Scalable Synthesis of Baccatin III Derivatives for Commercial Anticancer Drug Production
The pharmaceutical industry continuously seeks robust and scalable pathways for the production of complex antineoplastic agents, particularly taxane derivatives which remain a cornerstone in cancer therapy. Patent CN101130528A discloses a highly efficient process for the preparation of 14-beta-hydroxy-1,14-carbonate-deacetylbaccatin III, a critical intermediate for novel taxane derivatives with potent antitumor activity. This technology addresses the longstanding bottleneck of raw material scarcity by utilizing 10-deacetylbaccatin III, a compound abundantly available from European yew leaves, as the starting material. By shifting the synthetic reliance from scarce natural extracts to a semi-synthetic route based on accessible precursors, this method significantly enhances the feasibility of commercial-scale manufacturing. The process involves a sophisticated sequence of selective protection, oxidation, and functional group manipulation that ensures high stereochemical control and purity, meeting the stringent requirements of global regulatory bodies for active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 14-beta-hydroxy baccatin III derivatives has been severely constrained by the reliance on natural extraction from specific species such as the Tibetan yew (Taxus wallichiana). The concentration of the necessary 14-beta-hydroxy-deacetylbaccatin III precursor in these natural sources is extremely low, making the extraction process economically unviable and environmentally taxing. Conventional methods often suffer from inconsistent yields due to the variability of plant material and the complexity of isolating the target compound from a matrix of structurally similar taxanes. Furthermore, the extensive purification required to remove plant impurities and isomers adds significant cost and time to the manufacturing timeline. This dependency on limited natural resources creates a fragile supply chain that is vulnerable to agricultural fluctuations and geopolitical instability, posing a significant risk to the continuous production of life-saving anticancer medications.
The Novel Approach
The innovative method described in the patent overcomes these limitations by establishing a synthetic route starting from 10-deacetylbaccatin III, which can be harvested in large quantities from the more common European yew (Taxus baccata). This strategic shift in starting material fundamentally alters the economics of production, providing a stable and abundant feedstock that supports large-scale industrial operations. The process employs a unique protection strategy using tribromoacetyl or trialkylsilyl groups that facilitate selective reactions at the C-13 and C-14 positions without affecting other sensitive functional groups on the baccatin skeleton. By enabling the transformation of a readily available precursor into the high-value 14-beta-hydroxy derivative through a controlled chemical sequence, this approach eliminates the need for difficult natural extraction. The result is a streamlined manufacturing process that offers superior consistency, higher overall yields, and a drastically reduced environmental footprint compared to traditional extraction-based methods.
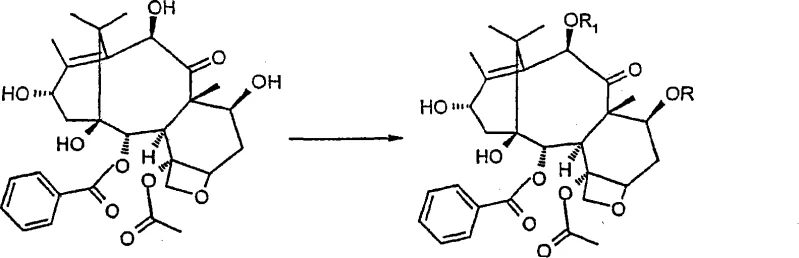
Mechanistic Insights into Selective Protection and Oxidation
The core of this synthesis lies in the precise manipulation of the baccatin III skeleton, beginning with the selective protection of the hydroxyl groups at the 7 and 10 positions. The reactivity of the hydroxyl groups follows a specific order, with C-7 and C-10 being more reactive towards acylation or silylation than C-1 and C-13. By exploiting this difference, the process selectively installs protecting groups such as tribromoacetyl esters, which serve a dual purpose: they protect the sensitive hydroxyls during subsequent oxidation steps and are designed to be easily removed later. The use of catalytic amounts of DMAP in the acylation step further enhances the efficiency of this protection, minimizing solvent waste and simplifying the workup procedure. This selective protection is crucial as it prevents unwanted side reactions at the C-7 and C-10 positions while leaving the C-13 position accessible for the critical oxidation step that follows.
Following protection, the synthesis proceeds with a two-step oxidation sequence at the C-13 position to introduce the hydroxyl group at the C-14 position. This transformation is achieved using manganese dioxide in solvents such as acetonitrile or acetone, which oxidizes the C-13 hydroxyl to a ketone and subsequently facilitates hydroxylation at the adjacent C-14 position. The reaction conditions are carefully controlled to ensure regioselectivity, preventing over-oxidation or degradation of the complex taxane core. Once the 14-beta-hydroxy group is established, a carbonate esterification is performed between the C-1 and C-14 hydroxyls using phosgene or triphosgene, creating a stable cyclic carbonate structure. The final steps involve the regioselective reduction of the C-13 carbonyl using sodium borohydride and the removal of the protecting groups, yielding the target 14-beta-hydroxy-1,14-carbonate-deacetylbaccatin III with high stereochemical purity.
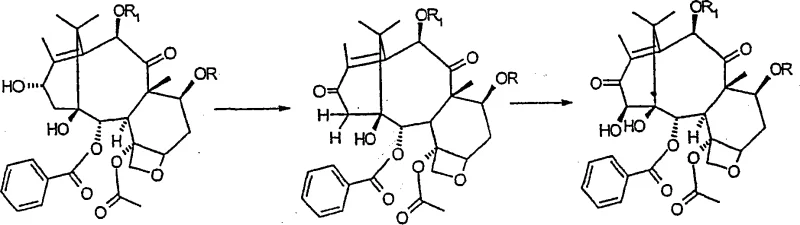
How to Synthesize 14-beta-hydroxy-1,14-carbonate-deacetylbaccatin III Efficiently
The synthesis of this critical taxane intermediate requires a disciplined approach to reaction conditions and reagent selection to ensure high yield and purity. The process begins with the dissolution of 10-deacetylbaccatin III in anhydrous methylene chloride, followed by the addition of trichloroacetic anhydride or trichloroacetyl chloride in the presence of a base like triethylamine. Careful temperature control during the protection phase is essential to prevent side reactions. Subsequent oxidation with activated manganese dioxide must be monitored closely via TLC to determine the optimal endpoint for the formation of the 13-dehydro derivative before proceeding to hydroxylation. The detailed standardized synthesis steps, including specific molar ratios, reaction times, and purification protocols, are outlined in the technical guide below to ensure reproducibility and safety in a manufacturing environment.
- Selectively protect the 7-position and 10-position hydroxyl groups of 10-deacetylbaccatin III using tribromoacetyl or trialkylsilyl reagents.
- Perform two-step oxidation at the 13-position using manganese dioxide to achieve hydroxylation at the 14-position.
- Execute carbonate esterification between the 1-position and 14-position hydroxyls, followed by reduction and deprotection to yield the final derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain leaders, the adoption of this synthetic route offers substantial strategic benefits that extend beyond mere technical feasibility. By decoupling production from the volatile supply of rare plant materials, manufacturers can secure a consistent and predictable flow of intermediates, thereby reducing the risk of production stoppages. The use of abundant European yew leaves as the starting material ensures that raw material costs remain stable and low, shielding the supply chain from the price spikes associated with scarce natural resources. Furthermore, the chemical steps involved utilize common, industrially available reagents such as manganese dioxide and sodium borohydride, which are easy to source in bulk quantities. This accessibility simplifies vendor management and reduces the complexity of the supply chain, allowing for more agile responses to market demand fluctuations.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive transition metal catalysts and complex purification steps associated with natural extraction, leading to significant operational cost savings. The use of tribromoacetyl protecting groups allows for simultaneous deprotection during the reduction step, which reduces the total number of unit operations and solvent consumption. This streamlining of the synthetic pathway directly translates to lower manufacturing costs per kilogram, enhancing the overall profitability of the final drug product without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: Relying on 10-deacetylbaccatin III from European yew provides a much more robust raw material base compared to the scarce Tibetan yew derivatives. This abundance ensures that production schedules can be maintained consistently, even during periods of high global demand for taxane-based therapies. The synthetic nature of the process also means that production is not subject to seasonal agricultural constraints, allowing for year-round manufacturing capabilities that strengthen the reliability of supply to downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The reaction conditions are mild and utilize solvents that are manageable within standard industrial waste treatment frameworks, facilitating easier compliance with environmental regulations. The high selectivity of the reactions minimizes the formation of by-products, reducing the burden on waste disposal systems and lowering the environmental impact of the manufacturing process. This scalability ensures that the process can be seamlessly transferred from pilot plant to full commercial production, supporting the growing global need for affordable anticancer medications.
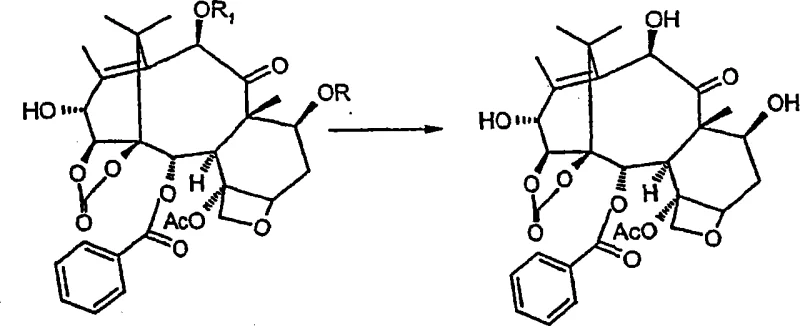
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these baccatin III derivatives. The answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the process capabilities and limitations. Understanding these details is essential for R&D teams evaluating the feasibility of this route for their specific product pipelines and for procurement teams assessing the long-term viability of the supply source.
Q: What is the primary advantage of using 10-deacetylbaccatin III over natural extraction for Baccatin III derivatives?
A: The primary advantage lies in the abundance of the starting material. 10-deacetylbaccatin III can be extracted in large quantities from European yew (Taxus baccata) leaves, whereas traditional methods rely on scarce 14-beta-hydroxy-baccatin III precursors from Tibetan yew, ensuring a more reliable and scalable supply chain for commercial manufacturing.
Q: How does the tribromoacetyl protection group improve the synthesis process?
A: The use of tribromoacetyl esters as protecting groups offers significant advantages during oxidation, carbonate esterification, and reduction steps. These groups are stable under reaction conditions yet can be removed simultaneously during the final reduction step, simplifying the workflow and reducing the number of purification stages required.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is designed for scalability. It utilizes common reagents like manganese dioxide and sodium borohydride, avoids expensive transition metal catalysts, and employs robust reaction conditions that are easily transferable from laboratory to commercial scale, ensuring consistent quality and yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Baccatin III Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable intermediates in the development of life-saving oncology drugs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in patent CN101130528A can be executed with precision at any volume. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Baccatin III derivatives meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of taxane synthesis, delivering high-quality intermediates that support your drug development and commercialization goals.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through advanced synthetic routes. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact us to request specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your project requirements. By partnering with us, you gain access to a wealth of chemical expertise and a supply chain dedicated to reliability and quality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
