Scalable Iodine-Promoted Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Commercial API Production
Scalable Iodine-Promoted Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Commercial API Production
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles, particularly 1,2,4-triazoles, which serve as critical scaffolds in numerous high-value active pharmaceutical ingredients. As detailed in patent CN113105402B, a novel preparation method has been developed that addresses longstanding challenges in synthesizing 3,4,5-trisubstituted 1,2,4-triazole compounds containing both trifluoromethyl and acyl groups. This technology is particularly relevant given the prevalence of this core structure in blockbuster drugs such as Maraviroc, Sitagliptin, and Deferasirox, as illustrated in the structural overview below. The ability to efficiently construct these complex molecular architectures without relying on precious metal catalysis represents a significant advancement for reliable pharmaceutical intermediate supplier networks aiming to optimize their production pipelines.
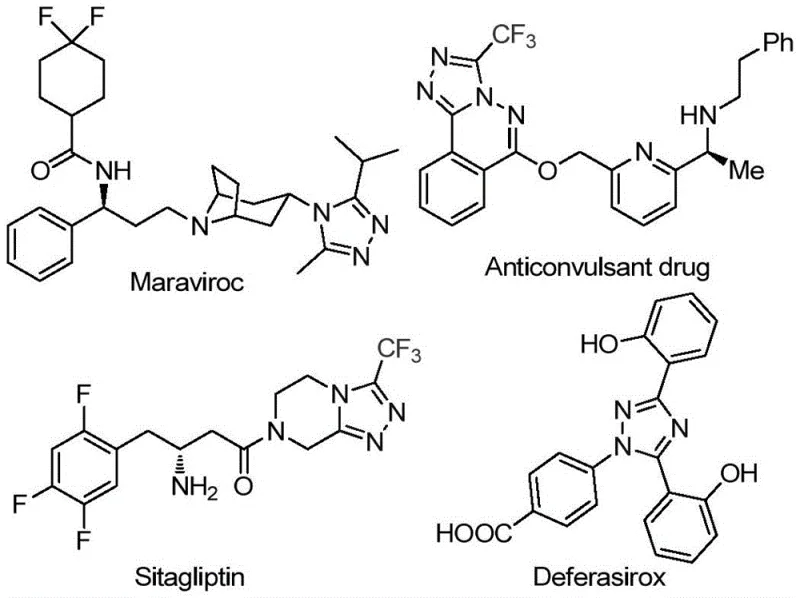
The strategic importance of this synthesis lies in its direct applicability to the manufacturing of advanced medicinal agents where metabolic stability and lipophilicity are paramount. The introduction of a trifluoromethyl group into the heterocyclic core significantly enhances the physicochemical properties of the parent compound, a feature highly sought after by R&D directors focusing on next-generation therapeutics. By leveraging this patented methodology, chemical manufacturers can access a versatile platform for generating diverse libraries of triazole derivatives, thereby accelerating the discovery and development phases for new drug candidates while ensuring compliance with stringent quality standards required for global market entry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted 1,2,4-triazole rings has often relied on transition metal-catalyzed cyclizations or multi-step sequences involving hazardous reagents and苛刻 reaction conditions. Traditional pathways frequently necessitate the use of expensive palladium or copper catalysts, which not only inflate the raw material costs but also introduce significant downstream processing challenges related to heavy metal removal. Furthermore, many existing protocols require strictly anhydrous and oxygen-free environments, demanding specialized equipment and inert gas handling that complicate operations on a large scale. These factors collectively contribute to extended lead times and increased operational expenditures, creating bottlenecks for procurement managers tasked with maintaining cost-effective supply chains for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN113105402B utilizes a metal-free promotion system driven by elemental iodine in dimethyl sulfoxide (DMSO). This innovative approach eliminates the need for toxic heavy metal catalysts entirely, thereby streamlining the purification process and reducing the environmental footprint of the synthesis. The reaction proceeds through a tandem sequence involving iodination and Kornblum oxidation followed by cyclization, all within a single pot under relatively mild thermal conditions. This simplicity translates directly into operational efficiency, allowing for easier scale-up from gram to kilogram levels without the need for complex engineering controls. For supply chain heads, this robustness ensures greater continuity of supply and reduced risk of batch failures due to sensitive reaction parameters.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The core of this synthetic breakthrough involves a sophisticated cascade reaction initiated by the interaction of aryl ethyl ketones with elemental iodine in a polar aprotic solvent. The mechanism likely proceeds via an initial iodination of the ketone alpha-position, followed by a Kornblum oxidation mediated by DMSO to generate an alpha-dicarbonyl intermediate in situ. This reactive species then undergoes condensation with trifluoroethylimide hydrazide to form a hydrazone intermediate, which subsequently cyclizes under the influence of the iodine and base system to yield the final 3,4,5-trisubstituted 1,2,4-triazole scaffold. Understanding this mechanistic pathway is crucial for R&D teams aiming to optimize reaction parameters for specific substrates, as it highlights the dual role of iodine as both an oxidant promoter and a cyclization facilitator.
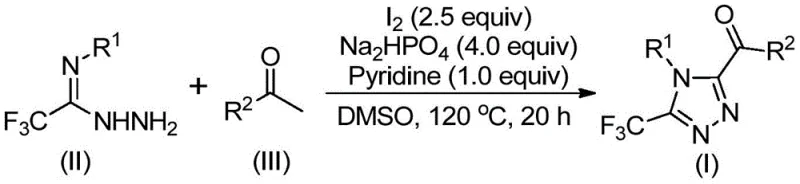
From an impurity control perspective, the absence of transition metals significantly simplifies the impurity profile of the final product. In conventional metal-catalyzed reactions, trace metal residues and metal-ligand complexes often persist through workup, requiring extensive scavenging steps that can reduce overall yield. In this iodine-promoted system, the primary byproducts are inorganic salts and organic species that are readily removed via standard aqueous workup and silica gel chromatography. The use of sodium dihydrogen phosphate and pyridine as additives further buffers the reaction environment, minimizing side reactions such as over-oxidation or polymerization. This results in a cleaner crude product, facilitating higher purity specifications and reducing the burden on analytical quality control laboratories during batch release testing.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for ease of operation, utilizing commercially available starting materials such as aryl ethyl ketones and trifluoroethylimide hydrazides. The process begins with the activation of the ketone substrate in DMSO, followed by the sequential addition of the hydrazine component and buffering agents. This one-pot strategy minimizes solvent swaps and intermediate isolation steps, which are common sources of yield loss in multi-step syntheses. For process chemists looking to implement this route, the detailed standardized synthesis steps are provided in the guide below, ensuring reproducibility and consistency across different production batches.
- Mix aryl ethyl ketone and elemental iodine in DMSO solvent and heat to 90-110°C for 4-6 hours to initiate oxidation.
- Add trifluoroethylimide hydrazide, sodium dihydrogen phosphate, pyridine, and additional iodine to the reaction mixture.
- Heat the mixture to 110-130°C for 12-20 hours to complete cyclization, then filter and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this iodine-promoted synthesis offers tangible strategic benefits beyond mere chemical elegance. The elimination of precious metal catalysts directly correlates to a reduction in raw material volatility, as the price of iodine is significantly more stable and lower than that of palladium or rhodium complexes. Additionally, the simplified workup procedure reduces the consumption of specialized scavenging resins and solvents, leading to substantial cost savings in waste management and disposal. These efficiencies allow manufacturers to offer more competitive pricing structures for high-purity pharmaceutical intermediates, enhancing their position in the global market.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with inexpensive elemental iodine drastically lowers the bill of materials for each production batch. Furthermore, the avoidance of rigorous anhydrous conditions reduces energy consumption associated with solvent drying and inert gas purging. This cumulative effect results in a leaner manufacturing process with lower overheads, enabling significant margin improvement without compromising product quality or yield.
- Enhanced Supply Chain Reliability: The starting materials, including various substituted aryl ethyl ketones and hydrazides, are commodity chemicals with robust global supply chains. This abundance ensures that production schedules are not held hostage by the scarcity of exotic reagents. Moreover, the tolerance of the reaction to ambient moisture and oxygen means that manufacturing can proceed with standard equipment, reducing the risk of unplanned downtime due to equipment failure or specialized maintenance requirements.
- Scalability and Environmental Compliance: The protocol has been demonstrated to scale effectively from laboratory to pilot scales, with consistent yields observed across different substrate classes. The use of DMSO, a high-boiling polar solvent, facilitates heat management in large reactors, while the absence of heavy metals simplifies regulatory filings and environmental impact assessments. This alignment with green chemistry principles supports corporate sustainability goals and eases the path to regulatory approval for new drug applications.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the process capabilities and limitations. These insights are derived directly from the experimental data and scope studies presented in the patent documentation, providing a realistic view of what can be achieved with this methodology in a commercial setting.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented process utilizes elemental iodine as a non-metallic promoter, completely avoiding the use of toxic heavy metal catalysts like palladium or copper, which simplifies purification and reduces regulatory burden.
Q: What are the optimal reaction conditions for scaling this process?
A: The reaction operates effectively in dimethyl sulfoxide (DMSO) at temperatures between 110°C and 130°C for the cyclization step, without requiring strict anhydrous or oxygen-free conditions, making it highly suitable for industrial scale-up.
Q: Is the substrate scope limited to specific aryl groups?
A: The method demonstrates broad functional group tolerance, successfully accommodating various substituted aryl groups including those with methyl, methoxy, chloro, and trifluoromethyl substituents at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in the timely delivery of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 3,4,5-trisubstituted 1,2,4-triazole intermediate meets the highest international standards for safety and efficacy.
We invite you to engage with our technical procurement team to discuss how this iodine-promoted synthesis can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in complex organic synthesis and pharmaceutical intermediate supply.
