Advanced Bismuth-Catalyzed Oxidative Cleavage for High-Purity Carbonyl Manufacturing
The chemical industry is constantly seeking more sustainable and economically viable pathways for the synthesis of high-value carbonyl compounds, which serve as critical building blocks in the pharmaceutical and agrochemical sectors. Patent CN1761640A introduces a groundbreaking methodology for the production of carbonyl compounds represented by formula (2) and formula (3) through the oxidative cleavage of vicinal diols. This innovation marks a significant departure from traditional oxidation methods that rely on hazardous heavy metals, proposing instead a robust system utilizing trivalent bismuth compounds in conjunction with a base and bromine sources. For R&D directors and process chemists, this patent offers a compelling solution to the longstanding challenge of achieving high selectivity without the environmental burden of chromium or lead waste. The technology enables the efficient transformation of diverse diol substrates into corresponding aldehydes or ketones under mild conditions, ensuring that the integrity of sensitive functional groups is maintained throughout the synthetic sequence.
Furthermore, the versatility of this bismuth-catalyzed system extends to a wide range of substituents, allowing for the customization of molecular architectures required in complex drug synthesis and specialty chemical manufacturing. By leveraging the unique redox properties of trivalent bismuth, the process achieves remarkable conversion rates while minimizing the formation of over-oxidized byproducts such as carboxylic acids, a common pitfall in conventional oxidation protocols. This level of control is paramount for manufacturers aiming to streamline purification processes and maximize overall throughput. As a reliable agrochemical intermediate supplier, understanding and adopting such advanced catalytic cycles is essential for maintaining competitiveness in a market that increasingly demands green chemistry solutions alongside cost efficiency.
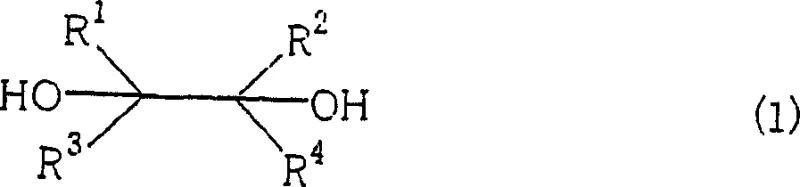
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of carbonyl compounds from vicinal diols has relied heavily on stoichiometric oxidants that pose severe environmental and safety risks. Traditional methods employing chromic acid, for instance, are notorious for generating vast quantities of toxic hexavalent chromium waste, which necessitates expensive and complex disposal procedures to meet stringent environmental regulations. Additionally, chromium-based oxidations often lack selectivity, frequently leading to the over-oxidation of aldehydes to carboxylic acids, thereby reducing the yield of the desired target molecule and complicating downstream purification. Similarly, the use of lead tetraacetate, while effective for certain substrates, is plagued by issues regarding storage stability and the inherent toxicity of lead, making it less desirable for large-scale operations where worker safety is a priority.
Alternative methods utilizing periodic acid or periodates offer better selectivity but come with their own set of economic drawbacks, primarily due to the high cost of these reagents when used in stoichiometric amounts. The reliance on pentavalent organic bismuth reagents in earlier literature also presented challenges, as these high-valent species are often expensive to synthesize and handle. Consequently, existing technologies have struggled to balance the triad of cost, safety, and efficiency, leaving a gap in the market for a method that can deliver high-purity products without the associated ecological footprint. For procurement managers, these legacy methods translate into volatile raw material costs and potential supply chain disruptions caused by regulatory crackdowns on heavy metal usage.
The Novel Approach
The methodology disclosed in CN1761640A revolutionizes this landscape by introducing a catalytic system based on trivalent bismuth compounds, which are significantly less toxic and more environmentally benign than their hexavalent counterparts. In this novel approach, the trivalent bismuth acts as a mediator in the presence of a base and a cheap bromine source, facilitating the oxidative cleavage of the carbon-carbon bond in vic-diols with exceptional precision. This system effectively circumvents the need for stoichiometric amounts of expensive oxidants, as the bromine serves as the terminal oxidant while the bismuth cycles through oxidation states to drive the reaction forward. The result is a process that not only reduces raw material costs but also simplifies the waste stream, as the resulting bismuth salts are easier to manage than chromium or lead residues.
Moreover, the reaction conditions are remarkably flexible, operating effectively across a temperature range of approximately -10°C to 100°C, which allows for fine-tuning based on the thermal sensitivity of the substrate. The inclusion of optional additives such as amides or imides, specifically succinimide, further enhances the reaction efficiency, pushing yields to impressive levels as demonstrated in the patent examples. This adaptability makes the process highly suitable for the commercial scale-up of complex aldehydes, providing a robust platform for manufacturing diverse chemical intermediates. By shifting away from hazardous reagents, manufacturers can achieve significant cost reduction in fine chemical manufacturing while simultaneously improving their sustainability profile.
Mechanistic Insights into Bismuth-Catalyzed Oxidative Cleavage
At the heart of this innovative process lies a sophisticated catalytic cycle involving the interplay between trivalent bismuth species and bromine. The trivalent bismuth compound, such as triphenylbismuth, likely functions as a Lewis acid activator that coordinates with the hydroxyl groups of the vic-diol substrate, increasing the electrophilicity of the carbon centers. Upon introduction of the bromine source, an active brominating species is generated in situ, which facilitates the formation of a cyclic bismuth intermediate or a bromo-ether species that primes the carbon-carbon bond for cleavage. The presence of a base, typically an alkali metal carbonate like potassium carbonate, is crucial for neutralizing the hydrogen bromide generated during the reaction and maintaining the optimal pH for the catalytic cycle to proceed without inhibition. This synergistic effect ensures that the oxidation proceeds smoothly to the carbonyl stage without stalling at intermediate oxidation states.
A particularly noteworthy aspect of the mechanism is the role of amide or imide additives, which appear to stabilize the transition state or perhaps regenerate the active bismuth species more efficiently. In the specific context of synthesizing pyrethroid intermediates, the transformation of diol (4) into aldehyde (5) exemplifies the high fidelity of this mechanism. The reaction preserves the stereochemistry of the cyclopropane ring, which is critical for the biological activity of the final agrochemical product. 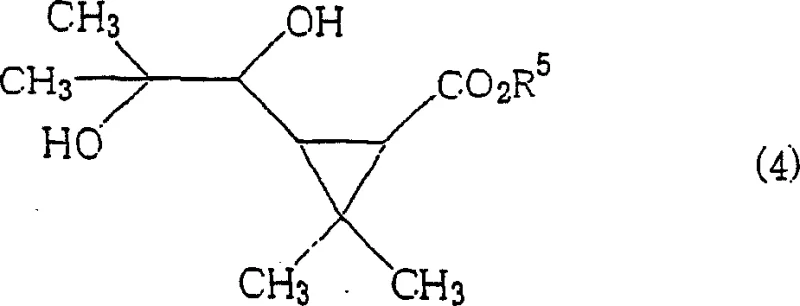
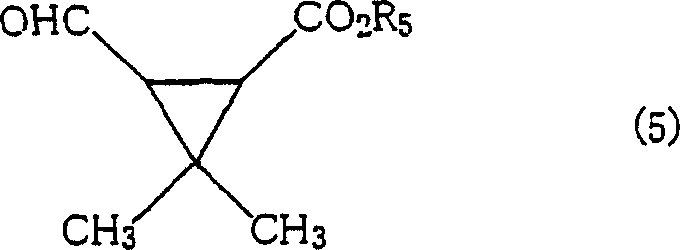 The ability to convert complex diols like 3,3-dimethyl-2-(2-methyl-1,2-dihydroxypropyl)cyclopropanecarboxylates into their corresponding formyl derivatives with yields reaching up to 97% underscores the mechanistic robustness. This level of impurity control is vital for R&D teams focused on developing high-purity OLED material or pharmaceutical intermediates where trace contaminants can compromise product performance.
The ability to convert complex diols like 3,3-dimethyl-2-(2-methyl-1,2-dihydroxypropyl)cyclopropanecarboxylates into their corresponding formyl derivatives with yields reaching up to 97% underscores the mechanistic robustness. This level of impurity control is vital for R&D teams focused on developing high-purity OLED material or pharmaceutical intermediates where trace contaminants can compromise product performance.
How to Synthesize Carbonyl Compounds Efficiently
The practical implementation of this bismuth-catalyzed oxidation involves a straightforward protocol that can be easily adapted for batch or continuous flow processing. The general procedure entails mixing the vic-diol substrate with the trivalent bismuth catalyst and the base in a suitable solvent such as acetonitrile, followed by the controlled addition of the bromine source. Detailed operational parameters, including specific molar ratios and addition rates, are critical for maximizing yield and minimizing side reactions.
- Prepare a reaction mixture containing the vic-diol substrate, a trivalent bismuth compound (e.g., triphenylbismuth), and a base (e.g., potassium carbonate) in a suitable solvent like acetonitrile.
- Optionally add an amide or imide compound such as succinimide to enhance reaction yield and selectivity during the oxidative process.
- Slowly add bromine or an inorganic bromine compound to the mixture at controlled temperatures between -10°C and 100°C, followed by filtration and concentration to isolate the carbonyl product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this bismuth-mediated process offers transformative benefits for supply chain resilience and cost management. The substitution of expensive and regulated oxidants like periodic acid or lead tetraacetate with inexpensive bromine and catalytic bismuth results in a drastic reduction in raw material expenditure. This shift not only lowers the direct cost of goods sold but also mitigates the financial risks associated with the fluctuating prices of specialty oxidizing agents. Furthermore, the elimination of toxic heavy metals from the waste stream significantly reduces the overhead costs related to environmental compliance, waste treatment, and disposal, contributing to substantial cost savings over the lifecycle of the product.
- Cost Reduction in Manufacturing: The use of catalytic amounts of bismuth rather than stoichiometric heavy metal reagents fundamentally alters the cost structure of carbonyl production. By avoiding the purchase of high-cost oxidants and reducing the volume of hazardous waste requiring specialized treatment, manufacturers can achieve a leaner cost base. The process efficiency, evidenced by high yields in patent examples, means less raw material is wasted, directly improving the atom economy of the synthesis. This economic efficiency is crucial for maintaining margins in the competitive agrochemical intermediate market.
- Enhanced Supply Chain Reliability: The reagents required for this process, including bromine, potassium carbonate, and triphenylbismuth, are commodity chemicals with stable and widespread global supply chains. Unlike niche oxidants that may suffer from supply shortages or geopolitical constraints, these materials are readily available from multiple vendors. This availability ensures consistent production schedules and reduces the risk of delays caused by raw material scarcity. For supply chain heads, this reliability translates into predictable lead times and the ability to scale production rapidly in response to market demand without bottlenecks.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of highly toxic byproducts make this process inherently scalable from laboratory to industrial tonnage. The simplified workup procedure, which often involves basic filtration and concentration, reduces the complexity of plant operations and the need for specialized containment equipment. Moreover, aligning with green chemistry principles by avoiding chromium and lead enhances the corporate sustainability profile, facilitating easier regulatory approvals and access to markets with strict environmental standards. This scalability ensures that the technology can support the commercial scale-up of complex polymer additives or fine chemicals without compromising safety.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this bismuth-catalyzed oxidative cleavage technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities.
Q: What are the primary advantages of using bismuth catalysts over chromium-based oxidants?
A: Bismuth catalysts eliminate the generation of toxic heavy metal waste associated with chromium and lead reagents, significantly reducing environmental compliance costs and simplifying waste disposal protocols in industrial settings.
Q: Can this process be scaled for the production of pyrethroid intermediates?
A: Yes, the patent explicitly demonstrates high-yield synthesis of key pyrethroid aldehyde intermediates, such as methyl 3,3-dimethyl-2-formylcyclopropanecarboxylate, proving its viability for large-scale agrochemical manufacturing.
Q: Does the reaction require expensive stoichiometric oxidants like periodates?
A: No, the method utilizes inexpensive bromine or inorganic bromine compounds as the terminal oxidant in the presence of a catalytic amount of bismuth, offering substantial cost reductions compared to traditional periodate methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl Compounds Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced synthetic methodologies to deliver superior chemical solutions to our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of carbonyl compounds meets the exacting standards required by the pharmaceutical and agrochemical industries. Our commitment to quality ensures that clients receive high-purity intermediates that facilitate seamless downstream synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this innovative bismuth-catalyzed route can be tailored to your specific manufacturing needs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your product portfolio. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this sustainable and efficient technology into your supply chain.
