Scalable Synthesis of Broad-Spectrum Carbapenem Antibiotics for Commercial Production
Scalable Synthesis of Broad-Spectrum Carbapenem Antibiotics for Commercial Production
The pharmaceutical landscape continuously demands novel antibiotic agents capable of overcoming resistant bacterial strains while maintaining metabolic stability. Patent CN1079224A introduces a significant advancement in the field of carbapenem chemistry, detailing a series of substituted carbapenem compounds characterized by a unique pyrrolidin-4-ylthio side chain linked to a phenyl formamyl group. These compounds exhibit broad-spectrum antibacterial activity against both Gram-positive and Gram-negative organisms, including aerobic and anaerobic bacteria. The core innovation lies in the specific substitution patterns on the phenyl ring, which enhance beta-lactamase stability and prolong the duration of action in mammalian systems. This technology represents a pivotal opportunity for manufacturers seeking to diversify their portfolio of high-purity antibiotic intermediates.
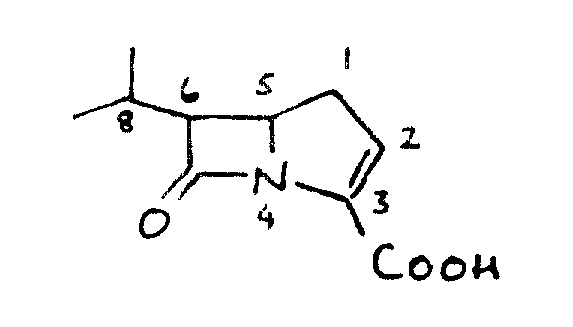
The structural versatility of these compounds allows for extensive modification at the R4 and R5 positions on the phenyl ring, enabling the tuning of pharmacokinetic properties without compromising the integrity of the beta-lactam nucleus. The patent outlines methods for preparing these compounds as pharmaceutically acceptable salts or in vivo hydrolyzable esters, ensuring flexibility in formulation development. For procurement specialists and supply chain managers, understanding the synthetic accessibility of these complex molecules is crucial for securing a reliable carbapenem intermediate supplier capable of meeting global demand.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of carbapenem antibiotics often faces significant hurdles regarding the stability of the beta-lactam ring during functionalization. Conventional routes frequently require harsh deprotection conditions that can lead to ring opening or epimerization at the C-1 position, resulting in reduced potency and increased impurity profiles. Furthermore, introducing complex side chains at the 2-position typically involves multiple protection and deprotection steps that lower overall yield and increase manufacturing costs. The sensitivity of the carbapenem nucleus to nucleophilic attack necessitates precise control over reaction conditions, which can be difficult to maintain on a commercial scale. These challenges often result in supply chain bottlenecks and inconsistent quality of the final active pharmaceutical ingredient.
The Novel Approach
The methodology described in CN1079224A overcomes these limitations through a strategic use of orthogonal protecting groups and mild coupling conditions. By utilizing allyl esters and nitrobenzyl groups, the synthesis allows for the construction of the complex side chain prior to its attachment to the carbapenem nucleus. This convergent approach minimizes the exposure of the sensitive beta-lactam ring to reactive conditions until the final stages. The key innovation involves the coupling of a protected mercapto-pyrrolidine derivative with a carbapenem phosphoric acid ester, a reaction that proceeds with high stereoselectivity. This ensures the preservation of the critical trans-configuration of the beta-lactam protons, which is essential for biological activity.
Mechanistic Insights into Carbapenem Side-Chain Coupling
The core of this synthetic strategy relies on the nucleophilic displacement of a leaving group on the carbapenem nucleus by the sulfur atom of the pyrrolidine side chain. Specifically, the reaction involves a carbapenem-2-phosphoric acid ester derivative reacting with a 4-mercapto-pyrrolidine-2-carboxamide intermediate. The phosphoric acid ester acts as an activated electrophile, facilitating the formation of the thioether bond under mild basic conditions, typically using organic amines like diisopropylethylamine. This mechanism avoids the use of heavy metal catalysts in the coupling step, thereby reducing the risk of metal contamination in the final product. The reaction is typically conducted at low temperatures, ranging from -25°C to room temperature, to prevent degradation of the reactants.
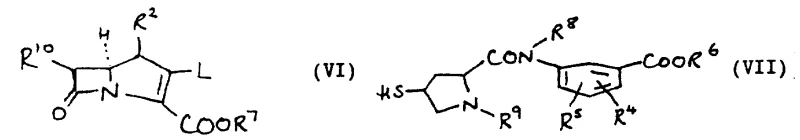
Following the coupling, the stereochemical integrity of the molecule is maintained through careful control of the reaction environment. The patent emphasizes the importance of the absolute stereochemistry at the 5, 6, and 8 positions, as well as the 2' and 4' positions on the pyrrolidine ring. The preferred configuration is (1R, 5S, 6S, 8R, 2'S, 4'S), which correlates with optimal antibacterial efficacy. The use of chiral precursors for the pyrrolidine ring ensures that the final product is obtained as a single diastereomer, simplifying downstream purification processes. This level of stereocontrol is vital for regulatory compliance and ensures consistent therapeutic performance across different batches.
How to Synthesize Carbapenem Derivatives Efficiently
The synthesis of these high-value antibiotic intermediates follows a logical sequence designed to maximize yield and purity while minimizing waste. The process begins with the preparation of the substituted aniline side chain, followed by its condensation with a proline derivative to form the pyrrolidine scaffold. This scaffold is then converted into a mercaptan and coupled with the carbapenem nucleus. The final step involves the simultaneous removal of protecting groups to reveal the active carboxylic acid functionalities. Detailed standardized synthesis steps are provided in the guide below to assist R&D teams in replicating this process.
- Preparation of the pyrrolidine-thio side chain via condensation of substituted aniline derivatives with proline intermediates.
- Coupling of the protected side chain mercaptan with a carbapenem phosphoric acid ester nucleus under basic conditions.
- Final deprotection of allyl and nitrobenzyl groups using palladium catalysis and hydrogenation to yield the active carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers substantial strategic benefits beyond mere chemical novelty. The modularity of the synthesis allows for the production of a wide library of analogues using a common set of intermediates, which significantly streamlines inventory management. By standardizing the core coupling and deprotection steps, manufacturers can reduce the complexity of their production schedules and mitigate the risk of supply disruptions associated with specialized reagents. The ability to generate diverse compounds from a single platform enhances the resilience of the supply chain against market fluctuations.
- Cost Reduction in Manufacturing: The use of palladium-catalyzed deprotection for allyl groups is a highly efficient method that operates under mild conditions, reducing energy consumption and equipment wear. Unlike harsh acidic or basic hydrolysis, this method preserves the integrity of the beta-lactam ring, leading to higher recovery rates of the active ingredient. The elimination of expensive transition metal catalysts in the coupling step further lowers the raw material costs. Additionally, the high stereoselectivity of the process reduces the need for costly chiral chromatography separations, resulting in significant operational savings.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as substituted nitrobenzoic acids and allyl bromide, are commodity chemicals available from multiple global sources. This diversity of supply reduces dependency on single-source vendors and mitigates the risk of geopolitical disruptions. The robustness of the reaction conditions means that the process can be transferred between manufacturing sites with minimal re-validation, ensuring continuity of supply. Furthermore, the stability of the protected intermediates allows for stockpiling, providing a buffer against sudden spikes in demand.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, utilizing solvents and reagents that are compatible with large-scale reactor systems. The deprotection steps generate benign byproducts that are easier to treat in standard wastewater facilities compared to traditional methods involving heavy metals or strong acids. The high atom economy of the coupling reaction minimizes waste generation, aligning with modern green chemistry principles. This environmental compatibility simplifies regulatory approvals and reduces the long-term liability associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the stability, synthesis, and application of these carbapenem derivatives. Understanding these aspects is critical for R&D directors evaluating the feasibility of incorporating these intermediates into their development pipelines. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation.
Q: What distinguishes these carbapenem derivatives from imipenem?
A: These compounds feature specific phenyl formamyl side chains at the 2-position which provide satisfactory stability to beta-lactamases and extraordinary duration of effect compared to earlier generations like imipenem.
Q: How is the stereochemistry controlled during synthesis?
A: The synthesis utilizes chiral starting materials and specific coupling conditions to maintain the critical 1R, 5S, 6S, 8R configuration essential for antibacterial activity.
Q: Are these intermediates suitable for large-scale manufacturing?
A: Yes, the process employs robust protecting group strategies like allyl esters which can be removed under mild palladium-catalyzed conditions, facilitating scalable production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbapenem Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, specializing in the production of complex pharmaceutical intermediates like the carbapenem derivatives described in CN1079224A. Our facility is equipped with state-of-the-art reactors and purification systems capable of handling sensitive beta-lactam chemistry with precision. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of major pharmaceutical companies. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which utilize advanced analytical techniques to verify the identity and potency of every batch.
We invite potential partners to engage with our technical procurement team to discuss how our capabilities can support your specific project needs. Whether you require custom synthesis of a specific analogue or large-scale supply of a standard intermediate, we are prepared to offer a Customized Cost-Saving Analysis tailored to your manufacturing constraints. Contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate your antibiotic development programs with reliable, high-quality intermediates.
