereochemical integrity. The use of Lewis acids such as boron trifluoride etherate further enhances the electrophilicity of the intermediate, facilitating the reaction under mild conditions that preserve sensitive functional groups. This level of control is essential for producing high-purity pharmaceutical intermediates that meet stringent regulatory requirements for impurity profiles.
Following the establishment of chirality, the synthesis employs a sophisticated intramolecular oxidative coupling to close the seven-membered ring, a transformation that mimics biosynthetic pathways found in nature. This step involves the oxidation of the phenolic moiety in the presence of hypervalent iodine reagents, generating a reactive radical or cationic species that attacks the adjacent aromatic ring. The resulting cyclization forms the allocolchicine core with high efficiency, demonstrating the power of biomimetic chemistry in simplifying complex total syntheses. Subsequent steps involve an oxidative dearomatization to generate a dienone system, followed by a Corey-Chaykovsky cyclopropanation. The final ring-opening rearrangement, mediated by trifluoroacetic acid, relieves the strain of the cyclopropane ring to expand the system into the characteristic tropolone ring of colchicine. This cascade of reactions showcases a deep understanding of physical organic chemistry, allowing for the construction of complex molecular architectures in a concise manner.
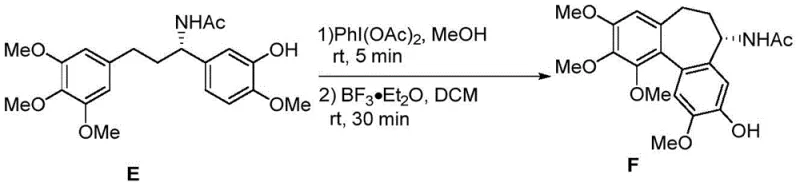
How to Synthesize Colchicine Efficiently
The execution of this synthesis requires precise control over reaction conditions, particularly regarding moisture sensitivity and temperature management during the organometallic steps. The process begins with the preparation of the allylic alcohol via Grignard addition, followed immediately by the asymmetric amination without the need for extensive purification, which streamlines the workflow. The subsequent Suzuki coupling utilizes standard palladium catalysis but requires careful optimization of the base and solvent system to ensure high conversion of the sterically hindered substrates. The oxidative coupling and ring-expansion steps demand strict anhydrous conditions to prevent side reactions and ensure the stability of the reactive intermediates. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below which outlines the exact protocols validated in the patent examples.
- Perform Grignard addition on isovanillin to generate allyl alcohol intermediate, followed by Ir-catalyzed asymmetric allylic amination to establish chirality.
- Execute Suzuki coupling with trimethoxyphenyl bromide to link the aromatic rings, forming the linear precursor.
- Complete the synthesis via intramolecular oxidative coupling to form allocolchicine, followed by oxidative dearomatization, cyclopropanation, and acid-mediated ring-opening rearrangement.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from extraction to this synthetic route offers profound strategic benefits that extend beyond mere cost considerations. The primary advantage lies in the decoupling of production from agricultural cycles, eliminating the risks associated with crop failures, seasonal shortages, and price volatility of raw plant materials. By utilizing commodity chemicals like isovanillin and vinylmagnesium bromide, manufacturers can secure a stable and predictable supply chain that is resilient to external shocks. Furthermore, the synthetic route allows for the production of colchicine and its analogs at scales that are simply impossible to achieve through extraction, enabling the pharmaceutical industry to meet growing global demand for gout medications and investigate new therapeutic indications without supply constraints. This reliability is paramount for maintaining continuous manufacturing lines and avoiding costly production stoppages.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive, commercially available starting materials and the elimination of expensive purification steps between certain intermediates. The high atom economy of the key coupling reactions and the ability to telescope multiple steps without isolation significantly reduce solvent consumption and waste generation. Moreover, the avoidance of precious metal catalysts in the later stages of the synthesis, relying instead on more abundant reagents for the ring expansion, contributes to a lower overall cost of goods sold. These efficiencies translate into substantial cost savings that can be passed down the supply chain, making the final API more affordable for healthcare systems while maintaining healthy margins for manufacturers.
- Enhanced Supply Chain Reliability: Synthetic manufacturing provides a level of consistency and scalability that natural extraction cannot match. Facilities can ramp up production capacity by simply adding more reactors, rather than waiting for the next harvest season. This agility allows suppliers to respond quickly to market fluctuations and emergency demands, such as sudden spikes in gout outbreaks or new clinical trial requirements. Additionally, the synthetic route produces a cleaner impurity profile compared to plant extracts, which often contain a complex mixture of related alkaloids that are difficult to separate. This purity reduces the burden on downstream processing and quality control, further enhancing the reliability of the supply chain and reducing the risk of batch rejections.
- Scalability and Environmental Compliance: The reaction conditions employed in this synthesis are generally mild, operating at temperatures ranging from 0°C to 90°C, which reduces energy consumption compared to high-temperature processes. The use of standard organic solvents like THF, methanol, and dichloromethane allows for established recycling and recovery protocols, minimizing environmental impact. The process avoids the use of highly toxic reagents in the final steps, facilitating easier waste treatment and compliance with increasingly stringent environmental regulations. This green chemistry profile not only lowers disposal costs but also aligns with the sustainability goals of modern pharmaceutical companies, making it an attractive option for long-term partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. They are derived from the specific experimental data and advantageous effects described in the patent documentation, providing clarity on the feasibility and benefits of adopting this route for industrial production. Understanding these details is crucial for technical teams evaluating the transfer of this technology to pilot or commercial scale.
Q: What is the key advantage of this synthetic route over traditional extraction?
A: Unlike plant extraction which suffers from low yields and seasonal variability, this chemical synthesis uses cheap commercial isovanillin and achieves an overall yield of approximately 30% with high stereocontrol, ensuring consistent supply.
Q: How is chirality controlled in this process?
A: Chirality is introduced early in the sequence via a highly enantioselective iridium-catalyzed asymmetric allylic amination reaction using a specific chiral phosphine ligand (S)-L, achieving >99% ee.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the route utilizes commercially available reagents, mild reaction conditions (mostly room temperature to 90°C), and avoids toxic heavy metal residues in the final steps, making it highly amenable to kilogram-to-ton scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Colchicine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this asymmetric synthesis technology for the global pharmaceutical market. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate complex laboratory routes like CN110963937B into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We are committed to delivering high-purity colchicine intermediates and APIs that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our focus on process optimization ensures that we can maximize yields and minimize costs, providing you with a competitive edge in the marketplace.
We invite you to collaborate with us to explore the full potential of this synthetic route for your specific drug development programs. Whether you require custom synthesis of colchicine analogs or large-scale production of the parent compound, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are eager to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your supply chain goals and accelerate your time to market.
