Advanced Continuous Flow Manufacturing of Edaravone for Global Pharmaceutical Supply Chains
Advanced Continuous Flow Manufacturing of Edaravone for Global Pharmaceutical Supply Chains
The pharmaceutical industry is constantly seeking robust, scalable, and safe manufacturing processes for critical active pharmaceutical ingredients and their intermediates. Patent CN111848517B, published in April 2023, introduces a groundbreaking preparation method for Edaravone, a potent free radical scavenger used extensively in the treatment of acute cerebral infarction. This technology represents a paradigm shift from traditional intermittent batch processing to modern continuous flow chemistry, utilizing microreactors or channel reactors to achieve unprecedented levels of control and efficiency. The structural integrity of the final product, 3-methyl-1-phenyl-2-pyrazoline-5-ketone, is paramount for its neuroprotective efficacy, and this new method ensures that integrity through precise kinetic management.
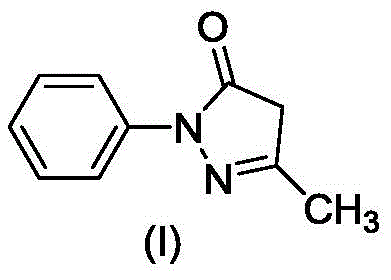
Edaravone functions by inhibiting lipid peroxidation and scavenging free radicals, thereby protecting vascular endothelial cells and neurons from oxidative damage. The traditional synthesis routes often suffer from significant drawbacks, including long reaction times, high energy consumption, and the generation of substantial impurities due to poor heat and mass transfer in large vessels. The innovation disclosed in CN111848517B addresses these pain points directly by employing a continuous loop reaction system. This approach not only enhances the safety profile by limiting the quantity of reactive material at high temperatures but also ensures a consistent product quality that is difficult to achieve with batch methods. For global supply chains, this translates to a reliable source of high-purity material that meets stringent regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
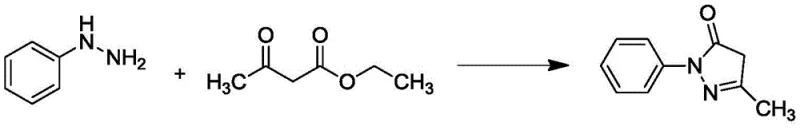
Historically, the industrial production of Edaravone has relied heavily on batch reactions conducted in large kettles or reaction bottles. These conventional methods typically involve heating a mixture of phenylhydrazine and ethyl acetoacetate in protic solvents like ethanol or acetic acid for several hours. The inherent limitations of this batch mode are multifaceted and pose serious challenges for modern manufacturing. Firstly, the heat transfer efficiency in large vessels is poor, leading to hot spots that can trigger side reactions and the formation of complex impurity profiles. Secondly, the long residence time at elevated temperatures increases the risk of product degradation and safety incidents, particularly when dealing with hydrazine derivatives which can be energetically unstable. Furthermore, the batch-to-batch variability is a persistent issue; slight differences in mixing speed, heating ramp rates, or cooling efficiency can result in fluctuating yields and purity levels, complicating quality control and regulatory compliance.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a continuous flow reactor, such as a microchannel reactor, to perform the cyclization reaction. This technology allows for the precise control of reaction parameters, including temperature, pressure, and crucially, residence time. By pumping phenylhydrazine and ethyl acetoacetate through a heated channel with a retention time of merely 2 to 20 minutes, the reaction reaches completion almost quantitatively. The high surface-area-to-volume ratio of the microreactor ensures rapid heat exchange, eliminating hot spots and maintaining a uniform reaction environment. This method supports both solvent-based and solvent-free conditions, offering flexibility in process design. The result is a streamlined operation that drastically reduces the generation of byproducts and eliminates the yield fluctuations common in batch processing.
Mechanistic Insights into Acid-Catalyzed Cyclization in Flow
The core chemical transformation involves the condensation of phenylhydrazine with ethyl acetoacetate to form the pyrazolone ring, a reaction that is fundamentally driven by nucleophilic attack followed by cyclization and elimination. In the continuous flow setup, this mechanism is optimized through the use of protic acids, such as acetic acid, hydrochloric acid, or sulfuric acid, which act as catalysts to accelerate the reaction kinetics. The presence of the acid protonates the carbonyl oxygen of the beta-keto ester, increasing its electrophilicity and facilitating the attack by the nucleophilic nitrogen of the phenylhydrazine. In a batch system, maintaining the optimal acid concentration and distribution throughout the bulk liquid is challenging, often leading to localized over-acidification or incomplete reaction zones. However, in the laminar flow regime of a microreactor, mixing occurs primarily through diffusion and chaotic advection, ensuring that the catalyst is uniformly distributed at the molecular level almost instantaneously upon contact.
Furthermore, the continuous flow environment provides superior control over the reaction trajectory, effectively suppressing the formation of impurities that arise from over-reaction or polymerization. The short residence time (as low as 2 minutes in optimized examples) means that once the Edaravone molecule is formed, it is quickly removed from the high-temperature zone, preventing thermal degradation. This kinetic control is critical for achieving the reported crude purity of greater than 99.0%. Additionally, the ability to operate under solvent-free conditions at temperatures between 130°C and 250°C leverages the melt state of the reactants and products to facilitate flow. This not only simplifies the downstream purification process by removing the need for solvent recovery but also aligns with green chemistry principles by minimizing waste generation. The precise stoichiometry control, with molar ratios tightly maintained between 0.8:1 and 1.2:1, further ensures that unreacted starting materials are minimized, simplifying the final isolation steps.
How to Synthesize Edaravone Efficiently
The synthesis of Edaravone via this continuous flow method offers a clear pathway for laboratories and manufacturing facilities to upgrade their production capabilities. The process begins with the preparation of the feed streams, where phenylhydrazine and ethyl acetoacetate are metered using high-precision pumps. Depending on the specific embodiment chosen, a protic acid catalyst may be introduced as a separate stream or pre-mixed with one of the reactants. The key to success lies in the selection of the reactor hardware—specifically, a microchannel or tubular reactor capable of withstanding the required temperatures and pressures while providing the necessary residence time. The reaction mixture is then heated to the target temperature, ranging from reflux conditions in solvent to over 140°C in solvent-free modes. Upon exiting the reactor, the effluent is immediately quenched, typically by pouring into water or an organic solvent, causing the product to precipitate as a high-purity solid. Detailed standardized synthetic steps for implementing this protocol are provided in the guide below.
- Prepare feed solutions of phenylhydrazine and ethyl acetoacetate, optionally with a protic acid catalyst like acetic acid.
- Pump the reagents simultaneously into a heated continuous flow reactor (microchannel or tubular) maintaining temperatures between 130°C and 250°C.
- Collect the effluent, quench with water or organic solvent, and filter the precipitated solid to obtain high-purity Edaravone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this continuous flow technology offers compelling strategic advantages that extend beyond simple chemical yield. The transition from batch to continuous processing fundamentally alters the cost structure and risk profile of Edaravone manufacturing. By eliminating the need for large reaction vessels and the associated infrastructure for heating and cooling massive batches, the capital expenditure for production capacity can be significantly optimized. Moreover, the drastic reduction in reaction time—from hours to minutes—means that the same physical footprint can produce a vastly higher volume of product over time, enhancing asset utilization rates. This efficiency gain is critical for meeting the demands of a growing market for neuroprotective agents without the need for proportional increases in facility size.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by several key factors inherent to the flow chemistry design. Firstly, the ability to operate under solvent-free conditions eliminates the substantial costs associated with purchasing, recovering, and disposing of large volumes of organic solvents. Secondly, the near-quantitative conversion rates and high crude purity reduce the burden on downstream purification units, such as crystallization and chromatography, leading to lower operational expenditures. Thirdly, the enhanced safety profile reduces insurance premiums and the need for extensive safety containment systems. Finally, the energy efficiency of microreactors, which require less energy to heat small volumes compared to large kettles, contributes to a lower overall carbon footprint and utility costs.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the variability of batch processes, where a single failed batch can disrupt delivery schedules for weeks. The continuous nature of this flow method ensures a steady, uninterrupted output of material, smoothing out production planning and inventory management. The consistency of the product quality, with purity levels consistently exceeding 99%, minimizes the risk of batch rejection during quality control testing. This reliability allows pharmaceutical customers to reduce their safety stock levels and operate with leaner inventories, knowing that the supply of this critical intermediate is stable and predictable. Furthermore, the use of commercially available starting materials like phenylhydrazine and ethyl acetoacetate ensures that raw material sourcing remains straightforward and resilient.
- Scalability and Environmental Compliance: Scaling chemical processes from the lab to the plant is notoriously difficult, often requiring complete re-optimization of parameters. Continuous flow chemistry offers a unique advantage here through "numbering up," where additional reactor modules are added in parallel to increase capacity without changing the reaction conditions. This modularity allows for flexible capacity expansion that matches market demand. From an environmental perspective, the process aligns with increasingly stringent global regulations on waste disposal. The reduction in solvent use and the minimization of byproduct formation significantly lower the volume of hazardous waste generated. This not only simplifies compliance with environmental permits but also enhances the corporate social responsibility profile of the manufacturing entity, a factor that is becoming increasingly important for multinational pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this continuous flow technology for Edaravone production. These insights are derived directly from the experimental data and technical specifications outlined in patent CN111848517B. Understanding these details is crucial for R&D teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of the supply source. The answers reflect the robustness of the method across different operating conditions, including variations in acid catalysts and temperature regimes.
Q: What are the primary safety advantages of the continuous flow method for Edaravone?
A: The continuous flow method significantly reduces the volume of reactive materials held at high temperatures at any given time, minimizing the risk of thermal runaway compared to large batch reactors.
Q: How does this process improve product purity compared to traditional batch synthesis?
A: By precisely controlling residence time (2-20 minutes) and temperature, the process prevents over-reaction and degradation, resulting in crude product purity exceeding 99.0%.
Q: Can this method be scaled for industrial production without solvents?
A: Yes, the patent explicitly describes a solvent-free variant where the temperature is maintained above the melting point of the product (130-250°C), facilitating green chemistry and reducing waste disposal costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Edaravone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing technologies like continuous flow chemistry requires a partner with deep technical expertise and a commitment to quality. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this patent can be fully realized at an industrial scale. Our facilities are equipped with state-of-the-art flow chemistry reactors and rigorous QC labs capable of verifying the stringent purity specifications required for pharmaceutical intermediates. We understand that consistency is key in the pharma supply chain, and our processes are designed to deliver batch-after-batch reproducibility that meets the highest global standards.
We invite potential partners to engage with our technical team to explore how this innovative synthesis route can optimize your supply chain for Edaravone. Whether you are looking to secure a long-term supply agreement or need assistance with process development, we are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value for your organization.
