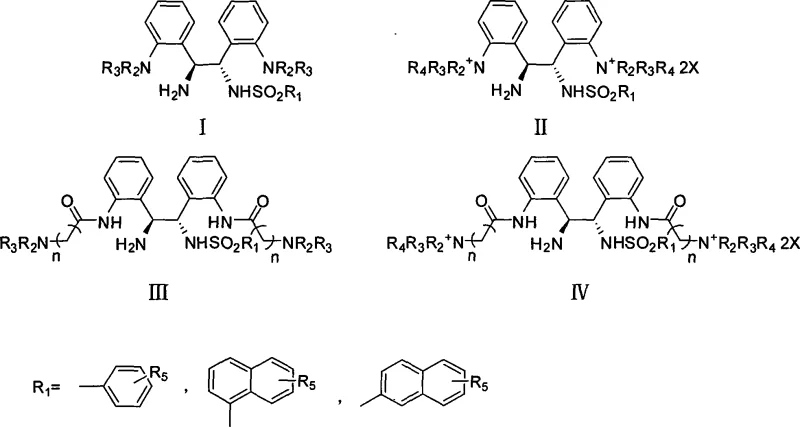
Advanced Aqueous Phase Asymmetric Catalysis with Novel Water-Soluble Chiral Diamine Ligands
Advanced Aqueous Phase Asymmetric Catalysis with Novel Water-Soluble Chiral Diamine Ligands
The landscape of green chemistry and sustainable pharmaceutical manufacturing is undergoing a significant transformation, driven by the urgent need to replace hazardous organic solvents with benign alternatives like water. Patent CN101074207A introduces a groundbreaking class of water-soluble chiral diamine derivatives, specifically optically pure N-monosulfonated acylation-1,2-bis(2'-aminophenyl)-1,2-ethylenediamine and its salts. These novel ligands represent a paradigm shift in asymmetric catalysis, enabling highly efficient transfer hydrogenation reactions directly in aqueous phases without the need for additional organic co-solvents or external surfactants. For R&D directors and process chemists, this technology offers a robust pathway to achieve high enantioselectivity while adhering to stringent environmental regulations. The core innovation lies in the molecular design that imparts intrinsic amphiphilicity to the catalyst, allowing it to function as a self-assembling nano-reactor in water.
From a supply chain and procurement perspective, the adoption of these water-soluble ligands addresses critical pain points associated with solvent recovery and waste disposal in API manufacturing. Traditional asymmetric hydrogenation processes often rely on volatile organic compounds (VOCs) that require complex distillation and treatment systems, inflating operational expenditures. By transitioning to an aqueous system, manufacturers can drastically simplify downstream processing, as the products can often be separated via simple extraction or filtration. This patent data underscores the viability of scaling these reactions from laboratory benchtop to industrial production, offering a reliable pharmaceutical intermediate supplier with the capability to deliver high-purity chiral building blocks with a significantly reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, asymmetric transfer hydrogenation has been predominantly conducted in organic solvents such as alcohols or dichloromethane, which pose significant safety and environmental hazards. While early attempts to move these reactions into aqueous media were made, they often suffered from poor solubility of the organic substrates, leading to sluggish reaction rates and low yields. For instance, previous methodologies described by Williams utilized alcohol-water two-phase systems, which failed to achieve a true single-phase aqueous reaction, limiting the efficiency of mass transfer. Furthermore, other approaches, such as those by Deng, required the addition of exogenous surfactants like SDS to solubilize the reactants. These surfactants not only add to the material cost but can also complicate product purification and potentially contaminate the final API, requiring rigorous removal steps that extend production lead times.
The Novel Approach
The technology disclosed in CN101074207A overcomes these barriers by integrating the solubilizing group directly into the chiral ligand structure. By introducing sulfonic acid groups or quaternary ammonium salts onto the diamine backbone, the resulting complexes become inherently water-soluble and amphiphilic. This structural modification allows the catalyst to spontaneously form stable micelles or capsules in water, creating a hydrophobic microenvironment that concentrates the organic substrate near the catalytic center. This "nano-reactor" effect dramatically enhances reaction kinetics and stereoselectivity, often surpassing the performance of traditional organic phase reactions. The result is a streamlined process that operates in pure water, eliminates the need for added surfactants, and achieves conversion rates and enantiomeric excess values comparable to or better than conventional methods, representing a major leap forward in cost reduction in API manufacturing.
Mechanistic Insights into Amphiphilic Catalyst Self-Assembly
The exceptional performance of these water-soluble chiral diamines stems from their unique ability to act as amphiphilic molecules that self-assemble in aqueous solutions. When the metal complex forms, the hydrophobic aromatic backbone of the ligand aggregates to minimize contact with water, while the hydrophilic sulfonate or quaternary ammonium groups interact with the solvent. This behavior leads to the formation of micellar structures that encapsulate the hydrophobic ketone or imine substrates within their core. This localized high concentration of substrate within the micelle significantly increases the frequency of collisions with the catalytic metal center, thereby accelerating the reaction rate. Moreover, the rigid chiral environment provided by the diamine backbone ensures that the hydride transfer occurs with high stereocontrol, consistently delivering products with enantiomeric excess values exceeding 95% and often reaching 98%.
Furthermore, the stability of these micellar assemblies contributes to the robustness of the catalytic system. Unlike small molecule surfactants that may form unstable emulsions, the catalyst-derived micelles provide a defined microenvironment that protects the active metal species from deactivation. This stability is crucial for maintaining catalytic turnover numbers over extended periods, which is a key factor for commercial viability. The patent data highlights that varying the length of the alkyl chains on the quaternary ammonium groups can fine-tune the micelle size and hydrophobicity, allowing for optimization based on specific substrate properties. This level of tunability provides process chemists with a versatile toolkit for adapting the catalytic system to a wide range of challenging transformations, ensuring high-purity chiral ligands are utilized effectively across diverse synthetic routes.
How to Synthesize Water-Soluble Chiral Diamine Efficiently
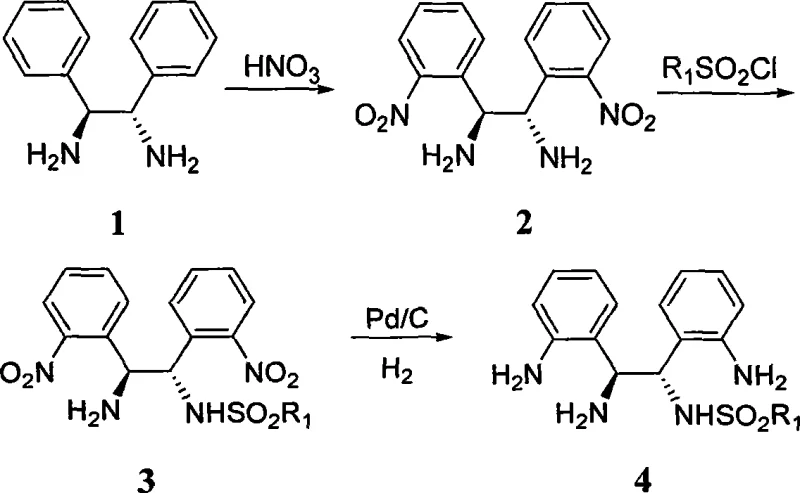
The preparation of these advanced ligands follows a logical and scalable synthetic sequence that begins with readily available optically pure 1,2-diphenylethylenediamine. The process involves a strategic nitration step to activate the aromatic rings, followed by a selective mono-sulfonylation to introduce the water-solubilizing moiety. The final reduction of the nitro groups yields the target diamine, which can be further derivatized to create quaternary ammonium salts or acylated derivatives. This modular approach allows for the generation of a library of ligands with varying solubility profiles and steric properties, catering to specific reaction requirements. The synthetic route is designed to be robust, avoiding sensitive reagents and extreme conditions that would hinder large-scale production.
- Nitration of optically pure 1,2-diphenylethylenediamine using concentrated nitric acid to obtain the dinitro intermediate.
- Selective mono-sulfonylation of the dinitro intermediate with sulfonyl chloride (R1SO2Cl) to introduce the water-solubilizing group.
- Catalytic hydrogenation using Pd/C to reduce the nitro groups to amino groups, yielding the final water-soluble chiral diamine ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this aqueous catalytic technology offers substantial strategic benefits beyond mere technical performance. The elimination of bulk organic solvents translates directly into reduced raw material costs and lower expenses associated with solvent recovery and waste treatment infrastructure. Since water is the primary medium, the safety profile of the manufacturing process is significantly enhanced, reducing the risk of fire and explosion hazards associated with flammable organic solvents. This improvement in safety can lead to lower insurance premiums and less regulatory burden, facilitating smoother operations in heavily regulated jurisdictions. Additionally, the simplified work-up procedures, often requiring only extraction or filtration, shorten the overall batch cycle time, thereby increasing plant throughput and asset utilization.
- Cost Reduction in Manufacturing: The inherent water solubility of the catalyst eliminates the need for purchasing and handling expensive organic solvents and external surfactants. By removing the requirement for complex solvent distillation and recycling units, capital expenditure for new facilities can be minimized, and operating costs for existing plants can be drastically lowered. The high catalytic efficiency means lower catalyst loading is required to achieve full conversion, further reducing the cost per kilogram of the final chiral intermediate. These factors combine to create a leaner, more cost-effective manufacturing process that improves overall profit margins.
- Enhanced Supply Chain Reliability: The starting materials for these ligands, such as diphenylethylenediamine and common sulfonyl chlorides, are commercially available and stable, ensuring a secure supply chain. The robustness of the aqueous reaction conditions means that production is less susceptible to disruptions caused by solvent quality variations or humidity issues that often plague organic synthesis. Furthermore, the ability to perform reactions at moderate temperatures reduces energy consumption, making the supply of these intermediates more resilient to fluctuations in energy prices. This reliability is critical for maintaining continuous production schedules for downstream API manufacturing.
- Scalability and Environmental Compliance: Scaling up aqueous reactions is generally safer and more straightforward than scaling organic processes, as heat transfer is more efficient in water and the risk of thermal runaway is mitigated. The technology aligns perfectly with Green Chemistry principles, significantly reducing the E-factor (mass of waste per mass of product) by minimizing organic waste generation. This environmental compliance is increasingly important for meeting corporate sustainability goals and satisfying the demands of eco-conscious stakeholders. The process facilitates the commercial scale-up of complex catalytic systems without the environmental liabilities associated with traditional methods.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this water-soluble catalytic technology. Understanding these details is essential for process development teams evaluating the feasibility of adopting this methodology for their specific pipeline compounds. The answers are derived directly from the experimental data and claims within the patent literature, ensuring accuracy and relevance for industrial application.
Q: What are the advantages of using water-soluble chiral diamines over traditional organic phase ligands?
A: Water-soluble chiral diamines allow reactions to proceed in pure aqueous media, eliminating the need for toxic organic solvents and simplifying product separation. They often exhibit amphiphilic properties that form micelles, enhancing reaction rates and enantioselectivity without added surfactants.
Q: Which metals are compatible with these novel ligands for asymmetric hydrogenation?
A: These ligands form active complexes with Group 8 transition metals, specifically Ruthenium (Ru), Rhodium (Rh), and Iridium (Ir). Rhodium complexes have shown particularly high activity and enantioselectivity (up to 98% ee) in aqueous phase transfer hydrogenation.
Q: Can these ligands be used for the synthesis of beta-adrenoceptor antagonists?
A: Yes, the technology is highly effective for the asymmetric transfer hydrogenation of omega-haloacetophenones, which are key precursors for beta-blockers like Terbutaline and Salbutamol, achieving high yields and enantiomeric excess in water.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Water-Soluble Chiral Diamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of aqueous phase asymmetric catalysis in modern drug discovery and development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale innovation to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise enantiomeric excess measurements essential for chiral pharmaceutical intermediates. We are committed to leveraging this advanced patent technology to deliver high-quality, cost-effective solutions that meet the evolving needs of the global pharmaceutical industry.
We invite you to collaborate with our technical procurement team to explore how these water-soluble ligands can optimize your current synthetic routes. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into the potential economic and environmental benefits for your specific projects. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us help you engineer a more sustainable and efficient supply chain for your critical chiral building blocks.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
