Advanced Boric Acid Catalysis for Scalable Chlorantraniliprole Manufacturing and Commercial Supply
Advanced Boric Acid Catalysis for Scalable Chlorantraniliprole Manufacturing and Commercial Supply
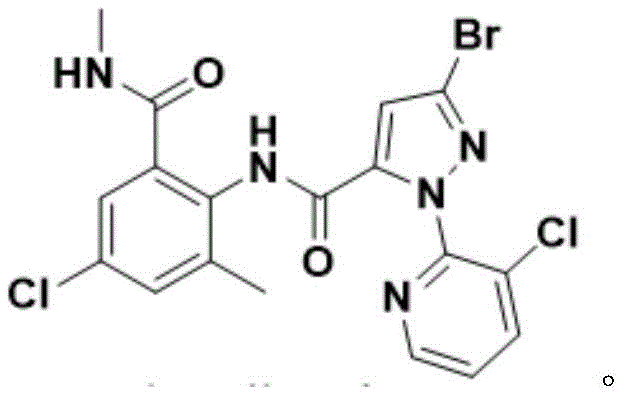
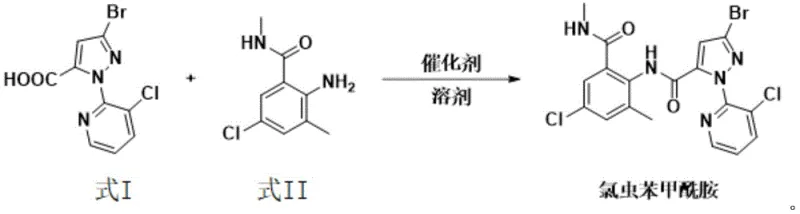
The global agrochemical sector is constantly seeking more sustainable and economically viable pathways for producing high-value insecticides, and the recent disclosure in patent CN114230555A offers a transformative approach to synthesizing chlorantraniliprole. This diamide insecticide, renowned for its efficacy against lepidoptera and other pests, has traditionally been manufactured through processes fraught with safety hazards and complex solvent recovery challenges. The new methodology leverages boric acid substances as condensation catalysts, facilitating a direct amidation reaction between 3-bromo-1-(3-chloro-2-pyridyl)-1H-pyrazole-5-carboxylic acid and 2-amino-5-chloro-N,3-dimethylbenzamide. By shifting away from toxic activating agents and water-miscible solvents, this innovation addresses critical pain points in industrial hygiene and waste management. For procurement and supply chain leaders, this represents a significant opportunity to secure a more reliable agrochemical intermediate supplier capable of delivering consistent quality with reduced environmental liability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of chlorantraniliprole has relied heavily on activation strategies involving methanesulfonyl chloride, a reagent known for its high toxicity and stringent transportation controls which pose severe risks to production personnel and facility safety. Furthermore, conventional protocols typically employ 3-methylpyridine as a base and acetonitrile as the reaction solvent, creating a nightmare scenario for downstream processing due to the mutual solubility of these components with water. Recovering acetonitrile from aqueous waste streams often necessitates complex rectification procedures, sometimes requiring double distillation or negative pressure operations, which drastically increases energy consumption and capital equipment investment. These inefficiencies not only inflate the cost of goods sold but also generate substantial three-waste discharge, complicating environmental compliance and sustainability reporting for large-scale manufacturers.
The Novel Approach
In stark contrast, the novel process detailed in the patent utilizes a boric acid catalyst system within a hydrophobic solvent environment, fundamentally altering the thermodynamics and kinetics of the amidation step. By employing solvents such as toluene, xylene, or chlorobenzene, which have boiling points higher than water, the reaction can be driven to completion through the continuous removal of water using a simple water separator or Dean-Stark apparatus. This eliminates the need for toxic activation reagents entirely and simplifies the workup procedure to basic filtration and washing steps. The reaction equation illustrates this elegant transformation where the carboxylic acid and amine couple directly under thermal conditions, bypassing the formation of hazardous intermediates.
Mechanistic Insights into Boric Acid-Catalyzed Amidation
The core of this technological breakthrough lies in the unique ability of boric acid and its derivatives, such as 3-nitrophenylboronic acid or 3,4,5-trifluorophenylboronic acid, to activate the carboxylic acid moiety towards nucleophilic attack by the amine. Unlike traditional coupling agents that form reactive esters or acid chlorides, boron species likely coordinate with the carbonyl oxygen, increasing the electrophilicity of the carbonyl carbon while simultaneously facilitating proton transfer events necessary for water elimination. This mechanism operates efficiently at elevated temperatures, typically 10-20°C above the boiling point of the chosen hydrophobic solvent, ensuring rapid kinetics without the degradation of sensitive functional groups. The use of a slight molar excess of the amine component (1:1.01 to 1:1.05 ratio) further drives the equilibrium towards the product side, ensuring high conversion rates essential for commercial viability.
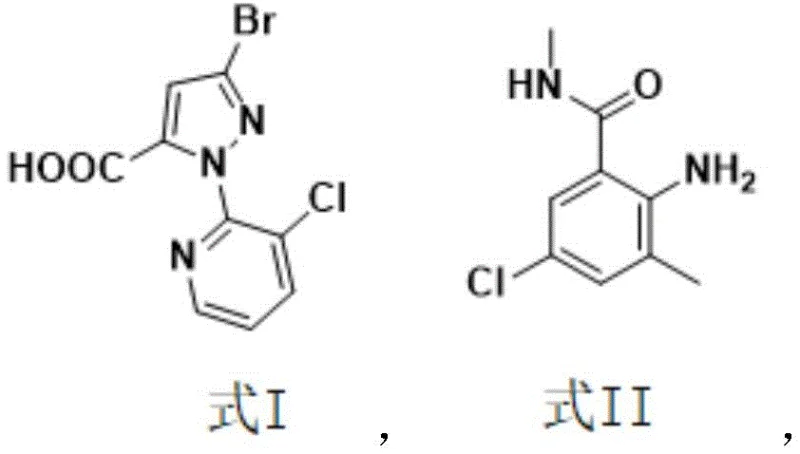
From an impurity control perspective, the boric acid catalyzed route offers superior selectivity, minimizing the formation of side products that often plague harsh activation methods. The process specifies stopping the reaction only when the content of the starting carboxylic acid (Formula I) drops below 0.5%, a rigorous standard that guarantees minimal residual starting material in the crude product. This high level of conversion reduces the load on subsequent purification steps, such as the methanol pulping described in the post-treatment phase, resulting in a final product with excellent purity profiles. For R&D directors, understanding this mechanistic nuance is crucial for scaling the process, as it highlights the importance of precise temperature control and water removal efficiency in maintaining reaction fidelity and minimizing impurity generation.
How to Synthesize Chlorantraniliprole Efficiently
The synthesis protocol outlined in the patent provides a robust framework for manufacturing chlorantraniliprole that balances reaction efficiency with operational simplicity. The procedure involves charging a reactor with the hydrophobic solvent, the pyrazole carboxylic acid, the benzamide amine, and the boric acid catalyst, followed by heating to reflux with continuous water separation. Detailed standardized synthetic steps see the guide below.
- Charge a reactor with hydrophobic solvent (toluene, xylene, or chlorobenzene), 3-bromo-1-(3-chloro-2-pyridyl)-1H-pyrazole-5-carboxylic acid, 2-amino-5-chloro-N,3-dimethylbenzamide, and a boric acid catalyst.
- Heat the mixture to 120-150°C using a Dean-Stark apparatus to continuously remove generated water while maintaining reaction temperature above the solvent boiling point.
- Monitor reaction progress until starting acid content is below 0.5%, then cool, filter, wash with solvent and water, pulp with methanol, and dry to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this boric acid catalyzed process translates into tangible strategic benefits that extend far beyond simple yield improvements. The elimination of methanesulfonyl chloride removes a major bottleneck related to hazardous material handling, storage, and regulatory compliance, thereby smoothing out logistics and reducing insurance and safety overheads. Additionally, the switch to hydrophobic solvents like toluene or xylene allows for straightforward solvent recovery through phase separation and simple distillation, avoiding the energy-intensive azeotropic breaking required for acetonitrile-water mixtures. This fundamental shift in process chemistry leads to substantial cost savings in utility consumption and waste treatment, making the overall manufacturing economics significantly more attractive.
- Cost Reduction in Manufacturing: The removal of expensive and toxic activating agents like methanesulfonyl chloride directly lowers raw material costs while simultaneously reducing the need for specialized corrosion-resistant equipment. The simplified solvent recovery system, which relies on gravity separation of water rather than complex fractional distillation trains, drastically cuts down on energy usage and maintenance requirements for recovery units. Furthermore, the high conversion rates achieved minimize the loss of valuable starting materials, ensuring that the input costs are maximized in terms of final product output, leading to a more competitive cost structure for the final agrochemical intermediate.
- Enhanced Supply Chain Reliability: By avoiding strictly controlled toxic reagents, the supply chain becomes more resilient to regulatory changes and transportation disruptions that often affect hazardous chemicals. The raw materials required, including boric acid derivatives and common hydrophobic solvents, are commodity chemicals with stable global availability, reducing the risk of supply shortages. This stability ensures consistent production schedules and reliable delivery timelines for downstream formulators, strengthening the partnership between the manufacturer and their clients in the global crop protection market.
- Scalability and Environmental Compliance: The process generates significantly less three-waste discharge compared to traditional methods, aligning perfectly with increasingly stringent global environmental regulations and corporate sustainability goals. The simplicity of the workup procedure, involving cooling, filtering, and washing, makes the process highly scalable from pilot plant to multi-ton commercial production without encountering the engineering complexities associated with handling toxic gases or difficult solvent separations. This ease of scale-up ensures that supply can be rapidly ramped up to meet market demand without compromising on environmental standards or operational safety.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route, derived directly from the patent specifications and experimental data provided. These insights are intended to clarify the operational parameters and benefits for stakeholders evaluating this technology for potential adoption or sourcing.
Q: What are the primary advantages of the boric acid catalyzed route over traditional methanesulfonyl chloride activation?
A: The boric acid catalyzed route eliminates the use of methanesulfonyl chloride, a highly toxic and strictly controlled reagent, significantly improving operational safety and reducing regulatory burdens. Furthermore, it replaces the difficult-to-recover acetonitrile and 3-methylpyridine system with hydrophobic solvents like toluene or xylene, allowing for simpler water separation and solvent recycling.
Q: How does the new process impact solvent recovery costs in chlorantraniliprole production?
A: By utilizing hydrophobic solvents such as toluene or xylene which form azeotropes with water, the process enables efficient water removal via a divider (Dean-Stark trap) without complex distillation trains. This contrasts sharply with prior art using acetonitrile, which is miscible with water and requires energy-intensive double rectification or negative pressure distillation for recovery.
Q: What represents the critical quality control parameter during the reaction phase?
A: The critical quality control parameter is the residual content of the starting carboxylic acid (Formula I), which must be controlled to be lower than 0.5% before stopping the reaction. This ensures high conversion rates and minimizes the burden on downstream purification steps, directly contributing to the final purity specifications required for agrochemical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chlorantraniliprole Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this boric acid catalyzed pathway to redefine the production standards for chlorantraniliprole and similar diamide insecticides. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into robust industrial realities. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of agrochemical intermediate meets the exacting requirements of global regulatory bodies and end-users.
We invite forward-thinking partners to engage with our technical procurement team to discuss how this advanced manufacturing route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, securing a competitive edge in the dynamic agrochemical marketplace through superior technology and reliable supply.
