Advanced Manufacturing of Erexib Intermediates: A Green, Scalable Route for Global Pharma Supply Chains
Advanced Manufacturing of Erexib Intermediates: A Green, Scalable Route for Global Pharma Supply Chains
The pharmaceutical industry constantly seeks robust, scalable, and environmentally sustainable pathways for the production of critical anti-inflammatory agents. Patent CN107586268B, published in early 2021, introduces a transformative preparation method for Erexib and its key intermediates, addressing long-standing inefficiencies in traditional synthesis routes. Erexib, chemically known as N-N-propyl-3-(4-methylphenyl)-4-(4-methylsulfonylphenyl)-2,5-dihydropyrrole-2-one, functions as a highly selective COX-2 inhibitor, offering potent anti-inflammatory effects while minimizing gastrointestinal side effects associated with non-selective inhibitors. The core innovation of this patent lies in the strategic replacement of hazardous acid chloride activation with modern condensing agents, specifically carbonyldiimidazole (CDI), which dramatically enhances both reaction yield and product purity. This technical breakthrough not only optimizes the chemical efficiency but also aligns with modern green chemistry principles by eliminating corrosive reagents and simplifying purification protocols.
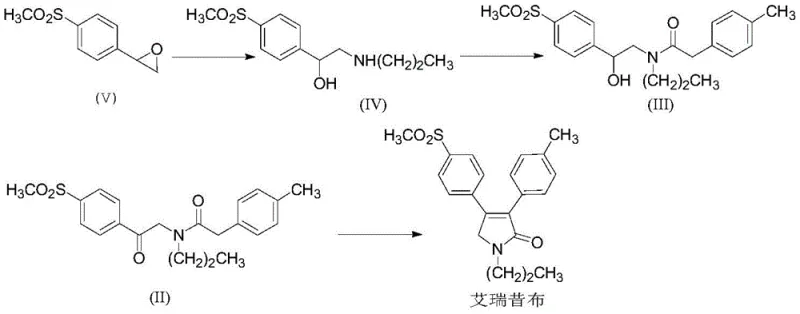
For procurement managers and supply chain directors, the implications of this methodology are profound. By shifting away from thionyl chloride (SOCl2) and avoiding complex column chromatography steps, the process significantly reduces operational hazards and waste generation. The patent details a comprehensive four-step sequence starting from p-methylsulfonylphenyl oxirane, proceeding through nucleophilic ring opening, acylation, oxidation, and finally cyclization. This streamlined approach ensures that the production of high-purity pharmaceutical intermediates can be achieved with greater consistency and lower environmental impact, making it an ideal candidate for reliable API intermediate supplier partnerships aiming for long-term commercial viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Erexib intermediates has relied heavily on the activation of carboxylic acids using thionyl chloride to form reactive acid chlorides. As disclosed in prior art such as CN1134413C, this conventional approach suffers from multiple critical drawbacks that hinder industrial scalability. Firstly, the use of SOCl2 introduces severe safety and environmental concerns due to its corrosive nature and the release of toxic sulfur dioxide and hydrogen chloride gases during the reaction. Secondly, the acylation step using these acid chlorides typically results in suboptimal yields, often hovering around 61 percent, coupled with product purities as low as 83.6 percent. Furthermore, alternative coupling agents like DCC (dicyclohexylcarbodiimide) generate dicyclohexylurea (DCU) as a byproduct, which is notoriously difficult to remove completely from the reaction mixture, thereby complicating downstream purification and increasing production costs. These factors collectively create a bottleneck for manufacturers seeking cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The novel approach detailed in patent CN107586268B fundamentally reengineers the acylation step by employing advanced condensing agents such as CDI, EDCI, or DIC. This method bypasses the need for unstable and hazardous acid chlorides entirely. In the optimized protocol, 4-methylphenylacetic acid is activated in situ using CDI in tetrahydrofuran at moderate temperatures (50°C), followed by the addition of the amine intermediate. This modification results in a staggering improvement in performance, with yields reaching up to 98 percent and purities exceeding 99.0 percent. The elimination of DCU byproducts and the avoidance of corrosive gases streamline the workup procedure, allowing for simple aqueous washes and solvent evaporation rather than complex chromatographic separations. This represents a paradigm shift towards safer, higher-yielding chemical manufacturing.
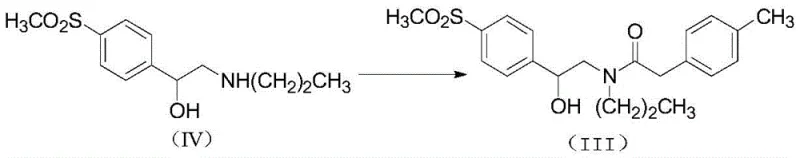
Mechanistic Insights into CDI-Mediated Amide Coupling and Oxidative Cyclization
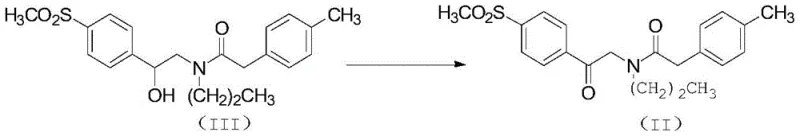
The mechanistic elegance of this synthesis lies in the precise control of reactivity at each stage. The acylation mechanism involves the nucleophilic attack of the carboxylic acid on the carbonyl carbon of CDI, forming a reactive acyl-imidazole intermediate. This species is highly electrophilic yet stable enough to be handled without immediate decomposition, allowing for a controlled reaction with the amine nucleophile (Compound IV). The leaving group, imidazole, is water-soluble and easily removed during the aqueous workup, contributing to the high purity of the resulting amide (Compound III). Following acylation, the synthesis proceeds to an oxidation step where the secondary alcohol moiety is converted to a ketone. The patent specifies the use of Jones reagent (chromium trioxide in sulfuric acid) or pyridine-chromium trioxide complexes. This oxidation is critical as it sets up the electronic environment necessary for the subsequent ring closure, transforming the flexible amide chain into a rigid precursor capable of cyclization.
The final mechanistic step is an intramolecular cyclization driven by base catalysis. Under alkaline conditions, typically using potassium carbonate (K2CO3) in an ethanol-water mixture, the alpha-proton adjacent to the newly formed ketone is deprotonated to form an enolate. This nucleophilic enolate then attacks the adjacent amide carbonyl carbon, displacing the amine leaving group and closing the five-membered pyrrolone ring. This cyclization is thermodynamically favorable and proceeds efficiently under reflux conditions. The patent highlights that this base-mediated cyclization significantly improves yield compared to previous methods, and the final product can be purified simply by recrystallization from ethanol, avoiding the need for silica gel column chromatography which is impractical for multi-kilogram production. This mechanistic clarity ensures reproducibility and robustness in a commercial setting.
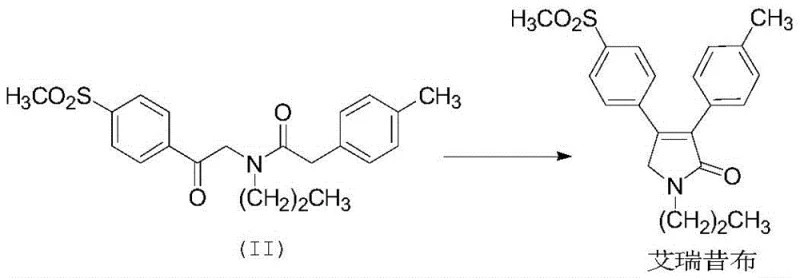
How to Synthesize Erexib Efficiently
The synthesis of Erexib described in this patent offers a clear, four-stage pathway that balances chemical efficiency with operational simplicity. The process begins with the ring-opening of an epoxide, followed by the critical amide coupling, oxidation, and final cyclization. Each step has been optimized to minimize impurities and maximize throughput, making it an attractive route for contract development and manufacturing organizations (CDMOs). The use of common solvents like methanol, tetrahydrofuran, and ethanol further enhances the practicality of this method, as these materials are readily available and easy to recover. For technical teams looking to implement this route, the key lies in maintaining strict temperature control during the exothermic ring-opening and ensuring complete activation of the acid during the coupling phase.
- Perform nucleophilic ring opening of p-methylsulfonylphenyl oxirane with n-propylamine to form the hydroxy-amine intermediate (IV).
- Execute amide coupling between intermediate (IV) and 4-methylphenylacetic acid using CDI as a condensing agent to yield compound (III) with high purity.
- Oxidize the secondary alcohol in compound (III) to a ketone using Jones reagent to obtain the diketo precursor (II).
- Complete the synthesis via intramolecular cyclization of compound (II) under alkaline conditions (e.g., K2CO3) to form the final pyrrolone ring of Erexib.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers substantial strategic benefits for procurement and supply chain stakeholders. The primary advantage is the drastic simplification of the purification process. By eliminating the need for column chromatography, which is solvent-intensive and time-consuming, manufacturers can significantly reduce processing time and solvent disposal costs. This directly translates to enhanced supply chain reliability, as batches can be turned around faster with consistent quality. Furthermore, the replacement of thionyl chloride with solid condensing agents like CDI improves workplace safety and reduces the regulatory burden associated with handling hazardous corrosive materials. This alignment with green chemistry standards not only mitigates environmental risk but also future-proofs the supply chain against increasingly stringent environmental regulations.
- Cost Reduction in Manufacturing: The shift to high-yield condensing agents eliminates the material losses associated with low-yielding acid chloride reactions. By achieving yields near quantitative levels (98 percent), the effective cost per kilogram of the active intermediate is drastically lowered. Additionally, the removal of expensive and labor-intensive chromatographic purification steps reduces operational expenditures related to silica gel, solvents, and labor hours. The ability to use simple recrystallization for purification further drives down variable costs, making the overall process economically superior to legacy methods.
- Enhanced Supply Chain Reliability: The starting material, p-methylsulfonylphenyl oxirane, is derived from thioanisole, a commodity chemical with a stable global supply. The robust nature of the synthetic steps, particularly the tolerance of the condensing agent method to minor variations, ensures consistent batch-to-batch quality. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream pharmaceutical clients. The simplified workflow also reduces the risk of production delays caused by complex purification bottlenecks.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations such as reflux, filtration, and crystallization that are easily transferable from pilot plant to commercial scale. The avoidance of heavy metal catalysts in the coupling step and the minimization of hazardous gas emissions contribute to a cleaner manufacturing footprint. This facilitates easier compliance with environmental, health, and safety (EHS) standards, reducing the likelihood of regulatory shutdowns and ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of adopting this technology for large-scale production.
Q: How does the new acylation method improve yield compared to traditional acid chloride routes?
A: The patented method replaces corrosive thionyl chloride (SOCl2) activation with carbonyldiimidazole (CDI) coupling. This shift eliminates the formation of difficult-to-remove urea byproducts associated with DCC and avoids the low yields (61%) typical of acid chloride methods, achieving yields up to 98% with 99% purity.
Q: What purification advantages does this process offer for large-scale manufacturing?
A: Unlike prior art requiring complex column chromatography, this process utilizes simple recrystallization techniques using common solvents like ethanol or acetone. This significantly simplifies post-treatment, reduces solvent consumption, and enhances suitability for industrial scale-up.
Q: Is the starting material p-methylsulfonylphenyl oxirane commercially viable?
A: Yes, the starting material can be efficiently synthesized from readily available thioanisole through a established four-step sequence involving acylation, oxidation, bromination, and reduction, ensuring a stable and continuous supply chain for the final API intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Erexib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, high-quality intermediate synthesis in the global pharmaceutical landscape. Our team of expert chemists has extensively analyzed the pathway disclosed in CN107586268B and possesses the technical capability to execute this advanced route with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Erexib intermediate meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this innovative technology for your drug development programs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can drive value and efficiency in your supply chain.
