Advanced Oxidative Synthesis of Tafamidis Intermediates for Commercial Scale-Up
Introduction to Next-Generation Tafamidis Synthesis
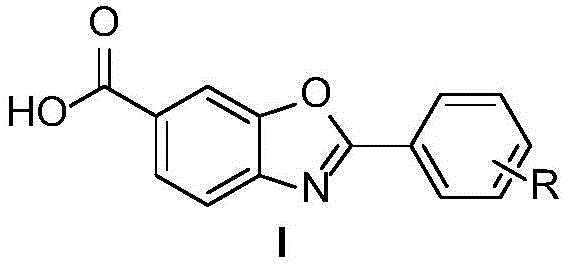
The pharmaceutical landscape for treating transthyretin-mediated amyloidosis (ATTR-CM) has been revolutionized by Tafamidis, necessitating robust and sustainable supply chains for its key intermediates. Patent CN113277993A introduces a groundbreaking synthetic methodology that fundamentally shifts the production paradigm from hazardous multi-step sequences to a streamlined, two-step oxidative protocol. This innovation leverages the oxidative power of sodium periodate and potassium permanganate to construct the critical benzoxazole scaffold directly from readily available 6-amino-m-cresol and various aldehyde derivatives. By bypassing traditional acylation and diazo-based esterification steps, this technology addresses long-standing pain points regarding environmental safety and process complexity. For global procurement leaders and R&D directors, this represents a pivotal opportunity to secure a reliable API intermediate supplier capable of delivering high-quality materials through a greener, more economically viable pathway that aligns with modern sustainability mandates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Tafamidis and its analogues has relied on convoluted pathways that pose significant logistical and safety challenges for large-scale manufacturing. Traditional routes typically involve the acylation of 4-amino-3-hydroxybenzoic acid using dichlorobenzoyl chloride, a reagent known for its corrosivity and potential to generate hazardous waste streams. Furthermore, subsequent steps often require the use of (trimethylsilyl)diazomethane for esterification, a compound that is not only expensive but also poses severe explosion risks and toxicity concerns, demanding specialized handling infrastructure. The cumulative effect of these harsh reagents, combined with the need for strong acids and bases for cyclization and hydrolysis, results in a process with low atom economy and a burdensome impurity profile. These factors collectively drive up the cost of goods sold (COGS) and complicate regulatory compliance, creating friction in the supply chain for cost reduction in pharmaceutical manufacturing.
The Novel Approach
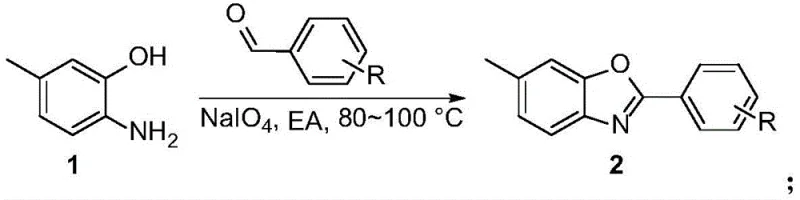
In stark contrast, the novel approach detailed in the patent utilizes a direct oxidative condensation strategy that dramatically simplifies the molecular construction of the benzoxazole core. By reacting 6-amino-m-cresol with substituted benzaldehydes in the presence of sodium periodate, the method achieves rapid cyclization under relatively mild thermal conditions (80-100°C) without the need for protecting groups or hazardous acyl chlorides. This is followed by a selective oxidation of the methyl group to a carboxylic acid using potassium permanganate, effectively installing the requisite pharmacophore in a single operation. This streamlined sequence not only reduces the total number of unit operations but also eliminates the generation of soluble toxic byproducts, as the reduced forms of the oxidants precipitate out of the reaction mixture. This technological leap provides a clear pathway for commercial scale-up of complex pharmaceutical intermediates, offering a distinct competitive advantage in terms of both operational safety and environmental footprint.
Mechanistic Insights into Oxidative Cyclization and Side-Chain Oxidation
The core of this synthetic breakthrough lies in the dual-oxidant system that orchestrates both ring formation and functional group transformation with high precision. In the first stage, sodium periodate (NaIO4) serves as the primary oxidant to facilitate the condensation between the amine and phenol moieties of the cresol substrate and the carbonyl carbon of the aldehyde. This oxidative cyclization proceeds through a mechanism that likely involves the formation of a Schiff base intermediate, which is subsequently oxidized to close the oxazole ring, releasing water and iodate species. The choice of ethyl acetate as a solvent is particularly strategic, as it provides excellent solubility for the organic substrates while allowing for easy separation of the inorganic iodate byproducts post-reaction. This step is remarkably tolerant of diverse electronic environments on the aldehyde ring, accommodating electron-withdrawing groups like halogens and trifluoromethyl substituents without compromising yield, ensuring versatility for derivative synthesis.
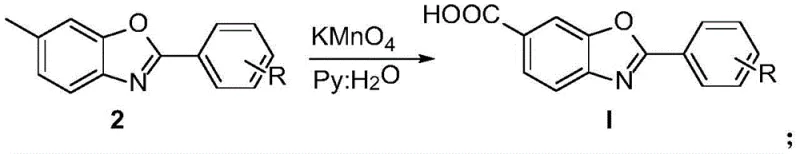
Following the construction of the heterocyclic core, the second stage employs potassium permanganate (KMnO4) in a pyridine-water mixture to effect the oxidation of the benzylic methyl group to a carboxylic acid. This transformation is critical for generating the final Tafamidis structure and is achieved under reflux conditions where the permanganate ion selectively attacks the benzylic position. The use of a biphasic or mixed solvent system involving pyridine helps to solubilize the organic intermediate while maintaining the oxidative strength of the permanganate. A key mechanistic advantage here is the insolubility of the resulting manganese dioxide (MnO2) precipitate, which allows for straightforward removal via filtration, thereby simplifying the workup procedure significantly compared to homogeneous oxidation methods. This rigorous control over oxidation states ensures that the sensitive benzoxazole ring remains intact while the side chain is fully converted, resulting in a high-purity product suitable for downstream API synthesis.
How to Synthesize Tafamidis Derivatives Efficiently
Implementing this synthesis requires precise control over stoichiometry and thermal parameters to maximize yield and minimize impurity formation. The process begins with the careful mixing of 6-amino-m-cresol and the chosen aldehyde in a molar ratio of approximately 1:2, alongside two equivalents of sodium periodate, ensuring complete conversion of the limiting amine reagent. The reaction is maintained at elevated temperatures (optimally 100°C) for a duration of 3 to 4 hours to drive the cyclization to completion. Upon cooling, the crude benzoxazole intermediate is isolated through standard extraction techniques, taking advantage of the phase separation between the organic solvent and the aqueous wash solutions. The subsequent oxidation step demands a slightly different solvent environment, utilizing a pyridine-water mix to support the permanganate oxidation, followed by acidification to precipitate or extract the final carboxylic acid product. For detailed operational parameters and safety protocols, please refer to the standardized guide below.
- Perform oxidative condensation of 6-amino-m-cresol with substituted benzaldehydes using sodium periodate (NaIO4) in ethyl acetate at 80-100°C to form the benzoxazole core.
- Oxidize the resulting 6-methylbenzoxazole intermediate using potassium permanganate (KMnO4) in a pyridine-water mixture under reflux conditions.
- Isolate the final Tafamidis derivative by acidification, extraction, and purification via column chromatography to ensure high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers profound benefits that extend beyond mere chemical efficiency, directly impacting the bottom line and supply resilience. By replacing exotic and hazardous reagents like dichlorobenzoyl chloride and diazomethane with commodity oxidants such as sodium periodate and potassium permanganate, the raw material cost structure is significantly optimized. These oxidants are globally available in bulk quantities, reducing the risk of supply disruptions that often plague specialized reagent markets. Furthermore, the elimination of strong acids and bases from the reaction matrix reduces the corrosion load on manufacturing equipment, extending asset life and lowering maintenance expenditures. The simplified workup, driven by the precipitation of inorganic byproducts, translates to reduced solvent consumption and shorter cycle times, which are critical factors for reducing lead time for high-purity pharmaceutical intermediates in a fast-paced market.
- Cost Reduction in Manufacturing: The economic viability of this route is underpinned by the drastic simplification of the synthetic sequence, which removes the need for expensive protection-deprotection strategies and hazardous reagent handling. Avoiding the use of (trimethylsilyl)diazomethane alone represents a substantial saving, as this reagent requires specialized storage and handling protocols that inflate operational costs. Additionally, the high atom economy of the oxidative cyclization means less waste is generated per kilogram of product, lowering waste disposal fees. The ability to recover and potentially recycle the insoluble iodate and manganese salts further enhances the process economics, creating a leaner manufacturing model that supports aggressive pricing strategies without sacrificing margin.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the reliance on widely sourced starting materials like 6-amino-m-cresol and various benzaldehydes, which are produced by multiple vendors globally. This diversification of the raw material base mitigates the risk of single-source bottlenecks that can halt production lines. The robustness of the reaction conditions, which tolerate a wide range of substituents, allows for flexible sourcing of aldehyde variants without requiring extensive process re-validation. Moreover, the stability of the intermediates and the final product ensures that inventory can be held safely, providing a buffer against market volatility and enabling just-in-time delivery models for downstream API manufacturers seeking a reliable API intermediate supplier.
- Scalability and Environmental Compliance: Scaling this process from laboratory to multi-ton production is facilitated by the heterogeneous nature of the byproduct formation, which simplifies filtration and isolation on a large scale. The absence of gaseous byproducts or highly exothermic runaway risks associated with diazo compounds makes the process inherently safer for large reactor vessels. From an environmental standpoint, the reduction in hazardous waste streams aligns with increasingly stringent global regulations on chemical manufacturing, easing the burden of environmental permitting and compliance reporting. This 'green' credential is increasingly valued by top-tier pharmaceutical companies who are mandating sustainable practices throughout their supply chains, making this route a future-proof choice for long-term partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative synthesis route for Tafamidis derivatives. These insights are derived directly from the experimental data and claims within patent CN113277993A, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding the nuances of reagent tolerance, workup procedures, and scalability is essential for R&D teams planning technology transfer. We encourage stakeholders to review these points to gain a comprehensive understanding of how this method compares to legacy processes in terms of efficiency, safety, and output quality.
Q: What are the primary safety advantages of this new Tafamidis synthesis route?
A: The process eliminates the use of hazardous dichlorobenzoyl chloride and explosive (trimethylsilyl)diazomethane, significantly reducing operational risks and environmental hazards associated with traditional acylation and esterification methods.
Q: How does the use of sodium periodate improve the reaction selectivity?
A: Sodium periodate acts as a mild yet effective oxidant that facilitates the direct oxidative cyclization of amino-cresols with aldehydes, tolerating various functional groups like halogens and trifluoromethyl groups without over-oxidation.
Q: Is this synthetic route scalable for industrial production of API intermediates?
A: Yes, the method utilizes commodity chemicals and generates insoluble byproducts (iodates and manganese dioxide) that are easily separated by filtration, simplifying downstream processing and making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tafamidis Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the oxidative route described in CN113277993A and is fully prepared to execute this methodology with the highest standards of quality and safety. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle oxidative chemistries safely, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of Tafamidis intermediate meets the exacting requirements of regulatory filings.
We invite you to collaborate with us to leverage this innovative synthesis for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in oxidative cyclization can drive value and reliability in your supply chain for high-purity cardiovascular drug intermediates.
