Advanced Semi-Synthetic Route for High-Purity Docetaxel Production
Introduction to Next-Generation Docetaxel Manufacturing
The global demand for effective oncology therapeutics continues to drive innovation in the synthesis of complex taxane derivatives, specifically Docetaxel. As detailed in the groundbreaking patent CN102887876A, a novel semi-synthetic methodology has been developed that fundamentally alters the economic and technical landscape of producing this critical active pharmaceutical ingredient. This invention leverages 10-deacetylbaccatin III (10-DAB) as the foundational scaffold, introducing a highly efficient photo-rotation side chain strategy that bypasses the traditional bottlenecks associated with chiral resolution and low-yield coupling reactions. By shifting the paradigm from extraction-dependent supply chains to robust chemical synthesis, this technology offers a sustainable pathway for securing the global supply of anti-cancer medications while preserving endangered Taxus plant resources.
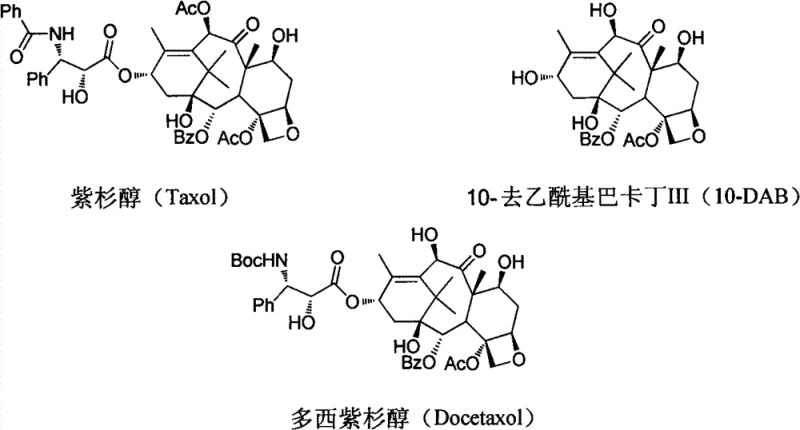
The core innovation lies in the strategic modification of the C-13 side chain and the protective group strategy on the baccatin core. Unlike conventional methods that struggle with isomer contamination and harsh reaction conditions, this improved process utilizes a unique oxazoline intermediate derived from tribromoacetaldehyde. This specific chemical architecture not only enhances the stereochemical fidelity of the final product but also drastically simplifies the purification workflow. For pharmaceutical manufacturers and procurement strategists, understanding the nuances of this patent is essential for evaluating potential partners who can deliver high-purity API intermediates with superior cost-efficiency and supply continuity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Docetaxel has been plagued by significant technical hurdles that inflate costs and compromise supply stability. Traditional routes often rely on the coupling of straight-chain isoserine side chains or four-membered azetidinone rings with the 10-DAB nucleus. These legacy processes are characterized by an excessive number of synthetic steps, each introducing opportunities for yield loss and impurity generation. Furthermore, the reliance on chiral resolution to separate enantiomers adds substantial complexity, requiring expensive chiral columns or resolving agents that are difficult to recycle. The resulting intermediates are frequently unstable, necessitating stringent storage conditions and rapid processing, which creates logistical friction in large-scale manufacturing environments.
Beyond the technical complexities, the economic implications of these conventional methods are severe. The low overall yields mean that a significant portion of the valuable 10-DAB starting material is wasted, driving up the raw material cost per kilogram of final API. Additionally, the use of harsh reagents and extreme reaction conditions often leads to the formation of difficult-to-remove by-products, forcing manufacturers to invest heavily in downstream purification technologies. For procurement managers, these factors translate into volatile pricing and long lead times, as production batches are more prone to failure or require extended quality control testing to ensure compliance with rigorous pharmacopeial standards.
The Novel Approach
The methodology disclosed in CN102887876A represents a decisive break from these inefficient traditions by introducing a streamlined, high-yield synthetic route. The cornerstone of this innovation is the use of (2R,3S)-3-phenylisoserine methyl ester reacting directly with tribromoacetaldehyde to form a stable oxazoline intermediate. This cyclization step is catalyzed efficiently by p-toluenesulfonic acid pyridinium salt in solvents like toluene or DMF, proceeding under relatively mild thermal conditions. This approach effectively locks the stereochemistry early in the synthesis, eliminating the need for later-stage chiral separations and ensuring that the optical purity of the side chain is maintained throughout the subsequent coupling reactions.
Moreover, the novel process incorporates a sophisticated protecting group strategy using trichloroacetyl groups for the 7-OH and 10-OH positions of the 10-DAB core. This specific choice of protecting groups facilitates smoother coupling reactions and allows for selective deprotection in the final stages without compromising the integrity of the sensitive taxane skeleton. The result is a process that is not only chemically elegant but also operationally robust, featuring fewer unit operations, simpler work-up procedures, and significantly higher throughput. For supply chain leaders, this translates to a more predictable production schedule and a reliable source of high-quality Docetaxel that meets the escalating demands of the global oncology market.
Mechanistic Insights into Oxazoline-Mediated Side Chain Synthesis
The mechanistic superiority of this process centers on the formation and reactivity of the tribromo-substituted oxazoline ring. In the initial step, the amino group of the phenylisoserine ester attacks the carbonyl carbon of tribromoacetaldehyde, followed by intramolecular cyclization involving the adjacent hydroxyl group. The presence of the three bromine atoms at the C-2 position of the oxazoline ring exerts a strong electron-withdrawing effect, which stabilizes the ring structure against premature hydrolysis while simultaneously activating it for the subsequent nucleophilic attack by the 13-hydroxyl group of the baccatin core. This electronic tuning is critical; it ensures that the side chain remains intact during the coupling phase but can be cleanly opened under specific acidic conditions to reveal the free amine required for the final biological activity.
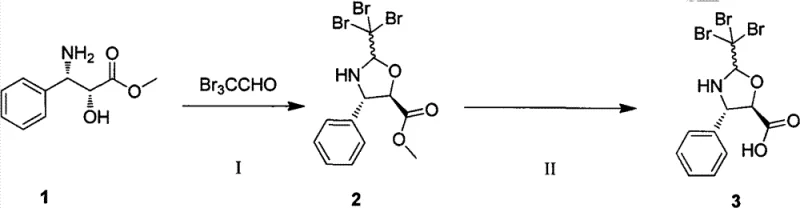
Comparative analysis with prior art, such as patent KR101032761B which utilizes a trichloromethyl substituent, highlights the profound impact of halogen selection on reaction kinetics and thermodynamics. The patent data explicitly demonstrates that replacing chlorine with bromine boosts the side chain synthesis yield from 81% to an impressive 89%. This increase is not merely incremental; it reflects a fundamental improvement in the transition state energy profile of the cyclization reaction. Furthermore, the total synthesis yield for Docetaxel using this tribromo-route reaches 82%, surpassing the 72% benchmark of previous methods. This mechanistic optimization directly correlates to reduced waste generation and lower solvent consumption, aligning with modern green chemistry principles.
Impurity control is another area where this mechanism excels. The rigid geometry of the oxazoline intermediate prevents the epimerization of the chiral centers, a common issue in linear side chain syntheses. By avoiding the formation of diastereomeric impurities, the process simplifies the purification burden significantly. The final crystallization steps are more efficient, yielding Docetaxel with a purity exceeding 99.6%, as confirmed by HPLC analysis in the patent examples. For R&D directors, this level of stereochemical control is paramount, as it ensures that the final drug product possesses the precise molecular configuration required for optimal binding to microtubules and maximum therapeutic efficacy.
How to Synthesize Docetaxel Efficiently
Implementing this advanced semi-synthetic route requires precise adherence to the reaction parameters outlined in the patent to maximize yield and purity. The process begins with the preparation of the specialized oxazoline side chain, followed by the protection of the 10-DAB nucleus, coupling, ring opening, amino protection, and final deprotection. Each step has been optimized to balance reaction rate with selectivity, ensuring that the complex polycyclic structure of the taxane is preserved. The following guide summarizes the critical operational phases derived from the experimental examples provided in the intellectual property documentation.
- Condense (2R,3S)-3-phenylisoserine methyl ester with tribromoacetaldehyde using PPTs catalyst to form the oxazoline intermediate.
- Protect the 7-OH and 10-OH positions of 10-DAB with trichloroacetyl groups under anhydrous conditions.
- Couple the protected 10-DAB with the side chain, followed by ring opening, Boc protection, and final ammonolysis to yield Docetaxel.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this improved semi-synthetic method offers tangible strategic benefits that extend beyond simple chemical yield. The elimination of complex chiral resolution steps and the reduction in total synthetic steps fundamentally alter the cost structure of Docetaxel manufacturing. By removing the need for expensive chiral stationary phases and reducing the number of isolation events, the process significantly lowers the variable cost of goods sold. This efficiency gain allows suppliers to offer more competitive pricing models, providing pharmaceutical buyers with greater flexibility in budgeting for their oncology portfolios while maintaining healthy margins.
- Cost Reduction in Manufacturing: The substantial increase in overall yield, rising from industry benchmarks of roughly 72% to over 82%, implies a drastic reduction in raw material waste. Since 10-DAB is a precious natural product derivative, maximizing its conversion into the final API is the single most effective lever for cost control. Furthermore, the use of common, low-toxicity reagents like formic acid and ammonia in the later stages reduces the expenditure on hazardous waste disposal and specialized containment infrastructure. These cumulative efficiencies create a leaner manufacturing process that is inherently more cost-effective than legacy routes.
- Enhanced Supply Chain Reliability: The robustness of the oxazoline intermediate contributes to greater supply chain resilience. Unlike unstable linear side chains that require immediate use or cryogenic storage, the cyclic oxazoline derivative is stable and can be stockpiled, decoupling the side chain synthesis from the final coupling step. This flexibility allows manufacturers to buffer against fluctuations in raw material availability and smooth out production schedules. Consequently, lead times for high-purity pharmaceutical intermediates are reduced, ensuring that downstream drug product manufacturers receive their materials on time, every time.
- Scalability and Environmental Compliance: The mild reaction conditions described in the patent, such as room temperature hydrolysis and ambient pressure couplings, facilitate seamless scale-up from pilot plants to multi-ton commercial reactors. There is no reliance on exotic high-pressure equipment or extreme cryogenic temperatures, which lowers the capital expenditure barrier for manufacturing partners. Additionally, the simplified work-up procedures, often involving standard extractions and crystallizations, minimize solvent usage and energy consumption. This alignment with environmental, social, and governance (ESG) goals makes the supply chain more sustainable and compliant with increasingly strict global regulatory standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this semi-synthetic technology. The answers are derived directly from the experimental data and claims presented in patent CN102887876A, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this route into their existing supply networks.
Q: What is the primary advantage of using tribromoacetaldehyde over trichloroacetaldehyde?
A: According to patent CN102887876A, substituting the trichloromethyl group with a tribromomethyl group increases the side chain synthesis yield from 81% to 89% and improves overall purity.
Q: How does this method improve supply chain reliability for Docetaxel?
A: The process eliminates difficult chiral resolution steps and utilizes mild reaction conditions, significantly reducing production lead times and ensuring consistent batch-to-batch quality.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the method features simple operational steps, low toxicity reagents, and easy post-treatment purification, making it highly adaptable for industrial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Docetaxel Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and scalable infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields demonstrated in patent literature can be replicated consistently in a GMP environment. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of Docetaxel intermediate meets the exacting standards required for oncology drug formulation.
We invite you to engage with our technical procurement team to discuss how this improved semi-synthetic route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this high-yield methodology. We are prepared to provide specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless integration of high-quality Docetaxel intermediates into your manufacturing pipeline.
