Scalable Heterogeneous Catalysis for High-Purity 2-Trifluoromethyl Benzimidazole Intermediates
Scalable Heterogeneous Catalysis for High-Purity 2-Trifluoromethyl Benzimidazole Intermediates
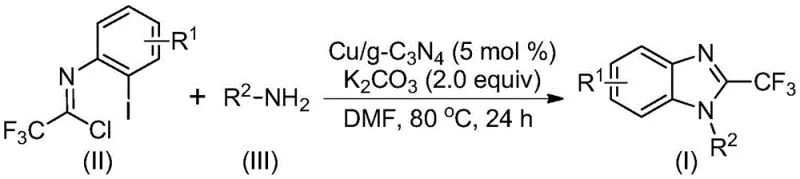
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing nitrogen-containing heterocycles, particularly those functionalized with fluorine atoms to enhance metabolic stability and bioavailability. Patent CN113429349A introduces a groundbreaking preparation method for 2-trifluoromethyl substituted benzimidazole compounds, utilizing a heterogeneous copper-doped carbon nitride catalyst. This technology represents a significant leap forward in synthetic efficiency, addressing long-standing challenges regarding catalyst recovery and substrate versatility. By leveraging cheap and easily obtainable starting materials such as trifluoroethylimidoyl chloride and various amines, this process offers a streamlined pathway to high-value intermediates. The innovation lies not just in the chemical transformation itself, but in the engineering of a catalytic system that balances high reactivity with the practical necessities of industrial manufacturing, such as recyclability and simplified downstream processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted benzimidazoles has been plagued by significant operational hurdles that impede large-scale production. Traditional routes often rely on the condensation of o-phenylenediamine with trifluoroacetic acid or trifluoroacetaldehyde, which frequently necessitates harsh reaction conditions and generates substantial waste. Alternative strategies involving the reductive cyclization of o-nitroanilines or direct trifluoromethylation of existing benzimidazole cores often suffer from narrow substrate scopes and the requirement for expensive, specialized reagents. Furthermore, earlier reports on copper-catalyzed cascade cyclizations utilized homogeneous catalytic systems. While effective in small-scale laboratory settings, homogeneous copper catalysts present a severe bottleneck for commercial scale-up due to the difficulty in removing trace metal residues from the final active pharmaceutical ingredient (API), a process that is both costly and time-consuming.
The Novel Approach
The methodology disclosed in the patent data circumvents these legacy issues by employing a heterogeneous copper-doped carbon nitride (Cu/g-C3N4) catalyst. This novel approach facilitates a cascade cyclization reaction between trifluoroethylimidoyl chloride and amines under mild thermal conditions, typically ranging from 70°C to 90°C. Unlike its homogeneous counterparts, this solid-state catalyst can be effortlessly separated from the reaction mixture through simple filtration, thereby eliminating the need for complex metal scavenging procedures. The process demonstrates exceptional tolerance for various functional groups on the aromatic ring, including halogens and alkyl groups, allowing for the synthesis of a diverse array of derivatives. This shift from homogeneous to heterogeneous catalysis fundamentally alters the economic and environmental profile of the synthesis, making it highly attractive for cost-sensitive manufacturing environments.
Mechanistic Insights into Cu/g-C3N4 Catalyzed Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of nucleophilic substitution and transition metal catalysis. Initially, the trifluoroethylimidoyl chloride and the amine undergo a nucleophilic addition-elimination reaction to form an amidine intermediate in situ. Subsequently, the copper species embedded within the carbon nitride matrix inserts into the carbon-iodine bond of the aryl ring, forming a transient organocopper complex. Under the basic conditions provided by potassium carbonate, a molecule of hydrogen iodide is eliminated, prompting the formation of a bivalent or trivalent cyclic copper intermediate. The final step involves a reductive elimination that closes the imidazole ring, releasing the desired 2-trifluoromethyl-substituted benzimidazole product and regenerating the active copper catalytic site. This cycle ensures high turnover numbers and consistent product quality throughout the reaction duration.
From a purity and impurity control perspective, the heterogeneous nature of the catalyst plays a pivotal role. In homogeneous systems, leached copper ions can coordinate with product molecules or side products, leading to difficult-to-remove impurities that complicate the purification profile. By confining the copper active sites within the g-C3N4 support, the patent describes a system where metal leaching is minimized, and the catalyst remains physically distinct from the soluble organic products. This physical separation capability is crucial for maintaining stringent purity specifications required by regulatory bodies. Additionally, the use of mild bases like potassium carbonate prevents the degradation of sensitive functional groups that might occur under stronger alkaline conditions, further ensuring a clean impurity profile and high isolated yields, often exceeding 90% for optimized substrates.
How to Synthesize 2-Trifluoromethyl Benzimidazole Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for replicating this high-efficiency transformation in a laboratory or pilot plant setting. The procedure is designed to be operationally simple, requiring standard equipment such as Schlenk tubes or stirred tank reactors capable of maintaining temperatures up to 90°C. The key to success lies in the precise stoichiometric balance of the reagents, specifically maintaining a molar ratio where the amine is in slight excess relative to the trifluoroethylimidoyl chloride to drive the equilibrium forward. The detailed standardized synthesis steps below outline the exact quantities and conditions required to achieve the reported high yields and purity levels.
- Mix copper-doped carbon nitride (5 mol %), potassium carbonate (2.0 equiv), trifluoroethylimidoyl chloride, and amine in an organic solvent such as DMF.
- Heat the reaction mixture to a temperature between 70°C and 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture to recover the heterogeneous catalyst, mix the filtrate with silica gel, and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this heterogeneous catalytic process translates directly into tangible operational improvements and risk mitigation. The shift away from homogeneous catalysis removes one of the most expensive and variable steps in pharmaceutical manufacturing: heavy metal removal. By utilizing a filterable solid catalyst, the process drastically simplifies the workup procedure, reducing the consumption of silica gel, solvents, and specialized scavenging resins. This simplification not only lowers the direct material costs but also shortens the overall production cycle time, allowing for faster throughput and improved responsiveness to market demands. The reliance on commercially available and inexpensive starting materials further stabilizes the supply chain against raw material price volatility.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of downstream purification bottlenecks. In traditional homogeneous copper catalysis, removing trace metals to meet ppm-level specifications often requires multiple chromatography steps or the use of expensive functionalized silicas. By employing a heterogeneous Cu/g-C3N4 catalyst that can be removed via simple filtration, manufacturers can significantly reduce the operational expenditure associated with purification. Furthermore, the patent data indicates that the catalyst retains substantial activity even after multiple recycling runs, which amortizes the initial catalyst cost over several batches, leading to a lower cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain continuity by reducing dependency on exotic or hard-to-source reagents. The starting materials, including trifluoroethylimidoyl chloride and various alkyl or aryl amines, are commodity chemicals available from multiple global suppliers. This multi-sourcing potential mitigates the risk of supply disruptions caused by single-vendor dependencies. Additionally, the mild reaction conditions (70-90°C) and the use of common solvents like DMF mean that the process can be executed in standard glass-lined or stainless steel reactors without requiring specialized high-pressure or cryogenic equipment, thereby increasing the number of qualified contract manufacturing organizations (CMOs) capable of producing the material.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this methodology is inherently designed for scalability. The heterogeneous catalyst minimizes the generation of heavy metal-contaminated waste streams, simplifying wastewater treatment and disposal compliance. The ability to recycle the catalyst reduces the total volume of solid waste generated per unit of product. Moreover, the high atom economy of the cascade cyclization, combined with high conversion rates, ensures that raw materials are efficiently utilized, minimizing the formation of by-products that would otherwise require energy-intensive separation processes. This aligns perfectly with modern green chemistry initiatives and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical disclosures found within the patent documentation, providing a reliable basis for feasibility assessments. Understanding these nuances is critical for R&D teams evaluating this route for potential technology transfer and for procurement teams assessing the long-term viability of the supply source.
Q: What are the primary advantages of using a heterogeneous catalyst in this synthesis?
A: The use of copper-doped carbon nitride (Cu/g-C3N4) allows for easy separation of the catalyst from the reaction mixture via simple filtration. Furthermore, the patent data indicates that this heterogeneous catalyst can be recycled and reused multiple times with only a partial reduction in catalytic efficiency, significantly lowering material costs compared to homogeneous systems.
Q: What is the substrate scope for the amine component in this reaction?
A: The method demonstrates broad substrate tolerance. The amine component (R2-NH2) can be varied to include C3 to C6 alkyl groups (such as n-butyl, n-pentyl, isopropyl), as well as aryl groups like phenyl and benzyl. This flexibility allows for the design of diverse benzimidazole libraries suitable for various drug discovery programs.
Q: What are the typical reaction conditions required for this transformation?
A: The reaction proceeds under relatively mild thermal conditions, typically requiring temperatures between 70°C and 90°C. The process utilizes potassium carbonate as a base and DMF as the preferred organic solvent, reacting for a period of 18 to 30 hours to achieve high yields, often exceeding 90% for optimized substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, scalable, and compliant manufacturing processes in the modern pharmaceutical landscape. The technology described in patent CN113429349A aligns perfectly with our commitment to delivering high-quality intermediates through innovative chemistry. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl benzimidazole meets the exacting standards required for drug substance manufacturing.
We invite potential partners to leverage our technical expertise to optimize their supply chains. By adopting this advanced heterogeneous catalytic route, your organization can achieve significant process intensification and cost efficiencies. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our team is ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can become your strategic partner in the reliable supply of high-purity pharmaceutical intermediates.
