Scalable UV-Induced Synthesis of 2-Aminobenzamide Intermediates for Commercial Production
Scalable UV-Induced Synthesis of 2-Aminobenzamide Intermediates for Commercial Production
Introduction to Advanced Photochemical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, sustainable, and cost-effective pathways to synthesize critical building blocks. A significant breakthrough in this domain is documented in patent CN114276203A, which introduces a novel ultraviolet light-induced method for synthesizing 2-aminobenzamide compounds. This technology represents a paradigm shift from traditional thermal or metal-catalyzed processes, offering a green chemistry alternative that aligns with modern environmental standards and efficiency demands. By leveraging photochemical energy at room temperature, this method bypasses the need for harsh reaction conditions, thereby reducing energy consumption and operational complexity. For R&D directors and procurement specialists, understanding the implications of this patent is crucial for optimizing supply chains and enhancing product purity profiles. The ability to produce high-purity pharmaceutical intermediates without heavy metal residues addresses a major pain point in downstream processing, particularly for active pharmaceutical ingredients (APIs) intended for human consumption. This report analyzes the technical merits and commercial viability of this innovation, providing actionable insights for stakeholders aiming to secure a competitive advantage in the global chemical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-aminobenzamide compounds has relied on a variety of conventional methodologies, each carrying significant drawbacks that hinder large-scale efficiency. Traditional routes often involve the ring-opening of isatoic anhydride, amination of anthranilic acid, or palladium-catalyzed carbonylation reactions. These methods frequently demand stringent control over water and oxygen content, requiring inert atmospheres and specialized equipment that drive up capital expenditure. Furthermore, the reliance on transition metal catalysts introduces the risk of heavy metal contamination, necessitating costly and time-consuming purification steps to meet regulatory purity specifications. The post-treatment processes associated with removing these metal residues not only increase waste generation but also reduce the overall atom economy of the synthesis. Additionally, many conventional pathways require elevated temperatures or pressures, posing safety risks and increasing energy costs. For supply chain managers, these complexities translate into longer lead times and higher vulnerability to disruptions, as the availability of specialized catalysts and the capacity for rigorous purification can become bottlenecks in production schedules.
The Novel Approach
In stark contrast to these legacy methods, the technology outlined in patent CN114276203A offers a streamlined, one-step solution that fundamentally simplifies the manufacturing landscape. This novel approach utilizes ultraviolet light to induce a simultaneous dehydrogenation N-acylation and nitro reduction reaction between 2-nitrobenzaldehyde compounds and primary amines. Operating effectively at room temperature, this photochemical process eliminates the need for external heating sources, resulting in substantial energy savings and a reduced carbon footprint. The reaction proceeds in a mixed solvent system of ethyl acetate and acetone, promoted by acetic acid, without the necessity for transition metals or strong bases. This metal-free nature is a critical advantage, as it inherently prevents heavy metal contamination, thereby simplifying downstream purification and ensuring higher product quality. The simplicity of the operation allows for easier scale-up, making it an attractive option for industrial applications where consistency and safety are paramount. By adopting this method, manufacturers can achieve yields as high as 92% while significantly reducing the environmental impact associated with chemical waste disposal.
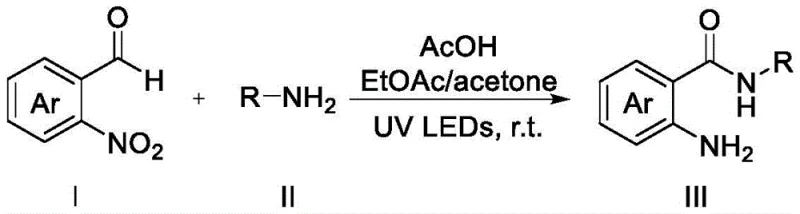
Mechanistic Insights into UV-Induced Cyclization and Reduction
Understanding the underlying mechanism of this photochemical transformation is essential for R&D teams aiming to optimize process parameters and ensure reproducibility. The reaction initiates when the 2-nitrobenzaldehyde substrate absorbs ultraviolet light, triggering a rearrangement to form a 2-nitrosobenzoic acid intermediate. This photo-excited state is highly reactive and, in the presence of acetic acid, creates an acidic environment that facilitates nucleophilic attack by the primary amine. The carboxyl group of the intermediate reacts with the amine to form an amide bond, while the nitroso group undergoes a series of transformations involving nucleophilic addition and elimination of an imine intermediate to yield a hydroxylamine species. Under the specific conditions of this system, the hydroxylamine group is subsequently reduced to an amino group, completing the formation of the target 2-aminobenzamide compound. This intricate cascade occurs seamlessly in a single pot, demonstrating the elegance of photochemical synthesis in constructing complex molecular architectures. The tolerance of this mechanism to various functional groups allows for a wide substrate scope, enabling the synthesis of diverse derivatives without compromising yield or selectivity.
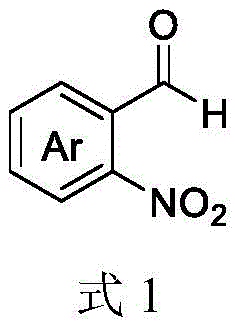
The versatility of this synthetic route is further evidenced by its compatibility with a broad range of substituents on both the aldehyde and amine components. The aromatic ring of the 2-nitrobenzaldehyde can accommodate various electron-donating or electron-withdrawing groups, such as halogens, alkyl chains, or alkoxy groups, without significantly impeding the reaction progress. Similarly, the primary amine component can vary from simple aliphatic chains to complex heterocyclic systems, including amino acid esters and benzyl derivatives. This structural flexibility is crucial for medicinal chemists who require access to diverse libraries of compounds for structure-activity relationship (SAR) studies. The mechanism ensures that even sterically hindered substrates can be converted efficiently, provided the optimal solvent ratio and light intensity are maintained. By controlling the molar ratios, specifically keeping the primary amine in excess, the reaction equilibrium is driven towards the product, minimizing the formation of side products and ensuring a clean impurity profile. This level of control is vital for maintaining the high purity standards required in the production of reliable pharmaceutical intermediates supplier networks.
How to Synthesize 2-Aminobenzamide Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific procedural guidelines to maximize efficiency and safety. The process begins with the precise preparation of the reaction mixture, where the stoichiometry of reagents plays a pivotal role in determining the final outcome. Operators must ensure that the solvent system consists of ethyl acetate and acetone in a specific ratio, typically around 6:1, as deviations can lead to diminished yields or incomplete conversions. The addition of acetic acid is not merely catalytic but serves to modulate the acidity of the medium, which is critical for the stability of the intermediates formed during the photochemical excitation. Once the mixture is prepared, it is subjected to irradiation using UV LEDs with a wavelength range of 365-375nm, which matches the absorption profile of the nitrobenzaldehyde substrate. Monitoring the reaction progress via techniques such as gas chromatography allows for real-time adjustments and ensures that the reaction is terminated at the optimal point to prevent degradation. Detailed standardized synthesis steps are provided below to guide technical teams through the execution of this protocol.
- Prepare the reaction mixture by combining 2-nitrobenzaldehyde compounds and primary amines in a mixed solvent of ethyl acetate and acetone with acetic acid.
- Maintain the reaction environment at room temperature while ensuring the molar ratio of primary amine is 2 to 4 times that of the aldehyde substrate.
- Irradiate the solution with 365-375nm UV LEDs for approximately 16 hours to complete the dehydrogenation N-acylation and nitro reduction simultaneously.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this UV-induced synthesis method offers compelling advantages that directly impact the bottom line and operational resilience. For procurement managers, the elimination of transition metal catalysts represents a significant opportunity for cost reduction in fine chemical manufacturing. Precious metals like palladium are not only expensive but also subject to volatile market pricing and supply constraints; removing them from the bill of materials stabilizes costs and reduces dependency on scarce resources. Furthermore, the absence of heavy metals simplifies the purification workflow, reducing the consumption of solvents and adsorbents typically used for metal scavenging. This streamlining of the post-reaction process translates into shorter cycle times and lower labor costs, enhancing overall throughput. For supply chain heads, the robustness of this method ensures enhanced supply chain reliability, as the raw materials involved are commodity chemicals that are readily available from multiple vendors. The mild reaction conditions also reduce the risk of safety incidents, lowering insurance premiums and compliance burdens associated with hazardous operations.
- Cost Reduction in Manufacturing: The economic benefits of this technology are driven primarily by the simplification of the process workflow and the removal of costly reagents. By avoiding the use of transition metals and specialized ligands, manufacturers can achieve substantial cost savings on raw material procurement. Additionally, the energy efficiency of running reactions at room temperature compared to high-temperature thermal processes results in lower utility bills over the lifespan of production campaigns. The reduction in waste generation, particularly hazardous metal-containing waste, lowers disposal fees and environmental compliance costs. These cumulative savings allow companies to offer more competitive pricing for high-purity pharmaceutical intermediates while maintaining healthy profit margins. The qualitative improvement in process efficiency means that resources can be reallocated to other areas of innovation and development.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the scarcity of specialized catalysts or the complexity of multi-step syntheses. This photochemical method mitigates those risks by utilizing widely available starting materials and standard laboratory equipment. The simplicity of the setup means that production can be easily transferred between facilities or scaled up without requiring bespoke infrastructure investments. This flexibility is crucial for responding to sudden spikes in demand or mitigating disruptions caused by geopolitical events affecting specific chemical supply lines. By reducing lead time for high-purity pharmaceutical intermediates, companies can improve their service levels to downstream customers, fostering stronger long-term partnerships. The reliability of the process ensures consistent quality output, which is essential for maintaining certification and trust in regulated markets.
- Scalability and Environmental Compliance: As regulatory pressure mounts on the chemical industry to adopt greener practices, this technology positions manufacturers at the forefront of sustainability. The commercial scale-up of complex pharmaceutical intermediates is often hindered by environmental constraints, but this method's low waste profile and energy efficiency facilitate smoother regulatory approvals. The absence of toxic heavy metals simplifies the handling of effluents and reduces the environmental footprint of the manufacturing site. This alignment with green chemistry principles enhances the corporate image and meets the increasing demand from clients for sustainably sourced ingredients. The scalability is further supported by the modular nature of LED photoreactors, which can be added incrementally to increase capacity without massive downtime. This adaptability ensures that production can grow in tandem with market needs without compromising on environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this UV-induced synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this method for adoption. Understanding these nuances helps in making informed decisions about process integration and resource allocation. The responses cover aspects ranging from catalyst requirements to scalability potential, ensuring a comprehensive overview of the technology's capabilities. Stakeholders are encouraged to review these points to assess the fit for their specific production requirements and strategic goals.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented UV-induced method operates without transition metals, bases, or specialized additives, significantly reducing raw material costs and eliminating heavy metal contamination risks.
Q: What are the optimal reaction conditions for maximum yield?
A: Optimal yields up to 92% are achieved using a 6:1 ratio of ethyl acetate to acetone as solvent, with acetic acid promotion, under 365-375nm UV irradiation at room temperature for 16 hours.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the method features mild conditions, simple operation, and avoids hazardous high-pressure or high-temperature requirements, making it highly feasible for commercial scale-up and continuous production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminobenzamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the UV-induced route for 2-aminobenzamide compounds. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into industrial reality. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest international standards. We understand that the transition to new synthetic routes requires confidence in technical execution and supply stability, which is why our team of experts works closely with clients to validate process feasibility and optimize yield. By leveraging our infrastructure, partners can accelerate their time-to-market for critical pharmaceutical intermediates while mitigating the risks associated with process development.
We invite you to explore how our capabilities can support your supply chain optimization goals. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate the practical benefits of this technology for your portfolio. Collaborating with us ensures access to a reliable supply of high-quality intermediates produced through cutting-edge, sustainable methods. Let us help you navigate the complexities of chemical manufacturing and secure a competitive edge in the global marketplace.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
