Advanced Biocatalytic Route for High-Purity Florfenicol Intermediate Manufacturing
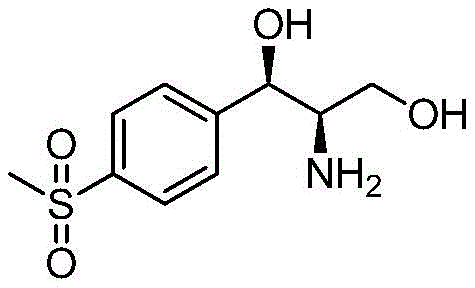
The pharmaceutical and veterinary industries are constantly seeking more efficient pathways to produce critical active ingredients, and the synthesis of Florfenicol intermediates stands as a prime example of this technological evolution. Patent CN109797174B introduces a groundbreaking biocatalytic approach for the preparation of (1R,2R)-2-amino-1-(4-(methylsulfonyl)phenyl)propane-1,3-diol, a pivotal precursor in the manufacture of the broad-spectrum antibacterial drug Florfenicol. This innovation leverages specific ketoreductases to achieve stereoselective reduction under remarkably mild conditions, addressing long-standing challenges related to yield, environmental impact, and cost efficiency in veterinary drug manufacturing. By shifting away from traditional chemical resolution or heavy metal-catalyzed hydrogenation, this method offers a sustainable alternative that aligns with modern green chemistry principles while ensuring the high optical purity required for regulatory compliance.
For procurement managers and supply chain directors, the adoption of this biocatalytic technology represents a strategic opportunity for cost reduction in veterinary antibiotic manufacturing. Traditional methods often rely on scarce resources or generate significant waste streams that require expensive disposal protocols. In contrast, the enzymatic route utilizes renewable biological catalysts that can be produced via fermentation, thereby stabilizing raw material costs and reducing dependency on volatile precious metal markets. Furthermore, the elimination of toxic heavy metals simplifies the purification process and minimizes the environmental footprint, which is increasingly critical for maintaining social license to operate in the global fine chemicals sector. This transition not only enhances supply chain reliability but also future-proofs production facilities against tightening environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this chiral diol intermediate has been plagued by inefficiencies inherent in classical chemical methodologies. One prominent prior art, US3927054, describes a chiral resolution method using D-tartaric acid, which suffers from a fundamental theoretical limitation where the maximum yield cannot exceed 50% without recycling the unwanted enantiomer. In practice, this results in significant material loss, with reported resolution yields as low as 41%, driving up the cost of goods sold due to poor atom economy. Another approach, disclosed in Fudan University patent CN105152989B, employs asymmetric hydrogenation using Ruthenium catalysts. While effective, this method introduces expensive transition metals into the process, necessitating rigorous and costly removal steps to meet strict residual metal specifications for pharmaceutical products. Additionally, the use of heavy metals poses significant environmental hazards and safety risks during large-scale operations, complicating waste management and increasing the overall operational expenditure for manufacturers.
The Novel Approach
The novel approach detailed in CN109797174B circumvents these drawbacks by employing engineered ketoreductases to catalyze the stereoselective reduction of a ketone precursor. This biocatalytic strategy operates under ambient pressure and moderate temperatures, typically between 20-40°C, which drastically reduces energy consumption compared to high-pressure hydrogenation or extreme pH chemical reactions. The enzyme demonstrates exceptional specificity, targeting the pro-chiral ketone group to form the desired (1R,2R) configuration with high fidelity. By integrating a cofactor regeneration system, such as glucose dehydrogenase (GDH) coupled with glucose, the process ensures a continuous supply of reducing equivalents (NADH/NADPH) without the need for stoichiometric amounts of expensive cofactors. This creates a closed-loop catalytic cycle that maximizes resource utilization and minimizes waste generation, offering a robust and scalable solution for industrial applications.
Mechanistic Insights into Ketoreductase-Catalyzed Stereoselective Reduction
At the heart of this innovation lies the precise molecular recognition and catalytic activity of specific ketoreductase enzymes, such as those identified by SEQ ID NO: 1, 2, or 6. These enzymes facilitate the hydride transfer from the reduced cofactor NADH or NADPH to the carbonyl carbon of the substrate, N-[1-hydroxymethyl-2-(4-methanesulfonyl-phenyl)-2-oxo-ethyl]-acetamide (Compound 3). The active site of the enzyme imposes a rigid steric environment that dictates the trajectory of the hydride attack, ensuring that reduction occurs exclusively from one face of the planar carbonyl group. This spatial constraint is what drives the formation of the specific (1R,2R) stereoisomer with exceptional diastereomeric and enantiomeric excess. The patent data highlights that certain variants, particularly Enzyme 6, exhibit superior activity and selectivity, achieving conversion rates of up to 94% with 100% de and 100% ee values, effectively eliminating the formation of unwanted stereoisomers that would otherwise complicate downstream purification.
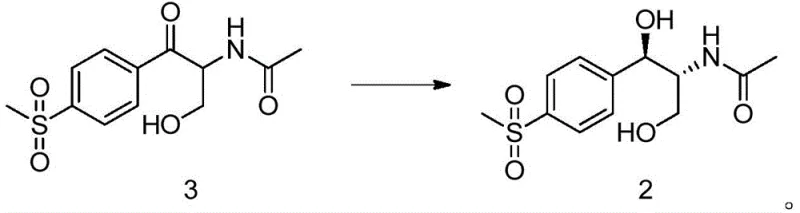
Beyond the primary reduction step, the control of impurities is meticulously managed through the specificity of the biocatalyst and the mildness of the reaction conditions. Unlike chemical reducing agents like sodium borohydride, which can be indiscriminate and reduce other functional groups or cause epimerization, the ketoreductase targets only the specific ketone moiety. The subsequent hydrolysis of the acetamide group to reveal the free amine is performed under controlled alkaline conditions, preserving the integrity of the newly formed chiral centers. The patent specifies that the final hydrolysis step yields the target diol with a 90% isolated yield and maintains the 100% ee purity established in the enzymatic step. This high level of impurity control reduces the burden on downstream crystallization and chromatography units, streamlining the overall manufacturing process and ensuring that the final API intermediate meets the stringent quality standards required for veterinary drug registration.
How to Synthesize Florfenicol Intermediate Efficiently
Implementing this biocatalytic route requires careful optimization of the reaction parameters to maximize enzyme performance and substrate solubility. The process begins with the preparation of a reaction体系 containing the ketone substrate and the recombinant ketoreductase, often expressed in E. coli hosts for high productivity. To sustain the catalytic cycle, a cofactor regeneration system is essential; the patent recommends using glucose dehydrogenase with glucose as the hydrogen donor, which is both cost-effective and easy to handle. The reaction pH is critically maintained between 8.0 and 9.0 using buffers like sodium carbonate, and the temperature is kept around 30°C to balance reaction rate with enzyme stability. Once the conversion is complete, indicated by HPLC analysis showing less than 2% residual starting material, the enzyme is denatured and removed, and the product is extracted and purified. The final step involves hydrolyzing the acetamide protecting group to yield the free amine diol.
- Prepare the reaction system containing the substrate N-[1-hydroxymethyl-2-(4-methanesulfonyl-phenyl)-2-oxo-ethyl]-acetamide (Compound 3) and the selected ketoreductase enzyme.
- Maintain the reaction pH between 7.0 and 10.0 (optimally 8.0-9.0) and temperature between 20-40°C, utilizing a cofactor regeneration system such as GDH/Glucose.
- Hydrolyze the resulting N-((1R,2R)-1,3-dihydroxy-1-(4-(methylsulfonyl)phenyl)propan-2-yl)acetamide (Compound 2) to obtain the final diol product.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and operational continuity, the shift to this enzymatic methodology offers profound commercial advantages that extend beyond simple yield improvements. The elimination of precious metal catalysts removes a significant variable cost component and mitigates the risk associated with the supply volatility of metals like Ruthenium. Furthermore, the aqueous nature of the biocatalytic process reduces the reliance on large volumes of organic solvents, leading to substantial cost savings in solvent procurement, recovery, and disposal. The mild operating conditions also translate to lower energy requirements for heating and cooling, contributing to a leaner manufacturing overhead. From a supply chain perspective, the ability to produce the enzyme via fermentation ensures a consistent and renewable source of the catalyst, decoupling production capacity from the constraints of chemical synthesis supply chains and enhancing overall resilience against market disruptions.
- Cost Reduction in Manufacturing: The transition from stoichiometric chiral resolving agents or expensive metal catalysts to recyclable biocatalysts fundamentally alters the cost structure of the synthesis. By avoiding the 50% yield loss typical of resolution methods and eliminating the need for costly metal scavengers, the overall material cost per kilogram of product is significantly reduced. Additionally, the high selectivity of the enzyme minimizes the formation of by-products, which reduces the load on purification equipment and lowers the consumption of auxiliary chemicals. This efficiency gain allows manufacturers to operate with higher throughput and lower variable costs, directly improving profit margins in a competitive generic drug market.
- Enhanced Supply Chain Reliability: Relying on biological catalysts produced through established fermentation technologies provides a more stable supply base compared to sourcing specialized chemical reagents that may have limited suppliers. The robustness of the enzyme under mild conditions means that production is less susceptible to interruptions caused by equipment failure or safety incidents associated with high-pressure or hazardous chemical processes. This reliability ensures consistent delivery schedules for downstream API manufacturers, reducing the risk of stockouts and enabling better inventory planning. The simplified workflow also shortens the overall production cycle time, allowing for faster response to fluctuations in market demand for Florfenicol.
- Scalability and Environmental Compliance: The biocatalytic process is inherently scalable, as demonstrated by the successful transition from laboratory to pilot scales in similar biotech applications. The absence of heavy metals and the use of biodegradable reagents simplify wastewater treatment and reduce the environmental liability of the manufacturing site. This alignment with green chemistry principles facilitates easier regulatory approval and helps companies meet increasingly strict environmental, social, and governance (ESG) targets. The reduced hazard profile also lowers insurance premiums and safety compliance costs, making the facility a more attractive asset for long-term investment and operation.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new manufacturing route is essential for stakeholders evaluating its adoption for commercial production. The following questions address common concerns regarding the practical implementation, quality attributes, and regulatory implications of the biocatalytic synthesis of Florfenicol intermediates. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making.
Q: What are the advantages of the enzymatic route over chemical resolution?
A: The enzymatic route described in CN109797174B avoids the theoretical 50% yield loss inherent in chiral resolution methods and eliminates the need for expensive heavy metal catalysts like Ruthenium, resulting in higher overall yields and reduced environmental toxicity.
Q: What is the stereochemical purity achievable with this method?
A: Experimental data from the patent indicates that using specific ketoreductases (such as SEQ ID NO: 6) can achieve conversion rates up to 94% with diastereomeric excess (de) values of 100% and enantiomeric excess (ee) values reaching 100%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates under mild conditions (20-40°C, pH 7-10) and uses renewable biocatalysts, making it highly scalable and safer for commercial manufacturing compared to high-pressure hydrogenation or harsh chemical resolution processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Florfenicol Intermediate Supplier
As the global demand for effective veterinary antibiotics continues to rise, securing a reliable florfenicol intermediate supplier is critical for maintaining uninterrupted API production. NINGBO INNO PHARMCHEM stands at the forefront of this technological shift, leveraging advanced biocatalytic platforms to deliver high-quality intermediates with unmatched consistency. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of major pharmaceutical companies. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch meets the highest standards of optical purity and chemical identity, minimizing the risk of downstream processing failures.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through innovative chemistry. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our biocatalytic route can enhance your operational efficiency. Please contact us to request specific COA data and route feasibility assessments, and let us partner with you to drive value and sustainability in your veterinary drug manufacturing operations.
