Advanced Synthesis of Beta-Enolimine Boron Difluoride Complexes for Commercial Photoluminescent Manufacturing
Advanced Synthesis of Beta-Enolimine Boron Difluoride Complexes for Commercial Photoluminescent Manufacturing
The landscape of organic optoelectronic materials is undergoing a significant transformation driven by the demand for highly efficient, visible-light emitters that offer superior stability and ease of manufacturing. A pivotal development in this sector is detailed in patent CN101108860A, which discloses a novel class of boron difluoride complexes featuring a beta-enolimine structure. Unlike traditional pyrromethene or beta-diketonate analogues that often suffer from limited conjugation or ultraviolet emission, these new compounds leverage a unique N,O-bidentate coordination environment to produce intense yellow-green fluorescence in the visible spectrum. This technological breakthrough addresses critical pain points for R&D directors seeking reliable high-purity photoluminescent materials by offering a synthesis route that is not only chemically elegant but also industrially viable. The ability to tune emission properties through simple substituent modifications while maintaining a robust core structure positions this technology as a cornerstone for next-generation fluorescent probes and photosensitizers.
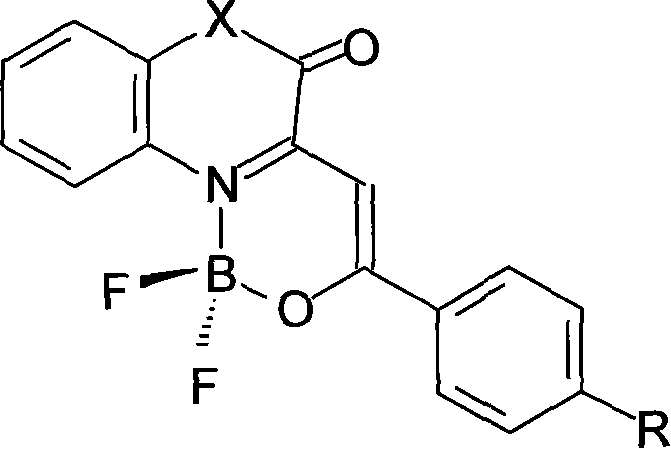
For procurement managers and supply chain heads, the implications of this patent extend far beyond mere chemical novelty. The synthesis methodology described eliminates the need for exotic catalysts or harsh reaction conditions, relying instead on readily available reagents like boron trifluoride etherate and common organic solvents. This simplicity translates directly into cost reduction in electronic chemical manufacturing, as it minimizes the operational complexity and safety hazards associated with more intricate coordination chemistry processes. Furthermore, the reported yields consistently exceed 90%, with crude purities often surpassing 98% after simple workup, which drastically reduces the burden on downstream purification units. By adopting this streamlined approach, manufacturers can achieve commercial scale-up of complex fluorescent intermediates with greater predictability and lower capital expenditure, ensuring a steady supply of critical materials for the burgeoning fields of bio-imaging and optoelectronic displays.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of organic boron difluoride complexes has been dominated by two primary structural motifs: the N,N-bidentate pyrromethene type and the O,O-bidentate beta-diketonate type. While these classes have been extensively researched, they present inherent limitations that restrict their utility in specific high-performance applications. As illustrated in the structural comparisons below, conventional N,N-ligands often require multi-step syntheses involving sensitive pyrrole derivatives, which can be costly and difficult to source in bulk quantities. Moreover, many existing beta-enolimine type complexes reported in prior art possess short conjugated systems that result in fluorescence emission falling within the ultraviolet region. This UV emission is suboptimal for direct human observation and limits their effectiveness in biological imaging where visible light penetration and detector sensitivity are paramount. The reliance on these older architectures often forces R&D teams to compromise between synthetic accessibility and optical performance, creating a bottleneck in the development of advanced photodynamic therapy agents.
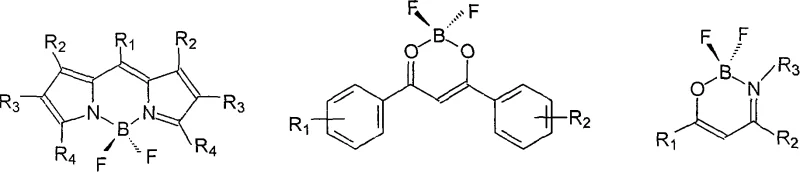
The Novel Approach
The innovation presented in the patent data overcomes these historical barriers by strategically integrating a beta-enaminone structure into a 3-(2-oxo-2-arylethylidene)-3,4-dihydroquinoxalin-2-one or benzoxazin-2-one scaffold. This structural engineering creates a tautomeric equilibrium that favors the formation of a beta-enolimine capable of stable N,O-bidentate coordination with the boron center. The result is a complex with an extended conjugated system that shifts the emission maximum to approximately 520nm, squarely within the yellow-green visible region where the human eye exhibits peak sensitivity. This approach not only enhances the optical properties but also simplifies the synthetic workflow. The ligands are derived from accessible precursors, and the complexation step is remarkably fast, requiring only brief reflux times. For a reliable photoluminescent material supplier, this represents a paradigm shift towards processes that deliver superior product performance without the traditional penalties of low yield or difficult purification, effectively bridging the gap between academic discovery and industrial reality.
Mechanistic Insights into Boron Trifluoride-Mediated Cyclization
The core of this synthesis lies in the efficient coordination of the boron atom to the tautomeric ligand system. The reaction mechanism initiates with the interaction between the beta-enaminone precursor and boron trifluoride diethyl etherate (BF3·OEt2) in a mixed solvent system. The Lewis acidic nature of the boron species facilitates the deprotonation and subsequent chelation of the ligand, locking it into the beta-enolimine form. This N,O-coordination is thermodynamically favored due to the formation of a stable six-membered ring involving the boron atom, the nitrogen of the quinoxalinone or benzoxazinone ring, and the oxygen of the enolate moiety. The use of a 1:1 volume ratio of acetic acid to toluene is critical; the acetic acid likely assists in proton transfer dynamics during complexation, while toluene provides the necessary thermal mass and solubility profile to maintain homogeneity during the reflux phase. This delicate balance of solvent polarity and acidity ensures that the reaction proceeds rapidly to completion without degrading the sensitive fluorophore core.
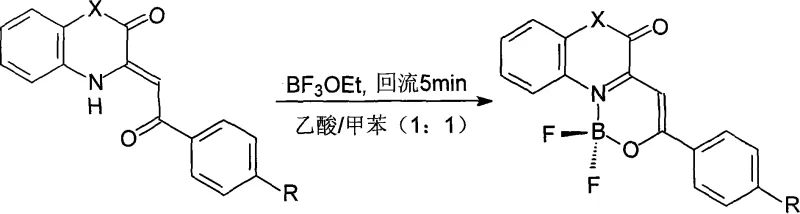
From an impurity control perspective, the mechanism offers distinct advantages that are crucial for maintaining stringent purity specifications. The rapid kinetics of the reaction, completed within just 5 minutes of reflux, minimize the residence time of intermediates that could otherwise undergo side reactions or decomposition. Furthermore, the product exhibits low solubility in the reaction medium upon cooling, leading to spontaneous precipitation. This "crash out" phenomenon acts as an intrinsic purification step, excluding soluble impurities and unreacted starting materials from the crystal lattice. The patent data indicates that substituents on the aryl ring (such as methoxy, chloro, or nitro groups) influence the molar absorptivity and fluorescence quantum yield but do not fundamentally alter the robustness of the cyclization mechanism. This mechanistic resilience ensures that the process remains consistent across a range of derivatives, providing R&D teams with a predictable platform for generating diverse libraries of fluorescent tags and sensors with minimal batch-to-batch variability.
How to Synthesize Beta-Enolimine Boron Difluoride Complexes Efficiently
The practical execution of this synthesis is designed for scalability and operational safety, making it ideal for transition from laboratory benchtop to pilot plant operations. The procedure avoids the use of moisture-sensitive reagents that require rigorous inert atmosphere techniques, as the reaction tolerates the conditions well enough to proceed efficiently with standard equipment. The key to success lies in the precise control of the cooling rate post-reflux; slow降温 (cooling) promotes the formation of well-defined crystals rather than amorphous powders, which facilitates easier filtration and washing. Detailed standardized operating procedures for this transformation, including specific stoichiometric ratios and temperature profiles optimized for different substituents, are outlined in the technical guide below to ensure reproducibility.
- Dissolve the beta-enaminone ligand precursor in a 1: 1 volume mixture of acetic acid and toluene.
- Add a 5-10 fold molar excess of boron trifluoride diethyl etherate to the reaction mixture under heating.
- Reflux the solution for 5 minutes, then slowly cool to room temperature to induce crystallization of the final complex.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the adoption of this synthesis route offers compelling economic and logistical benefits. The primary driver of value is the drastic simplification of the process workflow. By reducing the reaction time to merely minutes and eliminating the need for column chromatography in most cases, the throughput of the manufacturing facility can be significantly increased. This efficiency gain directly correlates to lower operational expenditures, as less energy is consumed for heating and stirring, and less solvent is required for purification. Additionally, the high thermal stability of the final products (with melting points often exceeding 300°C) reduces the risks associated with storage and transportation, allowing for more flexible logistics planning. These factors combine to create a supply chain that is both resilient and cost-effective, addressing the perennial challenge of reducing lead time for high-purity fluorescent intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of commodity chemicals like acetic acid and toluene significantly lowers the raw material cost base. Furthermore, the high crude purity (>98%) means that resource-intensive purification steps such as preparative HPLC or multiple recrystallizations are often unnecessary, leading to substantial savings in labor and consumables. The process efficiency ensures that the cost per gram of the final active material is minimized, enhancing the overall margin profile for downstream applications in medical diagnostics and display technologies.
- Enhanced Supply Chain Reliability: The starting materials, specifically the substituted 3-(2-oxo-2-arylethylidene) precursors, are synthesized from widely available building blocks, mitigating the risk of raw material shortages. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in environmental parameters, ensuring consistent output quality. This reliability is critical for long-term contracts with pharmaceutical and electronics clients who require guaranteed delivery schedules and consistent material performance to maintain their own production timelines without interruption.
- Scalability and Environmental Compliance: The solvent system used is relatively benign compared to halogenated alternatives often found in fine chemical synthesis, simplifying waste stream management and reducing the environmental footprint of the manufacturing process. The high yield (>90%) maximizes atom economy, meaning less waste is generated per unit of product. This aligns with modern green chemistry principles and regulatory standards, making it easier to obtain necessary environmental permits for scale-up. The ability to scale from grams to tons without re-optimizing the core chemistry provides a clear path for meeting growing market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these specialized boron complexes. Understanding these details is essential for integrating this technology into existing product lines or research pipelines. The answers are derived directly from the experimental data and characterization results provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers evaluating this material for potential adoption.
Q: What are the thermal stability characteristics of these boron difluoride complexes?
A: According to patent data, these complexes exhibit strong thermal stability with melting points ranging from 236°C to 326°C depending on the substituents, making them robust for various industrial processing conditions.
Q: How does the substitution pattern affect fluorescence intensity?
A: Electron-donating groups generally enhance fluorescence intensity compared to electron-withdrawing groups, although all variants maintain strong yellow-green emission in the visible region around 520nm.
Q: Is the purification process complex for large-scale production?
A: The process is highly scalable; crude products typically exceed 98% purity after simple filtration and washing, with recrystallization available as a secondary purification step if higher specifications are required.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Boron Difluoride Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the beta-enolimine boron difluoride architecture in advancing the field of functional materials. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate the methodologies described in patent CN101108860A into commercial reality. Our facilities are equipped to handle the specific solvent systems and thermal profiles required for this synthesis, ensuring that every batch meets the highest standards of quality. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, coupled with stringent purity specifications enforced by our rigorous QC labs. Whether you require custom derivatives for specific emission wavelengths or bulk supply for device fabrication, our team is ready to support your objectives with precision and speed.
We invite you to explore how this advanced chemistry can optimize your product portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data for existing analogues and conduct comprehensive route feasibility assessments for novel targets. Let us collaborate to accelerate your development timeline and secure a competitive advantage in the global market for high-performance fluorescent materials.
