Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
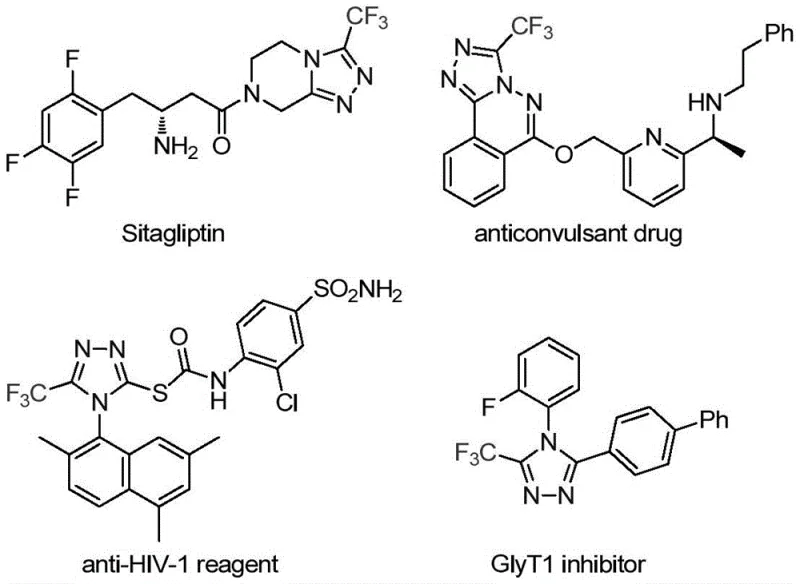
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are critical for enhancing metabolic stability and bioavailability. Patent CN113307778A introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, utilizing a synergistic molybdenum and copper co-catalytic system. This technology addresses the long-standing challenges associated with traditional triazole synthesis, such as harsh reaction conditions and limited substrate scope. By leveraging cheap and easily obtainable starting materials like trifluoroethylimidoyl chloride and functionalized isonitriles, this process offers a streamlined pathway to high-value intermediates used in blockbuster drugs like Sitagliptin and various agrochemical agents. For R&D directors and procurement managers alike, this represents a significant opportunity to optimize supply chains and reduce manufacturing costs through a more efficient, one-pot synthetic strategy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied on methodologies that pose significant safety and operational hurdles for large-scale manufacturing. Traditional routes often involve the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones, processes that frequently require hazardous hydrazine derivatives and stringent temperature controls. Furthermore, copper-catalyzed multi-component reactions utilizing diazonium salts and trifluorodiazoethane, while effective in academic settings, introduce substantial risks due to the explosive nature of diazo compounds and the complexity of handling gaseous reagents. These conventional approaches often suffer from poor atom economy, generate significant waste streams, and exhibit limited tolerance for sensitive functional groups, thereby restricting their utility in the late-stage functionalization of complex pharmaceutical intermediates.
The Novel Approach
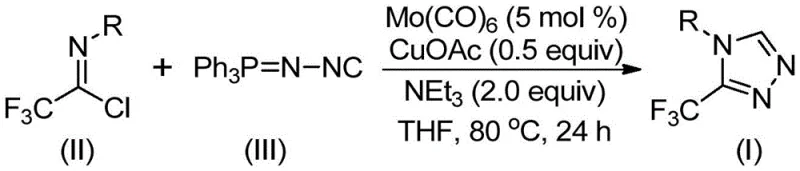
In stark contrast, the methodology disclosed in patent CN113307778A utilizes a sophisticated yet operationally simple co-catalytic system involving molybdenum hexacarbonyl and cuprous acetate to drive the cycloaddition of trifluoroethylimidoyl chloride with functionalized isonitriles. This novel approach eliminates the need for dangerous diazonium salts or unstable hydrazines, instead relying on stable, commercially available liquid or solid reagents that can be handled with standard laboratory equipment. The reaction proceeds under mild thermal conditions, typically between 70°C and 90°C, in common aprotic solvents like THF, which facilitates easier solvent recovery and recycling. By shifting the mechanistic paradigm to a metal-promoted [3+2] cycloaddition followed by triphenylphosphine oxide elimination, this method achieves high reaction efficiency and excellent yields across a broad range of substrates, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming to modernize their production capabilities.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The success of this synthetic route lies in the intricate interplay between the molybdenum and copper catalysts, which orchestrate the activation of the isonitrile and the subsequent ring closure. Mechanistically, the molybdenum hexacarbonyl serves as a crucial metal activator that coordinates with the functionalized isonitrile (Ph3P=N-NC), increasing its nucleophilicity and facilitating its attack on the electrophilic carbon of the trifluoroethylimidoyl chloride. Simultaneously, the cuprous acetate acts as a promoter for the [3+2] cycloaddition step, stabilizing the transition state and ensuring the formation of the five-membered triazole ring intermediate with high regioselectivity. The presence of triethylamine as a base is essential for neutralizing the hydrochloric acid byproduct generated during the initial addition, driving the equilibrium forward and preventing catalyst deactivation. This dual-catalyst system ensures that the reaction proceeds smoothly even with sterically hindered or electronically diverse substrates, providing a level of control that single-metal catalysts often fail to achieve in complex heterocyclic synthesis.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions commonly associated with radical pathways or uncontrolled hydrazine condensations. The specific coordination environment created by the Mo/Cu system directs the reaction exclusively towards the desired 1,2,4-triazole scaffold, significantly reducing the formation of regioisomers or polymeric byproducts. Furthermore, the final step involves the elimination of triphenylphosphine oxide, a solid byproduct that can be easily removed during the aqueous workup or silica gel filtration, simplifying the purification process. For quality control teams, this means a cleaner crude reaction profile and higher purity specifications for the final API intermediate, reducing the burden on downstream crystallization steps and ensuring consistent batch-to-batch quality essential for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
To implement this cutting-edge synthesis in a pilot or production setting, operators must adhere to precise stoichiometric ratios and reaction parameters to maximize yield and minimize waste. The process begins with the careful selection of the organic solvent, with tetrahydrofuran (THF) being the preferred medium due to its ability to effectively dissolve both the metal catalysts and the organic substrates while promoting high conversion rates. The molar ratio of reactants is critical, with a slight excess of the functionalized isonitrile (1.5 equivalents) relative to the trifluoroethylimidoyl chloride (1.0 equivalent) recommended to ensure complete consumption of the limiting reagent. Catalyst loading is optimized at 5 mol % for molybdenum hexacarbonyl and 0.5 equivalents for cuprous acetate, balancing cost efficiency with reaction kinetics. Detailed standardized synthetic steps for this procedure are provided in the guide below.
- Combine molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieves in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride (Formula II) and functionalized isonitrile (Formula III, Ph3P=N-NC) to the reaction mixture under inert atmosphere.
- Heat the reaction mixture to 70-90°C for 18-30 hours, then filter and purify via column chromatography to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Mo/Cu co-catalyzed methodology translates into tangible strategic benefits regarding cost structure and supply reliability. The shift away from hazardous, specialty reagents like diazonium salts towards commodity chemicals such as trifluoroethylimidoyl chloride and triethylamine drastically simplifies the sourcing landscape. These starting materials are widely available from multiple global vendors, mitigating the risk of supply chain disruptions caused by the monopoly of a single supplier or regulatory restrictions on controlled substances. Furthermore, the use of earth-abundant copper salts alongside catalytic amounts of molybdenum ensures that the raw material costs remain low and stable, insulating the production budget from the volatility often seen with precious metal catalysts like palladium or rhodium.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the elimination of expensive and dangerous reagents, coupled with a simplified workup procedure that reduces solvent consumption and waste disposal costs. By avoiding the need for cryogenic conditions or high-pressure equipment required by alternative methods, the capital expenditure for reactor infrastructure is significantly lowered. The high reaction efficiency and yield reported in the patent data mean that less raw material is wasted per kilogram of product, directly improving the overall cost of goods sold (COGS) for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route against variations in substrate electronics ensures consistent production output even when switching between different batches of starting materials. Since the reagents are stable and shelf-stable, inventory management becomes more predictable, allowing for just-in-time manufacturing strategies without the fear of reagent degradation. This reliability is crucial for maintaining continuous supply lines to downstream API manufacturers, preventing costly production stoppages and ensuring that delivery commitments to global pharmaceutical partners are met without delay.
- Scalability and Environmental Compliance: The mild reaction conditions (70-90°C) and the use of THF, a solvent with well-established recovery protocols, make this process highly amenable to scale-up from gram to multi-ton quantities. The absence of heavy metal waste streams associated with stoichiometric oxidants or toxic hydrazine byproducts simplifies environmental compliance and wastewater treatment. This green chemistry profile not only reduces the environmental footprint of the manufacturing site but also aligns with the increasingly stringent sustainability criteria demanded by major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazole synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on reaction scope, safety, and scalability. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing production workflows.
Q: What are the key advantages of the Mo/Cu co-catalyzed method over traditional triazole synthesis?
A: Unlike traditional methods requiring harsh hydrazinolysis or dangerous diazonium salts, this patent describes a mild one-pot cycloaddition at 70-90°C using commercially available trifluoroethylimidoyl chloride and functionalized isonitriles, significantly improving safety and operational simplicity.
Q: Can this synthesis method be scaled for industrial production of API intermediates?
A: Yes, the patent explicitly demonstrates that the reaction conditions are mild and the operation is simple, allowing for easy expansion to gram-level reactions and providing a solid foundation for subsequent scale production and application in pharmaceutical manufacturing.
Q: What is the substrate scope for the R group in this triazole synthesis?
A: The method exhibits wide functional group tolerance, successfully accommodating R groups such as phenethyl, substituted phenyl (with methyl, methoxy, fluoro, chloro, nitro, or t-butyl substituents), and naphthyl groups, yielding products with high efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Mo/Cu co-catalyzed synthesis described in patent CN113307778A for the production of high-purity pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole delivered meets the exacting standards required for global drug registration and clinical trials.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced synthetic technology for their next-generation drug candidates. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is built on a foundation of innovation, reliability, and cost-effectiveness.
