Advanced Nickel-Catalyzed Cross-Coupling for Scalable Production of High-Purity Aryl Intermediates
The chemical manufacturing landscape is constantly evolving, driven by the need for safer, more efficient, and scalable synthetic routes. A pivotal advancement in this domain is documented in patent CN1289190C, which outlines a particularly advantageous process for the preparation of aryl compounds via cross-coupling reactions. This technology specifically addresses the longstanding challenges associated with nickel-catalyzed couplings of aryl halides with Grignard reagents. Traditionally, such reactions have been plagued by stringent temperature requirements and safety hazards, but this innovation introduces a novel protocol that utilizes a specialized nickel catalyst and a controlled addition strategy. By shifting the operational paradigm from cryogenic conditions to moderate or elevated temperatures, this method unlocks new possibilities for the commercial scale-up of complex pharmaceutical intermediates and fine chemicals. For industry leaders seeking a reliable pharma intermediates supplier, understanding the mechanistic and operational shifts detailed in this patent is crucial for optimizing production lines and reducing overall manufacturing costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical precedents for nickel-catalyzed cross-coupling, such as those described in Inorganica Chimica Acta, rely heavily on heterogeneous Ni/C catalysts prepared under strictly inert conditions. A critical bottleneck in these legacy processes is the requirement to add Grignard reagents at extremely low temperatures, typically around -78°C. This cryogenic necessity is not merely an inconvenience; it represents a substantial barrier to industrial implementation due to the immense energy costs and specialized equipment required to maintain such conditions. Furthermore, the thermal profile of these reactions is notoriously difficult to manage. When the reaction mixture is warmed from -78°C to room temperature or reflux, there is a significant risk of a delayed reaction onset. This delay can lead to a sudden, uncontrolled release of heat, creating severe safety hazards, particularly when handling large volumes of reactive Grignard reagents. Additionally, the catalyst precursors used in these older methods are air-sensitive, necessitating complex storage and handling protocols under argon atmospheres, which further complicates the supply chain and increases operational overhead.
The Novel Approach
In stark contrast to the cumbersome legacy techniques, the process disclosed in CN1289190C introduces a streamlined workflow that fundamentally alters the sequence of reagent addition and temperature management. The core innovation lies in pre-charging the reactor with the substituted aryl compound and the nickel catalyst, heating the mixture to the desired reaction temperature ranging from 20°C to 120°C, and only then metering in the Grignard reagent. This reversal of the addition order eliminates the need for cryogenic cooling entirely, allowing the reaction to proceed under thermally stable conditions that are easily achievable in standard industrial reactors. By maintaining the reaction at a constant elevated temperature during the addition, the process ensures immediate reaction initiation, thereby preventing the accumulation of unreacted reagents and the associated risk of thermal runaway. Moreover, the nickel catalyst employed in this novel approach is designed to be air-stable in its precursor form, removing the need for inert gas storage and significantly simplifying the logistical framework required for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Nickel-Catalyzed Cross-Coupling
The efficacy of this synthetic route is underpinned by the unique properties of the supported nickel catalyst. The catalyst is typically prepared by impregnating a high-surface-area support material, such as activated carbon, alumina, or silica, with an aqueous solution of a nickel salt like nickel(II) nitrate or chloride. Following impregnation, the material is dried and reduced, often using hydrogen or chemical reducing agents, to generate active Ni(0) species on the surface. What distinguishes this catalyst system is its robustness; the precursor materials are insensitive to oxygen, allowing them to be handled in air before being introduced into the reaction vessel. Once in the reactor, often in the presence of phosphine ligands like triphenylphosphine to enhance selectivity and rate, the nickel centers facilitate the oxidative addition of the aryl halide. This step is critical for activating the carbon-halogen bond, setting the stage for the subsequent transmetallation with the Grignard reagent. The careful selection of the support material, with surface areas ranging from 100 to 1600 m²/g, ensures high dispersion of the active metal sites, maximizing catalytic efficiency and minimizing the loading of expensive nickel metal required per batch.
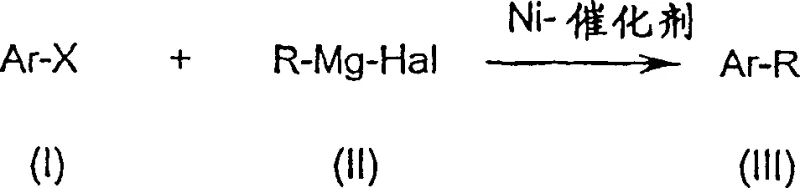
Controlling the impurity profile is paramount for any high-purity OLED material or API intermediate, and this process offers distinct advantages in that regard. The key mechanism for impurity control is the controlled concentration of the Grignard reagent within the reaction mixture. In conventional batch processes where all reagents are mixed at low temperature and then warmed, the local concentration of the Grignard reagent can fluctuate wildly, leading to homocoupling side reactions or over-alkylation. By metering the Grignard reagent slowly into the heated mixture containing the aryl halide and catalyst, the concentration of the organometallic species is kept consistently low throughout the reaction course. This kinetic control favors the desired cross-coupling pathway over competing side reactions. Furthermore, the use of specific phosphorus-containing components, such as triarylphosphines or trialkylphosphites, can further tune the electronic environment of the nickel center, enhancing the specificity of the transmetallation step and ensuring that the final aryl compound is obtained with purity levels often exceeding 95%, thus meeting the rigorous standards required for commercial scale-up of complex polymer additives and electronic chemicals.
How to Synthesize Aryl Compounds Efficiently
Implementing this synthesis strategy requires a precise adherence to the operational parameters defined in the patent to ensure both safety and yield. The process begins with the preparation or procurement of the supported nickel catalyst, which serves as the engine for the transformation. Once the catalyst is ready, the reactor setup involves charging the aryl halide substrate and the catalyst into the vessel along with a suitable solvent such as tetrahydrofuran or toluene. The mixture is then brought to the target reaction temperature, which is significantly higher than traditional methods, typically between 35°C and 100°C. Only after thermal equilibrium is reached is the Grignard reagent introduced via a metering pump or dropping funnel. This specific sequence is critical for managing the exotherm and ensuring high conversion. For a detailed breakdown of the specific quantities, timing, and workup procedures, please refer to the standardized synthesis guide below.
- Preparation of the air-stable nickel catalyst precursor by impregnating a support material like activated carbon or silica with aqueous nickel salts, followed by drying and reduction.
- Pre-charging the reactor with the substituted aryl halide, the nickel catalyst, and solvent, then heating the mixture to the target reaction temperature between 20°C and 120°C.
- Metering the Grignard reagent slowly into the heated reaction mixture to maintain low concentration, ensuring safe exotherm control and high selectivity before final workup.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel nickel-catalyzed process represents a strategic opportunity to optimize both cost structures and operational resilience. The elimination of cryogenic conditions is perhaps the most immediate driver for cost reduction in fine chemical manufacturing. Maintaining reactors at -78°C requires specialized refrigeration units and consumes vast amounts of energy, whereas operating at ambient or slightly elevated temperatures utilizes standard heating utilities that are far more economical and widely available. This shift not only lowers the direct utility costs but also reduces the capital expenditure required for plant infrastructure, as standard glass-lined or stainless steel reactors can be utilized without modification. Furthermore, the simplified catalyst handling removes the need for complex inert gas blanketing systems during storage and transfer, reducing the consumption of nitrogen or argon and minimizing the risk of supply interruptions due to gas shortages. These factors combine to create a leaner, more cost-effective production model that enhances the overall competitiveness of the supply chain.
- Cost Reduction in Manufacturing: The economic benefits of this process extend beyond simple utility savings. By utilizing air-stable catalyst precursors, the manufacturing facility avoids the costs associated with glovebox operations or specialized inert storage containers. The ability to run the reaction at higher concentrations and without the need for extreme cooling allows for smaller reactor volumes to produce the same amount of product, effectively increasing the throughput of existing assets. Additionally, the high selectivity of the reaction minimizes the formation of byproducts, which reduces the burden on downstream purification steps such as distillation or chromatography. This reduction in waste treatment and purification complexity translates directly into lower variable costs per kilogram of finished product, providing a substantial margin improvement for high-volume production runs.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of raw material handling and the fragility of sensitive reagents. The robustness of the nickel catalyst described in this patent significantly mitigates these risks. Since the catalyst precursor does not degrade upon exposure to air, it can be sourced from a wider range of suppliers and transported using standard logistics channels without the need for expedited or climate-controlled shipping. This flexibility reduces lead time for high-purity intermediates and ensures that production schedules are not derailed by minor delays in raw material delivery. Moreover, the safety profile of the reaction, characterized by controlled exotherms and the absence of cryogenic hazards, reduces the likelihood of unplanned shutdowns due to safety incidents, thereby guaranteeing a more consistent and reliable flow of goods to downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production is fraught with challenges, particularly regarding heat transfer and safety. This process is inherently scalable because the heat generation is managed by the rate of reagent addition rather than by external cooling capacity. This means that the reaction can be safely performed in large-scale reactors without the risk of thermal runaway that plagues many exothermic couplings. From an environmental perspective, the use of supported nickel catalysts allows for easier recovery and potential reuse of the metal, aligning with green chemistry principles by reducing heavy metal waste. The higher atom economy and reduced solvent usage associated with the higher concentration operation further contribute to a lower environmental footprint, facilitating compliance with increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled a set of frequently asked questions based on the technical disclosures within the patent. These queries address common concerns regarding reaction conditions, catalyst stability, and product quality. Understanding these details is essential for R&D directors who are assessing the compatibility of this route with their existing infrastructure. The answers provided are derived directly from the experimental data and descriptions found in the patent documentation, ensuring accuracy and relevance for technical decision-making.
Q: How does this nickel-catalyzed process improve safety compared to conventional methods?
A: Conventional methods often require adding Grignard reagents at cryogenic temperatures like -78°C, which poses significant thermal control risks upon warming. This patented process operates at 20-120°C and controls the exotherm by metering the reagent, eliminating the danger of delayed reaction onset and runaway heat generation.
Q: What are the storage requirements for the nickel catalyst used in this synthesis?
A: Unlike traditional heterogeneous catalysts that require inert atmosphere storage, the nickel catalyst precursors described in this patent are oxygen-insensitive when wet or dried properly. They can be stored in air without special protective gas measures, significantly simplifying logistics and handling.
Q: Can this method achieve high purity for pharmaceutical intermediates?
A: Yes, the process yields aryl compounds with purity typically above 95% and yields exceeding 85% of theory. The controlled addition of reagents minimizes side reactions and byproduct formation, making it highly suitable for producing high-purity pharmaceutical and agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN1289190C for the production of high-value aryl compounds. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this nickel-catalyzed process are fully realized in a commercial setting. Our facilities are equipped with state-of-the-art reactors capable of precise temperature control and reagent metering, which are critical for executing this synthesis safely and efficiently. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch of aryl intermediates meets the exacting standards required for pharmaceutical and electronic applications. Our commitment to quality assurance ensures that the impurity profiles are tightly controlled, delivering materials that are ready for immediate use in your downstream synthesis.
We invite you to collaborate with us to leverage this innovative chemistry for your next project. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements and target molecules. By partnering with us, you gain access to our deep expertise in nickel catalysis and process optimization. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can accelerate your development timelines and secure a robust, cost-effective supply chain for your critical aryl building blocks.
