Scalable Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazole Intermediates
Scalable Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazole Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing complex heterocyclic scaffolds, particularly those incorporating fluorine atoms which are pivotal for enhancing metabolic stability and bioavailability. Patent CN113105402B introduces a significant advancement in this domain by disclosing a novel preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds. These structural motifs are ubiquitous in high-value medicinal chemistry, serving as core components in blockbuster drugs such as Maraviroc, Sitagliptin, and Deferasirox, as illustrated in the structural diversity of bioactive molecules below. The ability to efficiently synthesize these trifluoromethyl-containing triazoles represents a critical capability for any reliable pharmaceutical intermediate supplier aiming to support the development of next-generation therapeutics.
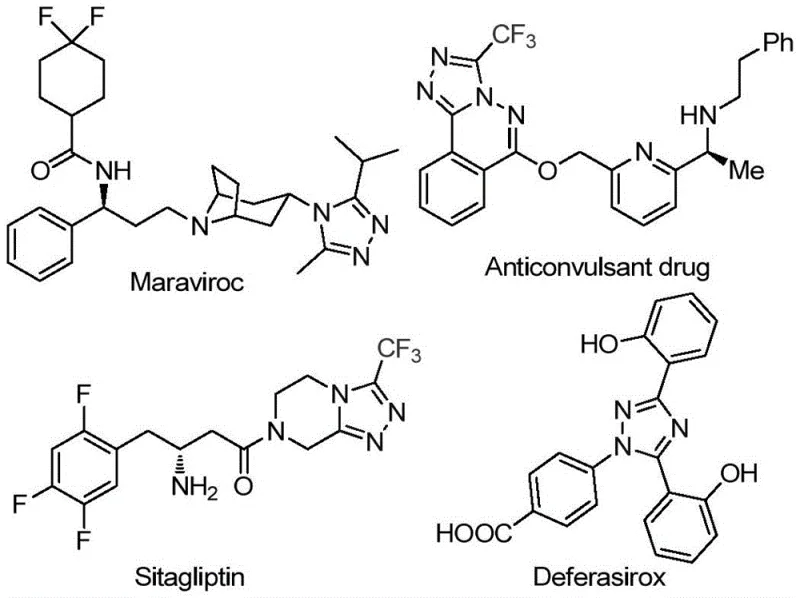
This patented technology addresses the growing demand for high-purity pharmaceutical intermediates by offering a synthetic route that bypasses the limitations of traditional transition-metal catalysis. By leveraging a non-metallic iodine-promoted system, the process not only reduces the environmental footprint associated with heavy metal waste but also streamlines the purification workflow, which is a major pain point in commercial scale-up of complex pharmaceutical intermediates. The method utilizes readily available aryl ethanones and trifluoroethylimine hydrazides as starting materials, ensuring a stable supply chain and predictable cost structures for procurement teams managing large-volume API production campaigns.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted 1,2,4-triazole rings, especially those bearing both trifluoromethyl and acyl groups, has relied heavily on transition metal catalysts or harsh reaction conditions that pose significant challenges for industrial application. Conventional pathways often necessitate the use of expensive copper or palladium complexes, which not only inflate the raw material costs but also introduce severe complications regarding residual metal limits in the final active pharmaceutical ingredient. Furthermore, many existing protocols require stringent anhydrous and oxygen-free environments, demanding specialized equipment and increasing the operational complexity and energy consumption of the manufacturing process. These factors collectively hinder the cost reduction in pharmaceutical intermediate manufacturing and can lead to extended lead times due to the need for rigorous metal scavenging and purification steps.
The Novel Approach
In stark contrast, the methodology outlined in CN113105402B employs a metal-free strategy driven by elemental iodine in a dimethyl sulfoxide (DMSO) solvent system, effectively circumventing the drawbacks of heavy metal catalysis. This innovative approach allows the reaction to proceed under relatively mild conditions without the need for inert atmosphere protection, thereby drastically simplifying the operational requirements and reducing the capital expenditure needed for reactor setup. The use of inexpensive and commercially abundant reagents such as aryl ethanones and iodine ensures that the cost of goods sold (COGS) remains competitive, making this route highly attractive for high-purity pharmaceutical intermediate production. Additionally, the broad substrate tolerance demonstrated in the patent examples suggests that this platform technology can be adapted to synthesize a wide array of analogues, providing R&D directors with the flexibility to explore diverse chemical spaces for drug discovery programs.
Mechanistic Insights into Iodine-Promoted Cyclization
The core of this synthetic breakthrough lies in the tandem oxidative cyclization mechanism facilitated by the iodine-DMSO system. The reaction initiates with the iodination and subsequent Kornblum oxidation of the aryl ethanone substrate, transforming it into an reactive aryl diketone intermediate in situ. This oxidative activation is crucial as it generates the electrophilic species necessary for the subsequent condensation step. Following this initial transformation, the trifluoroethylimine hydrazide acts as a nucleophile, attacking the activated carbonyl species to form a hydrazone intermediate. This sequence avoids the isolation of unstable intermediates, telescoping multiple synthetic steps into a single pot operation which enhances overall atom economy and process efficiency.
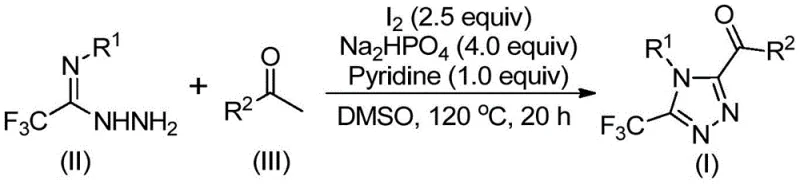
Upon formation of the hydrazone, the reaction mixture undergoes an intramolecular cyclization promoted by the continued presence of iodine and the basic environment provided by pyridine and sodium dihydrogen phosphate. This cyclization step closes the triazole ring, establishing the stable 1,2,4-triazole core while retaining the critical trifluoromethyl group at the 3-position and the acyl group at the 5-position. The mechanistic pathway is designed to minimize side reactions and byproduct formation, resulting in a cleaner crude reaction profile that facilitates easier downstream processing. For R&D teams, understanding this mechanism is vital for troubleshooting and optimizing reaction parameters such as temperature and stoichiometry to maximize yield and purity for specific substrate combinations.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The practical execution of this synthesis involves a straightforward two-stage heating protocol that can be easily implemented in standard glass-lined or stainless steel reactors. The process begins by dissolving the aryl ethanone and a portion of iodine in DMSO, followed by heating to promote the initial oxidation. Subsequently, the remaining reagents including the hydrazide, base, and additional iodine are introduced to drive the cyclization to completion. The detailed standardized synthesis steps, including precise molar ratios and temperature ramps validated across multiple examples in the patent, are outlined in the guide below to ensure reproducible results for process chemists.
- Mix aryl ethyl ketone and iodine in DMSO, heating to 90-110°C for 4-6 hours to initiate oxidation.
- Add sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the solution to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and purification.
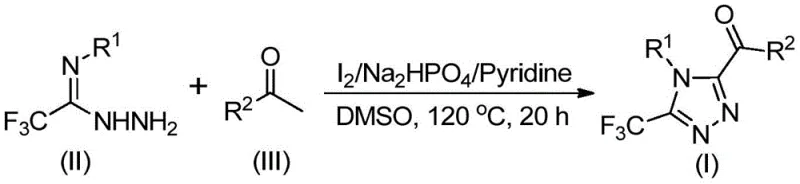
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this iodine-promoted synthesis offers substantial strategic advantages for procurement managers and supply chain heads looking to optimize their vendor networks and reduce manufacturing overheads. The elimination of precious metal catalysts removes a significant cost driver and mitigates the risk of supply chain disruptions associated with scarce metals like palladium. Furthermore, the use of commodity chemicals such as DMSO, iodine, and simple aryl ketones ensures that raw material sourcing is robust and less susceptible to geopolitical volatility, thereby enhancing supply chain reliability for long-term production contracts.
- Cost Reduction in Manufacturing: The absence of expensive transition metal catalysts and the ability to operate without strict anhydrous conditions significantly lowers the operational expenditure associated with this synthesis. By removing the need for specialized metal scavenging resins and complex purification trains required to meet strict residual metal specifications, manufacturers can achieve substantial cost savings in the final processing stages. This economic efficiency is further amplified by the high atom economy of the tandem reaction, which minimizes waste generation and maximizes the utilization of starting materials, directly contributing to a lower cost per kilogram of the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The reliance on widely available, off-the-shelf reagents such as aryl ethanones and elemental iodine ensures a stable and continuous supply of inputs for production. Unlike proprietary catalysts that may have single-source suppliers and long lead times, the commodities used in this process can be sourced from multiple global vendors, reducing the risk of production stoppages due to material shortages. This diversification of the supply base allows for better inventory management and more flexible procurement strategies, ensuring that delivery schedules for critical API intermediates are met consistently.
- Scalability and Environmental Compliance: The simplicity of the reaction conditions, which do not require high-pressure equipment or cryogenic cooling, makes this process inherently scalable from laboratory benchtop to multi-ton commercial production. The use of DMSO, a solvent with well-established recovery and recycling protocols, aligns with modern green chemistry principles and facilitates compliance with increasingly stringent environmental regulations. The reduced generation of heavy metal waste simplifies effluent treatment processes, lowering the environmental compliance burden and associated disposal costs for manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazole synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of adopting this route for industrial applications.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN113105402B utilizes elemental iodine as a non-metallic promoter, eliminating the need for costly heavy metal catalysts and simplifying downstream purification.
Q: What are the typical reaction conditions for this triazole formation?
A: The process involves a two-stage heating protocol in DMSO: an initial oxidation phase at 90-110°C followed by a cyclization phase at 110-130°C, without requiring strict anhydrous or anaerobic conditions.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method is easily scalable from gram level to industrial production due to the use of cheap, readily available raw materials and simple operational procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development and commercialization. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and robust. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee batch-to-batch consistency and regulatory compliance.
We invite potential partners to engage with our technical procurement team to discuss how this advanced iodine-promoted synthesis can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this metal-free route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific project requirements, ensuring a successful and profitable partnership.
