Transforming Picloram Waste into High-Value 2,3,5-Trichloropyridine via Advanced Catalytic Recycling
Introduction to Patent CN111909079B: A Breakthrough in Waste Valorization
The global agrochemical industry faces increasing pressure to adopt sustainable manufacturing practices, particularly in the management of hazardous production waste. Patent CN111909079B presents a transformative resource treatment method for picloram solid slag, a notorious byproduct in herbicide synthesis that has historically posed significant disposal challenges. This technology outlines a sophisticated three-stage chemical pathway—comprising decarboxylation, diazotization-chlorination, and selective dechlorination—to upcycle this waste stream into 2,3,5-trichloropyridine, a high-value intermediate with robust market demand. By shifting the paradigm from waste disposal to resource recovery, this process not only mitigates environmental liabilities but also creates a new revenue stream from what was previously considered a liability. For R&D directors and supply chain leaders, this represents a critical opportunity to integrate circular economy principles into their sourcing strategies for key pyridine derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
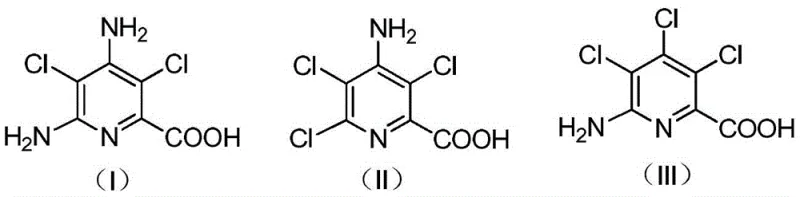
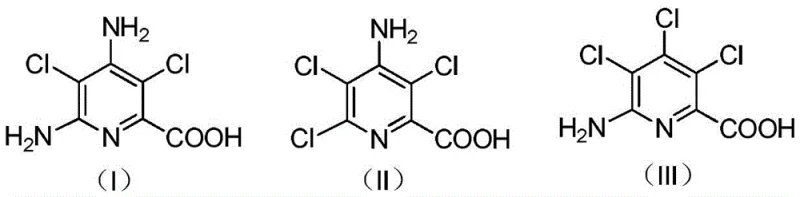
Traditionally, the treatment of picloram production waste has been limited to basic disposal or low-value recovery methods that fail to address the complex chemical nature of the slag. The solid residue generated during picloram synthesis is a heterogeneous mixture containing significant amounts of inorganic salts and complex organic impurities, primarily amino-substituted pyridine acids. As illustrated in the structural analysis below, these organic components include various isomers and salts (Formulas I, II, and III) that are difficult to separate and purify using standard crystallization techniques. Conventional approaches often result in the accumulation of hazardous solid waste or the production of low-grade intermediates that require extensive further processing, leading to inflated operational costs and inefficient resource utilization. The poor solubility of these organic components in common solvents further complicates the separation process, making traditional recycling economically unviable.
The Novel Approach
The innovative methodology described in CN111909079B overcomes these barriers by employing a targeted chemical modification strategy that converts the complex slag mixture into a unified, high-value product. Instead of attempting to separate the individual impurities directly, the process first subjects the entire organic fraction to a copper-catalyzed decarboxylation, effectively stripping the carboxyl groups to simplify the molecular architecture. This is followed by a rigorous diazotization and chlorination sequence that replaces amino groups with chlorine atoms, homogenizing the mixture into 2,3,4,5-tetrachloropyridine. Finally, a controlled dechlorination step selectively removes the chlorine at the 4-position, yielding 2,3,5-trichloropyridine. This approach bypasses the need for difficult physical separations of the initial sludge, turning a chemically messy problem into a streamlined synthetic route that maximizes atom economy and product value.
Mechanistic Insights into Copper-Catalyzed Decarboxylation and Zinc-Mediated Dechlorination
The core of this recycling technology lies in its precise control over reaction conditions to manage the complex impurity profile of the starting material. The initial decarboxylation step utilizes copper oxide as a catalyst in a specialized solvent system of 1,3-dimethyl-2-imidazolidinone and hydrochloric acid. This specific solvent choice is critical, as it facilitates the dissolution of the otherwise insoluble organic salts found in the slag while maintaining the thermal stability required for efficient CO2 evolution at 100-120°C. Following this, the diazotization step employs sodium nitrite and concentrated hydrochloric acid to convert amino groups into diazonium salts, which are subsequently displaced by chloride ions. This sequence is vital for eliminating nitrogen-containing impurities that could otherwise contaminate the final chloropyridine product, ensuring that the intermediate 2,3,4,5-tetrachloropyridine is formed with high fidelity.
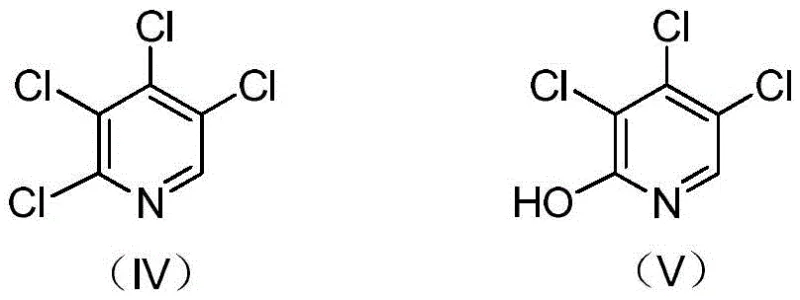
Impurity control is further refined during the conversion of the decarboxylated mixture to the tetrachloro-intermediate. A key challenge in this transformation is the formation of hydroxy-chloropyridine byproducts, such as the compound shown in Formula V, which can arise from incomplete chlorination or hydrolysis side reactions. The process addresses this by utilizing thionyl chloride in the presence of a catalytic amount of N,N-dimethylformamide, which acts as a potent chlorinating agent to convert these hydroxy impurities back into the desired chloro-species. The final dechlorination step leverages zinc powder in a methanol-water binary solvent system to selectively reduce the 4-chloro position. The polarity of this mixed solvent system is optimized to balance the solubility of the substrate and the reactivity of the zinc, preventing over-reduction to 2,5-dichloropyridine while ensuring complete conversion of the tetrachloro-precursor.
How to Synthesize 2,3,5-Trichloropyridine Efficiently
The synthesis of 2,3,5-trichloropyridine from picloram slag requires careful optimization of reaction parameters to ensure high yield and purity suitable for commercial applications. The process begins with the preparation of the decarboxylated mixture, followed by the critical diazotization-chlorination sequence to generate the tetrachloro-intermediate. The final reduction step demands precise control over the zinc powder stoichiometry and solvent composition to maximize selectivity for the 2,3,5-isomer. Detailed operational parameters, including specific temperature ramps, addition rates, and workup procedures, are essential for replicating the high efficiency demonstrated in the patent examples. For a comprehensive guide on executing this synthesis at scale, please refer to the standardized protocol below.
- Perform copper oxide-catalyzed decarboxylation of picloram solid slag in 1,3-dimethyl-2-imidazolidinone solvent with hydrochloric acid at 100-120°C to remove carboxyl groups.
- Subject the decarboxylated solid to diazotization using sodium nitrite and concentrated hydrochloric acid, followed by chlorination with thionyl chloride to yield 2,3,4,5-tetrachloropyridine.
- Execute selective dechlorination of 2,3,4,5-tetrachloropyridine using zinc powder in a methanol-water mixed solvent system to obtain the target 2,3,5-trichloropyridine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this recycling technology offers profound strategic advantages beyond simple cost arithmetic. By transforming a waste product into a valuable feedstock, manufacturers can significantly decouple their raw material costs from volatile commodity markets, creating a more resilient and predictable cost structure. The ability to source 2,3,5-trichloropyridine from internal waste streams reduces dependency on external suppliers of virgin petrochemical feedstocks, thereby enhancing supply chain security and reducing lead times associated with traditional procurement channels. Furthermore, the elimination of hazardous waste disposal fees and the potential for carbon credit generation through waste valorization contribute to a substantially improved bottom line, aligning financial performance with sustainability goals.
- Cost Reduction in Manufacturing: The primary economic driver of this process is the substitution of expensive virgin raw materials with low-cost or negative-cost waste feedstocks. By utilizing picloram solid slag, which is often incurred as a disposal liability, the effective cost of goods sold for the resulting 2,3,5-trichloropyridine is drastically reduced compared to traditional synthesis routes. Additionally, the process utilizes common and inexpensive reagents such as zinc powder, methanol, and hydrochloric acid, avoiding the need for precious metal catalysts or exotic ligands that typically inflate production costs. The recovery and recycling of solvents like 1,3-dimethyl-2-imidazolidinone further enhance the economic viability by minimizing consumable expenses.
- Enhanced Supply Chain Reliability: Integrating this recycling loop creates a closed-loop supply chain that is less susceptible to external market disruptions. Since the feedstock is generated internally as a byproduct of existing herbicide production, the availability of raw materials is inherently linked to the company's own production volume, ensuring a consistent and reliable supply of the intermediate. This vertical integration reduces the risks associated with supplier bankruptcy, geopolitical instability, or logistics bottlenecks that frequently plague global chemical supply chains. Consequently, manufacturers can guarantee more stable delivery schedules to their downstream customers, strengthening long-term contractual relationships.
- Scalability and Environmental Compliance: The unit operations involved in this process—filtration, crystallization, and distillation—are standard industrial practices that scale linearly from pilot to commercial production without requiring specialized equipment. This simplicity facilitates rapid capacity expansion to meet growing market demand for pyridine intermediates. From an environmental perspective, the process significantly reduces the volume of solid hazardous waste requiring landfilling or incineration, helping companies meet increasingly stringent regulatory compliance standards. The conversion of toxic slag into a useful chemical product also enhances the corporate sustainability profile, which is becoming a critical factor in vendor selection for major multinational agrochemical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this picloram slag recycling technology. These insights are derived directly from the experimental data and process descriptions within patent CN111909079B, providing a factual basis for evaluating the feasibility of this route. Understanding these nuances is essential for technical teams assessing the integration of this process into existing manufacturing lines.
Q: What are the primary organic impurities found in picloram solid slag?
A: The solid slag primarily contains complex amino-substituted pyridine acids, specifically compounds corresponding to structural formulas (I), (II), and (III), which are salts or derivatives of 4-amino-3,5,6-trichloropicolinic acid.
Q: How does this process handle hydroxy-chloropyridine impurities?
A: During the diazotization and chlorination steps, hydroxy impurities (such as formula V) are formed but are effectively removed or converted during the subsequent thionyl chloride treatment and distillation separation, ensuring high purity of the tetrachloropyridine intermediate.
Q: Why is zinc powder used in the final dechlorination step?
A: Zinc powder acts as a reducing agent in a mixed solvent of methanol and water, selectively removing the chlorine atom at the 4-position of 2,3,4,5-tetrachloropyridine to yield the desired 2,3,5-trichloropyridine with high selectivity and yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,5-Trichloropyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of waste-to-value technologies in reshaping the agrochemical intermediate landscape. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the one described in CN111909079B can be seamlessly transitioned from the laboratory to full-scale manufacturing. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of 2,3,5-trichloropyridine meets the exacting standards required for downstream herbicide synthesis. We are committed to delivering high-purity agrochemical intermediates that empower our clients to achieve their sustainability and efficiency targets.
We invite you to collaborate with us to explore how this recycling technology can optimize your supply chain and reduce your overall manufacturing footprint. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and waste profiles. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how we can serve as your strategic partner in sustainable chemical manufacturing.
