Advanced Manufacturing of High-Purity DOPO: A Technical Breakthrough for Global Supply Chains
Advanced Manufacturing of High-Purity DOPO: A Technical Breakthrough for Global Supply Chains
The global demand for halogen-free flame retardants has necessitated a rigorous re-evaluation of synthetic pathways for key intermediates like 9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide, commonly known as DOPO. Patent CN102229624B introduces a transformative synthesis and purification protocol that addresses the longstanding inefficiencies of traditional manufacturing. This technology leverages a controlled, multi-stage addition of phosphorus trichloride and a novel azeotropic dehydration technique to achieve exceptional purity levels exceeding 99% without the burden of high-vacuum distillation. For R&D directors and procurement strategists, this represents a pivotal shift towards more robust, cost-effective, and scalable production methodologies. The structural integrity and chemical stability of the final product are paramount for its application in circuit cards, semiconductor sealing materials, and advanced polymers.
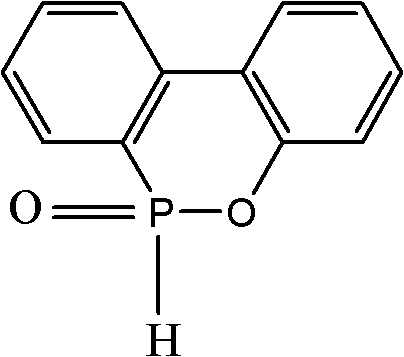
Furthermore, the elimination of complex vacuum requirements directly translates to reduced capital expenditure and simplified operational protocols, making this a highly attractive route for reliable flame retardant intermediate supplier networks. The patent data underscores a yield improvement to greater than 88%, validating the efficiency of the new catalytic system. By integrating these technical advancements, manufacturers can secure a competitive edge in the specialty chemical market, ensuring consistent quality and supply continuity for downstream applications in electronics and materials science.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of DOPO has been dominated by processes described in earlier patents such as DE 2034887 and US 5717127, which rely heavily on harsh reaction conditions and complex separation techniques. These conventional methods typically involve the generation of the intermediate 6-chloro-6H-dibenzo[c,e][1,2]oxaphosphorin (CDOP) followed by high-vacuum distillation to isolate the product or its precursors. The requirement for high-vacuum equipment imposes severe constraints on reactor design, significantly increasing both the initial investment and the ongoing maintenance costs for production facilities. Moreover, the high temperatures often necessary for these vacuum distillations promote undesirable side reactions, leading to increased impurity profiles that complicate downstream purification. The energy consumption associated with maintaining high vacuum and elevated temperatures is substantial, creating an environmental and economic burden that limits the scalability of these older technologies. Consequently, the final product often suffers from variable purity, necessitating additional refining steps that further erode profit margins and extend lead times for high-purity organophosphorus compounds.
The Novel Approach
In stark contrast, the methodology outlined in CN102229624B presents a streamlined, atmospheric-pressure alternative that fundamentally reengineers the reaction landscape. By employing a specific three-stage addition of phosphorus trichloride, the process effectively manages the exothermic nature of the initial phosphorylation, thereby suppressing the formation of byproducts at the source. The subsequent conversion of the intermediate to 2'-hydroxybiphenyl-2-phosphonic acid (HPPA) and its cyclodehydration is achieved through azeotropic distillation rather than high-vacuum stripping. This shift allows the reaction to proceed at significantly lower temperatures, typically between 80°C and 120°C, preserving the thermal stability of the sensitive phosphorus-heterocyclic framework. The use of metal salt catalysts during the cyclization step further accelerates the reaction kinetics, reducing the overall cycle time and enhancing throughput. This approach not only simplifies the equipment requirements but also ensures a consistently high purity of the final DOPO crystal, meeting the stringent specifications required for electronic grade applications.
Mechanistic Insights into Controlled Phosphorylation and Catalytic Cyclization
The core innovation of this synthesis lies in the precise kinetic control exerted during the formation of the CDOP intermediate. Instead of a bulk addition of phosphorus trichloride, which can lead to runaway exotherms and localized hot spots, the patent mandates a fractional dosing strategy. Approximately 70% to 85% of the phosphorus trichloride is introduced at ambient temperature, establishing the initial reaction matrix with anhydrous zinc chloride as the Lewis acid catalyst. As the temperature rises to the 105°C to 130°C range, a second aliquot of 10% to 20% is added, followed by the final portion at 140°C to 155°C. This staged addition ensures that the concentration of the reactive phosphorus species remains optimal for the electrophilic attack on the o-phenylphenol substrate, minimizing oligomerization and other degradation pathways. The result is a cleaner reaction mixture that facilitates easier isolation of the CDOP liquid, setting a strong foundation for the subsequent hydrolysis and cyclization steps.
Following the hydrolysis of CDOP to HPPA, the critical cyclodehydration step is driven by a metal-catalyzed azeotropic mechanism. The presence of metal salts, such as ferric chloride or zinc sulfate, activates the phosphonic acid group, lowering the activation energy required for the intramolecular condensation that forms the P-O-C heterocyclic ring. By utilizing aromatic solvents like xylene or chlorobenzene, the water generated during cyclization is continuously removed via azeotropic distillation at moderate reflux temperatures. This continuous removal of water shifts the equilibrium towards the product side according to Le Chatelier's principle, driving the reaction to completion without the need for extreme thermal stress. The careful selection of the metal catalyst and solvent system ensures that the cyclization proceeds rapidly and selectively, yielding a white solid powder with minimal colored impurities, which is essential for achieving the target purity of ≥99% in the final DOPO product.
How to Synthesize DOPO Efficiently
The synthesis of DOPO via this patented route involves a sequence of four distinct operational units: phosphorylation, hydrolysis, cyclodehydration, and final purification. Each step is optimized to maximize yield and minimize waste, leveraging standard chemical engineering unit operations that are easily adaptable to existing infrastructure. The process begins with the controlled reaction of o-phenylphenol and phosphorus trichloride, followed by a carefully managed hydrolysis to precipitate the phosphonic acid intermediate. The detailed standardized synthesis steps below outline the specific parameters for temperature, stoichiometry, and solvent selection required to replicate the high-efficiency results reported in the patent documentation.
- React o-phenylphenol with anhydrous zinc chloride and phosphorus trichloride (added in three portions) at 158-175°C to form CDOP liquid.
- Hydrolyze the CDOP liquid with water and an alcohol solvent under reflux to precipitate 2'-Hydroxybiphenyl-2-phosphonic acid (HPPA) crystals.
- Perform cyclodehydration on HPPA using an aromatic solvent and metal salt catalyst via azeotropic distillation at 80-120°C.
- Purify the resulting solid powder using an aliphatic hydrocarbon solvent to obtain final DOPO with purity ≥99%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis technology offers profound strategic benefits that extend beyond mere technical performance. The elimination of high-vacuum distillation equipment represents a significant reduction in capital intensity, allowing for faster deployment of production capacity and lower depreciation costs per unit. Furthermore, the ability to operate at lower temperatures and atmospheric pressure reduces energy consumption and enhances operator safety, aligning with modern sustainability goals and regulatory compliance standards. The robustness of the process ensures a stable supply of high-quality intermediates, mitigating the risks associated with batch-to-batch variability that often plague complex organophosphorus manufacturing.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of energy-intensive high-vacuum systems and the reduction of reaction temperatures. By avoiding the need for specialized vacuum pumps and the associated maintenance, facilities can realize substantial operational savings. Additionally, the improved yield of over 88% means that less raw material is wasted per kilogram of finished product, directly improving the cost of goods sold. The use of recyclable aromatic solvents in the cyclization step further contributes to cost efficiency by minimizing solvent purchase and disposal expenses, creating a leaner and more profitable manufacturing model for polymer additive production.
- Enhanced Supply Chain Reliability: The simplified equipment requirements and milder reaction conditions contribute to higher equipment availability and reduced downtime for maintenance. Unlike high-vacuum systems which are prone to leaks and seal failures, the atmospheric pressure setup is inherently more robust and easier to troubleshoot. This reliability translates into more predictable production schedules and shorter lead times for customers requiring urgent deliveries of flame retardant intermediates. The consistent quality of the output also reduces the need for re-processing or rejection of off-spec batches, ensuring a steady flow of inventory to meet market demand without interruption.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial quantities is straightforward due to the absence of complex vacuum constraints that often limit reactor size. The use of standard reflux and filtration techniques allows for easy replication in larger vessels, facilitating rapid capacity expansion to meet growing global demand. From an environmental perspective, the lower energy footprint and the potential for solvent recovery align with green chemistry principles, reducing the facility's overall carbon emissions. This compliance with environmental standards not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a responsible partner in the sustainable chemicals industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this DOPO synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is critical for stakeholders evaluating the feasibility of adopting this method for their own production lines or sourcing strategies.
Q: How does this new DOPO synthesis method improve upon traditional high-vacuum processes?
A: Traditional methods rely on high-vacuum distillation for both the intermediate CDOP and the final dehydration of HPPA, requiring expensive equipment and high energy consumption. This patented process eliminates the need for high-vacuum apparatus during purification and utilizes azeotropic distillation for cyclodehydration, significantly reducing equipment complexity and operational costs while maintaining purity above 99%.
Q: What is the significance of adding phosphorus trichloride in three portions?
A: Adding phosphorus trichloride in three distinct stages (70-85% initially, then 10-20% at 105-130°C, and the remainder at 140-155°C) allows for precise control over the exothermic reaction rate. This strategy prevents localized overheating and minimizes side reactions, leading to a cleaner reaction profile and higher overall yields compared to single-addition methods.
Q: Can this process be scaled for industrial production of flame retardant intermediates?
A: Yes, the process is designed for scalability. By avoiding high-vacuum constraints and utilizing standard reflux and filtration techniques with recyclable aromatic solvents, the method simplifies the engineering requirements for large-scale reactors. The reported yield of over 88% and purity of 99% demonstrate its viability for commercial manufacturing of complex organophosphorus compounds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DOPO Supplier
The technical advancements detailed in patent CN102229624B highlight the immense potential for optimizing the production of critical flame retardant intermediates. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into industrial reality. Our commitment to stringent purity specifications and the operation of rigorous QC labs guarantees that every batch of DOPO meets the exacting standards required for high-performance applications in electronics and polymers. We understand that consistency is key, and our state-of-the-art facilities are designed to maintain the precise control necessary for complex organophosphorus synthesis.
We invite you to collaborate with us to leverage these process improvements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can drive value and security for your business. Let us be your partner in delivering high-quality chemical solutions with unmatched reliability and efficiency.
